Maintenance for Implants Based On Diagnostic Categories
A patient-specific maintenance regimen plays a pivotal role in the success of the dental implant process.
This course was published in the December 2019 issue and expires December 2022. John B. Wilson, DDS, MS, has no commercial conflicts of interest to disclose. Although not in conjunction with this article, Thomas G. Wilson Jr., DDS, has previously received grants, research support and honorarium from Straumann and Osteogenics This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Explain the role of the maintenance visit within the context of the overall dental implant process.
- Describe key elements of a dental implant maintenance visit.
- List clinical signs of peri-implant disease.
Dental implants are the optimal method for replacing missing teeth in the vast majority of cases. A primary reason is that when an individualized maintenance program is included in the dental implant process, implants offer excellent long-term success rates. However, implant failures can occur for a number of reasons, including a lack of oral hygiene and/or inadequate maintenance.
Teeth and implant issues share many similar etiologies. Therefore, a typical implant maintenance visit is similar in many ways to a maintenance visit for natural teeth. Because oral biofilm is associated with implant loss, clinical emphasis should be placed on evaluating the peri-implant tissues for the presence of inflammation and/or bone loss. If disease is diagnosed around an implant, appropriate therapy should be provided to reduce the probability of failure. While there is controversy concerning some aspects of maintenance care, most clinicians would agree that routine radiographic evaluation, occlusal analysis, reduction of biomass burden, and personal oral hygiene instruction should be part of each maintenance visit.
The efficacy of maintenance in the dental implant process has been strongly substantiated in the dental literature.1–7 This article will detail current approaches to maintenance for patients with dental implants.
EXTRAORAL AND INTRAORAL EXAMINATIONS
The health of the patient can directly affect dental implant health. Therefore, a typical maintenance visit starts with an overview of the patient’s medical history. This includes any changes in health, hospitalizations, and medication history. The patient’s vital signs (including blood pressure) should be recorded. Any concerns or changes in the patient’s dental or medical history should also be noted.
Because occlusal relationships can affect the longevity of implants, an extraoral examination — including evaluation of the temporomandibular joints and muscles of mastication — is prudent. This is followed by a thorough intraoral examination to check for any abnormalities. The fact these examinations were performed and any positive findings should also be documented.
During the dental implant maintenance examination, tooth mobility is determined using two methods: bidigital mobility (Figure 1), and fremitus (i.e., tooth movement in function). Fremitus is often seen when no tooth mobility is evident using the bidigital approach. Restorations are evaluated for fractures, marginal integrity and movement, and the teeth are examined for evidence of dental caries. The examination and any positive findings should be carefully recorded. Implant mobility without pain is usually indicative of a problem with the crown or abutment, while intense pain usually indicates implant failure.
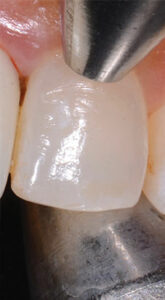
ends of two instruments
are used to determine
tooth mobility
PROBING
It is difficult to detect early issues with the peri-implant tissues without using a periodontal probe (Figure 2). Although probing around implants was once controversial, the 2017 guidelines from the American Academy of Periodontology and the European Federation of Periodontology (AAP/EFP) Consensus Conference suggest routine probing around implants is the current standard of care.8 However, the guidelines draw no conclusions on what constitutes health or disease in relation to probing depths. Nevertheless, the AAP/EFP data indicate that probing is the most accurate way to diagnose the presence of inflammation around these fixtures. A gentle probing pressure of 15 newton centimeter (Ncm) has been recommended around implants versus the 25 Ncm recommended for teeth; this lighter pressure around implants may reduce false positives for bleeding upon probing (BOP).
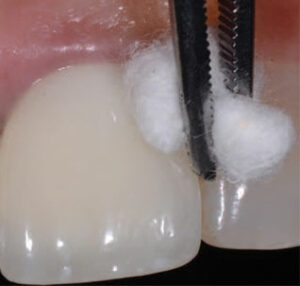
The authors advocate the use of a plastic periodontal probe because it is flexible and presents less risk of damaging the implant surface or implant components. If a plastic probe is not available, a metal probe may be used with care. Probing depths and signs of BOP should be recorded for future comparison. Clinically, BOP around implants with cemented restorations in patients with good personal oral hygiene usually indicates the presence of excess cement in the peri-implant tissues (Figure 3).
EVALUATION OF PERI-IMPLANT SOFT TISSUES
The dental implant process involves ongoing therapy and evaluation; for example, in addition to probing, clinicians should carefully examine the peri-implant soft tissues. Any deviation from normal color, thickness, height or presence of keratinization should be charted. A coral pink color of the gingiva is noted as a sign of health, with exceptions for racial melanin pigmentation. Regarding the dentition, an adequate band of keratinized and attached gingival tissue (ideally, 2 mm in the absence of exceptional plaque control) has been associated with increased soft tissue health. While debate continues about the need for keratinized tissue around implants, studies have found reduced signs of inflammation and bone loss around implants with at least 2 mm of keratinization9 and adequate tissue thickness of at least 2 mm.10
Following the examination, a diagnosis can be made. According to the AAP/EFP conference, peri-implant health is defined as a healthy soft tissue and bone interface with the implant. A patient considered to have peri-implant health will generally present with probing depths of 5 mm or less, and with no more than 2 mm of bone loss that remains stable, with no signs of inflammation. Peri-implant mucositis is characterized by BOP and tissue color changes shading toward red. The AAP/EFP conference papers assume this problem is related only to bacterial plaque. Peri-implantitis is defined as progressive loss of bone seen on radiographs and/or increased probing depths and is associated with biomass burden.8
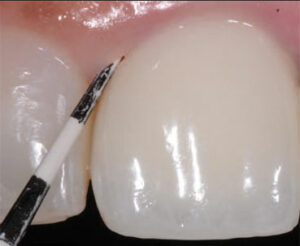
used to examine peri-implant tissues.
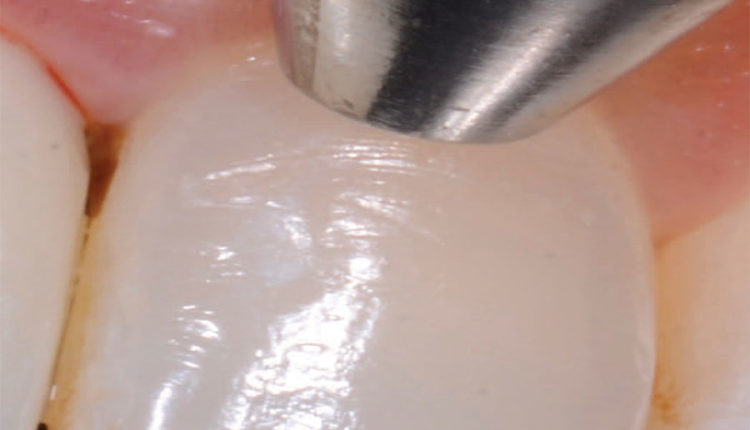
Diagnosis: Peri-implant Health — Maintenance: Assuming there are no signs of inflammation or continued bone loss in these cases, a conservative approach to reducing biofilm is suggested. Sterile saline on a cotton pellet is used to reduce the biologic load on the implant surface. A metal curet used to debride the sulcus should be turned away from the implant surface to remove any debris in the sulcus, taking care to not touch the implant or restorative surface. The implant crown may be instrumented with a conventional rubber cup; however, use of an ultrasonic, sonic or piezo device on the implant, abutment or implant crown should be avoided due to the possibility of damaging the implant surface.
Maintenance for Peri-implant Tissue Problems: The use of any device that will create aerosols should be preceded by a 30-second rinse with 0.12% chlorhexidine gluconate. This significantly reduces the cultivatable microflora within the oral cavity, thus reducing the possible spread of these bacteria to the dental team and patients.11 Next, clinicians should polish the natural teeth and implant prostheses with a rubber cup, after which calculus removal (both supra- and subgingival) is performed around the natural dentition.
Diagnosis: Peri-implant Mucositis, Option 1: Periodontal mucositis is frequently seen in clinical practice.12 One approach to dealing with this condition emphasizes removal of biofilm and material adherent to the implant surface in a manner that will minimally damage the implant surface. Adherents of this approach believe that small fragments of titanium can set up an infection in the soft tissues that can eventually lead to implant loss.13
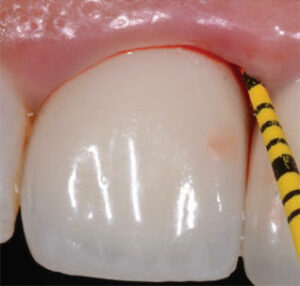
In this approach, a cotton pellet dipped in saline solution is suggested for removal of bacteria on the subgingival portions of the implant (Figure 4). The goal is to remove bacteria while leaving the surface as intact as possible, thus reducing the amount of titanium released into the soft tissues. Deposits found on implant surfaces are best removed with a sharp manual curet. This is suggested because a recent study demonstrated the use of other mechanical devices may result in removal of the implant’s titanium surface, with subsequent deposition into the surrounding tissues.14 Next, a sweep of the implant sulcus is made using a curet (normally metal), with the blade turned toward the sulcus wall (Figure 5). The purpose of this sweep is to remove particles of cement and titanium (as well as microbial deposits) that may have accumulated in the soft tissue and avoid damaging the implant surface.
In these cases, the patient’s oral hygiene is reinforced and twice-daily chlorhexidine rinses are suggested. The area is reevaluated in one month. If the patient’s personal oral hygiene is good and BOP is found at the second visit, it is highly probable that foreign bodies are present in the surrounding soft tissues. If the prosthesis is cemented, the foreign bodies will most likely be excess cement, or in some cases titanium. If this proves to be the case, surgical removal of the soft tissues immediately surrounding the implant is often indicated to facilitate removal of embedded foreign bodies. It is suggested these materials be removed after elevating a flap.
The flap is used to provide visual access to the implant surface and allow removal of excess cement and biofilm. Cement removal from the implant surface should be accomplished with a sharp metal curet, and every attempt should be made to reduce the damage to the implant surface so as to minimize the release of titanium particles. This approach can be greatly enhanced with the use of the dental videoscope. If surgery is needed, referral to a specialist should be considered. It should be noted that regaining health of the peri-implant tissues is delayed compared to tissues with gingivitis.15
Diagnosis: Peri-implant Mucositis, Option 2: Other clinicians believe the release of titanium particles into the surrounding tissues is of no consequence, and that thorough cleaning of the implant surface is needed to remove any adherent foreign material. Various instruments have been suggested for this purpose, including plastic, stainless steel, titanium and gold-tipped scalers. Ultrasonic tips have also been used with traditional, plastic-coated and metal-alloy tips. Likewise, the use of polishing paste, air-powder abrasives and glycine powder has been suggested.4 It is the authors’ contention that all of these methods have the potential to cause inflammation of the surrounding tissues. This hypothesis is currently being debated.
Diagnosis: Peri-Implantitis: When continued bone loss associated with clinical signs of inflammation is seen (i.e., peri-implantitis), steps should be taken to halt — and hopefully reverse — the process. Peri-implantitis is defined as progressive bone loss seen one year following placement of an implant prosthesis of 3 mm or greater, in combination with inflammation and probing depths of 6 mm or greater.8 The results of treatment have been mixed; consequently, referral to a specialist with experience treating this condition is strongly suggested.
OCCLUSAL CONSIDERATIONS
The effect of occlusion on bone loss around implants is controversial. However, it is well documented that excessive occlusal forces can result in fracture of implant crowns, screws and, in some cases, the implants themselves. As a result, it is suggested the effect of bite forces be regularly evaluated. Excessive bite forces can result from improper occlusal relationships, especially clenching and bruxing.
Clinical signs of these forces can be evidenced by fracture of the overlying restoration, loose or broken occlusal screws, and fractured or mobile implants. Some patients develop temporomandibular joint symptoms, while others exhibit fremitus or cold sensitivity on teeth in the arch opposing the implants. Due to the potential harm from excessive bite forces, the authors feel that in most cases fabrication of a maxillary hard-acrylic full coverage night guard is appropriate to help protect implants and implant restorations. This is especially true for patients with parafunctional habits.
RADIOGRAPHIC EVALUATION
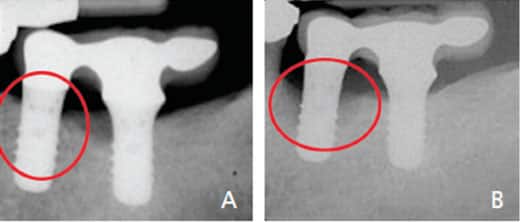
Radiographs are an important means of evaluating implant health. Right-angle radiographs, either periapicals or vertical bitewings, should be taken at appropriate intervals, starting just after the overlying prosthesis is placed. For patients with no clinical signs of inflammation or increased probing depths, this would be every five years. For patients showing signs of peri-implant disease, more frequent radiographic evaluation is appropriate (Figures 6A and 6B).
Systemic modifiers of periodontal and peri-implant diseases — including smoking, diabetes, thyroid and parathyroid disorders, and other systemic diseases or habits — can have negative effects on the tissues surrounding dental implants. For this reason, patients should be questioned about any changes in their general health that might have negative effects on their implants; in addition, appropriate behavioral changes should be addressed.
Following evaluation of the patient’s oral hygiene and self-care regimen, appropriate instruction and suggestions for modification can be offered. For example, a soft manual or electric toothbrush should be used for a minimum of two minutes at a 45-degree angle to the gingival margin. Interproximal cleaning aids, such as floss and/or interproximal brushes, should be used as appropriate, depending on the size of the interproximal space. In areas where the gingival margin of the implant is apical to adjacent teeth, sulcular cleaning aid, such as a Sulcabrush, end-tuft brush or implant care brush, may be helpful. The positive effect on health resulting from good oral hygiene cannot be overemphasized.
MAINTENANCE INTERVALS
As part of the dental implant process, establishing individualized maintenance intervals for partially dentate patients should be determined according to their periodontal status and the status of the peri-implant tissues.16,17 In general, this would mean an individual with a healthy periodontium and peri-implant tissues would be seen for maintenance at yearly intervals. One study concluded that compliance to suggested maintenance intervals was better for implant patients than those diagnosed with periodontitis.18 Patients with gingivitis (or peri-implant mucositis not associated with excess cement) are usually seen every six months, while those who have periodontitis with increased bone loss around their implants should be seen more frequently.19 Individuals with a history of periodontitis who maintain regular maintenance visits, as well as high levels of personal oral hygiene, have been shown to have minimal bone loss around their implants.20,21 Overall, the dental implant maintenance schedule should be determined by the individual’s specific disease profile, as ongoing maintenance is critical to the survival of these devices.
SUMMARY
Dental implants should be maintained on a regular schedule. Approaches to treating peri-implant disease are controversial at this time, and practitioners should consider treatment carefully, as incorrect treatment of early peri-implant disease may contribute to later implant complications.
REFERENCES
- Monje A, Aranda L, Diaz KT, et al. Impact of maintenance therapy for the prevention of peri-implant diseases: a systematic review and meta-analysis. J Dent Res. 2016;95:372–379.
- Salvi GE, Lang NP. Diagnostic parameters for monitoring peri-implant conditions. Int J Oral Maxillofac Implants. 2004;Suppl 19:116–127.
- Armitage GC, Xenoudi P. Post-treatment supportive care for the natural dentition and dental implants. Periodontol 2000. 2016;71:164–184.
- Todescan S, Lavigne S, Kelekis-Cholakis A. Guidance for the maintenance care of dental implants: clinical review. J Can Dent Assoc. 2012;78:c107.
- Costa FO, Takenaka-Martinez S, Cota LO, Ferreira SD, Silva GL, Costa JE. Peri-implant disease in subjects with and without preventive maintenance: a 5-year follow-up. J Clin Periodontol. 2012;39:173–181.
- Jepsen S, Berglundh T, Genco R, et al. Primary prevention of peri-implantitis: managing peri-implant mucositis. J Clin Periodontol. 2015;42(Suppl 16):S152–S157.
- Lang NP, Wilson TG Jr, Corbet EF. Biological complications with dental implants: their prevention, diagnosis and treatment. Clin Oral Implants Res. 2000;11(Suppl 1):146–155.
- Caton JG, Armitage G, Berglundh T, et al. A new classification scheme for periodontal and peri-implant diseases and condition — introduction and key changes from the 1999 classification. J Periodontol. 2018;45(Suppl 20):S1–S8.
- Boynueğri D, Nemli SK, Kasko YA. Significance of keratinized mucosa around dental implants: a prospective comparative study. Clin Oral Implants Res. 2013;24:928–933.
- Linkevicius T, Apse P, Grybauskas S, Puisys A. The influence of soft tissue thickness on crestal bone changes around implants: a 1-year prospective controlled clinical trial. Int J Oral Maxillofac Implants. 2009;24:712–719.
- Yadav S, Kumar S, Srivastava P, Gupta KK, Gupta J, Khan YS. Comparison of efficacy of three different mouthwashes in reducing aerosol contamination produced by ultrasonic scaler: a pilot study. In J Dent Sci. 2018;10:6–10.
- Pontoriero R, Tonelli MP, Carnevale G, Mombelli A, Nyman SR, Lang NP. Experimentally induced peri-implant mucositis. A clinical study in humans. Clin Oral Implants Res. 1994;5:254–259.
- Wilson TG Jr, Valderrama P, Burbano M, et al. Foreign bodies associated with peri-implantitis human biopsies. J Periodontol. 2015;86:9–15.
- Harrel SK, Wilson TG Jr, Pandya M, Diekwisch TG. Titanium particles generated during ultrasonic scaling of implants. J Periodontol. 2019;90:241–246.
- Salvi GE, Aglietta M, Eick S, Sculean A, Lang NP, Ramseier CA. Reversibility of experimental peri-implant mucositis compared with experimental gingivitis in humans. Clin Oral Implants Res. 2012;23:182–190.
- Tonetti MS, Chapple IL, Jepsen S, Sanz M. Primary and secondary prevention of periodontal and peri-implant diseases: introduction to, and objectives of the 11th European Workshop on Periodontology consensus conference. J Clin Periodontol. 2015;42(Suppl 16):S1–S4.
- Aguirre-Zorzano LA, Estefania-Fresco R, Telletxea O, Bravo M. Prevalence of peri-implant inflammatory disease in patients with a history of periodontal disease who receive supportive periodontal therapy. Clin Oral Implants Res. 2015;26:1338–1344.
- Cardaropoli D, Gaveglio L. Supportive periodontal therapy and dental implants: an analysis of patients’ compliance. Clin Oral Implants Res. 2012;23:1385–1388.
- Swierkot K, Lottholz P, Flores-de-Jacoby L, Mengel R. Mucositis, peri-implantitis, implant success, and survival of implants in patients with treated generalized aggressive periodontitis: 3- to 16-year results of a prospective long-term cohort study. J Periodontol. 2012;83:1213–1225.
- Aguirre-Zorzano LA, Vallejo-Aisa FJ, Estefania-Fresco R. Supportive periodontal therapy and periodontal biotype as prognostic factors in implants placed in patients with a history of periodontitis. Med Oral Patol Oral Cir Bucal. 2013;18:e786–e792.
- Pjetursson BE, Helbling C, Weber HP, et al. Peri-implantitis susceptibility as it relates to periodontal therapy and supportive care. Clin Oral Implants Res. 2012;23:888–894.
From Decisions in Dentistry. November/December 2019;5(10):32—35.




