Innovations in Endodontic Cleaning and Disinfection
The challenges inherent to this portion of endodontic treatment have led to technologies designed to help ensure clinical success.
This course was published in the April 2018 issue and expires April 2021. The author has no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
OBJECTIVES
- List some of the inherent challenges clinicians face in endodontic cleaning and disinfection.
- Describe various approaches to endodontic cleaning and disinfection that have been designed to address the problem of pulpal debridement and microbial disinfection.
- Discuss the use of calcium hydroxide as an interim measure in endodontic therapy, and the resulting difficulty in removing the material using conventional techniques.
Introduction
Innovation often presents in two forms: 1) Incremental. 2) Disruptive. Most innovations in root canal therapy are incremental and appeal to a broad audience of dentists classified as “late majority” adopters of technology. Some of the technologies discussed in this article are incremental innovations (e.g., passive ultrasonic activation, sonic activation and negative apical pressure irrigation). One of the technologies discussed in this article — the GentleWave System — classifies as a disruptive technology and will appeal to a smaller segment of “innovative and progressive” dentists.
The GentleWave Procedure represents an inflection point in root canal therapy. This inflection point, or shift in market fundamentals, manifests in referring dentists who demand improvements in efficacy for the patients they refer to endodontists. In addition, patients are now taking a more active role in their health care decisions. Inflection points represent an exciting time for the future of root canal therapy and dentistry, in general.
The success of endodontic therapy hinges on thorough canal cleaning and disinfection techniques
Innovation drives dental science forward and helps clinicians achieve better patient outcomes. Due to the rapid pace of innovation in dentistry, clinicians can feel lost if they don’t stay abreast of new systems designed to improve treatment efficiency and efficacy. With the introduction of the dental operating microscope, surgical ultrasonics and other surgical instruments, the field of endodontics has seen significant advances in the last 20 years. In particular, cone beam computed tomography (CBCT) has fundamentally changed the practice of endodontics by allowing clinicians to view true alveolar breakdown related to an infection of endodontic origin in three dimensions. The use of CBCT imaging allows clinicians to visualize the anatomy, canal location and shape, and even helps establish a reasonably accurate working length — all prior to access.1 With proper visualization of the anatomy, providers can select nickel titanium shaping files that are extremely resistant to cyclic fatigue and, thus, file separation.2 Now, canals can be safely and efficiently shaped within minutes. However, the challenge of canal cleaning and disinfection persists.
If clinicians can locate (using CBCT) and shape canals within minutes, are techniques available to adequately clean the entire root canal system? Apart from the main canal, lateral canals, secondary and accessory canals, isthmuses and other deep apical anatomy — such as apical deltas, and anastomoses, fins and crevices — are unseen by the human eye and especially difficult to treat. This is a significant and clinically relevant challenge. In addition, it has been suggested that techniques commonly used to irrigate and chemically debride vital and necrotic pulp tissue, microorganisms and biofilm from the entire root canal system are inadequate.3
This article will review some of the advances in root canal cleaning and disinfection technologies. Most of today’s irrigation systems, whether they activate irrigants (e.g., passive ultrasonic or sonic irrigation), apply photons into the sodium hypochlorite to incite microbubbles (e.g., photon-induced photoacoustic streaming [PIPS]), or introduce the irrigants to the apex by employing negative apical pressure, yield cleaner root canal systems with very little apical extrusion.4 Recently, a new system was developed that uses a multimodality approach (i.e., degassed fluids, useful cavitation, multisonic acoustic waves, and negative apical pressure) to remove pulpal tissue and pathogenic microbes. Each of these advances has been designed to help solve the problem of pulpal debridement and microbial disinfection, which is central to the success of endodontic therapy.

CHEMOMECHANICAL PREPARATION
Although the presence of microorganisms was observed in necrotic dental pulp more than 100 years ago,5 Kakehashi et al6 was the first to demonstrate a causal relationship between the presence of bacteria and apical periodontitis. Pathobiological studies soon followed, and microorganisms as the etiology of endodontic disease now forms the rational basis for prevention and treatment of endodontic pathology. To this end, irrigation techniques serve to eliminate a milieu favoring microbial persistence.
Grossman and Meiman7 laid the groundwork for contemporary irrigation when they demonstrated the solvent properties of sodium hypochlorite on pulp tissue in 1941. Today, sodium hypochlorite and ethylenediaminetetraacetic acid (EDTA) are the mainstay irrigant solutions applied via syringe. Empirical evidence, however, continues to reveal that mechanical preparation and tactile administration of irrigants into the canal space insufficiently removes microorganisms, biofilm and other debris.
The root canal system is anatomically complex; thus it should not be surprising that 20% to 80% of the canal surface area remains unprepared using various instrumentation techniques.8,9 One study showed that shaping the main canals of mesial roots of lower molars with various files resulted in 20% to 35% of unprepared canals (not including isthmi).9 Similarly, another study showed that, on average, only 65% to 75% of the canal walls are touched with files.10 When evaluating the apical 4 mm “red zone,” Peters et al11 reported that ~19% and 44% of the canal surface was unprepared after preparation up to a size 20/.06 and 30/.06, respectively. Unprepared canal walls are major harbors of remnant pulp and microbes, which may threaten endodontic success.12
Conventional endodontic therapy using both rotary instrumentation and standard needle irrigation (SNI) with sodium hypochlorite also shows incomplete removal: According to Molina et al,3 only 68% of the root canal surface was debrided with this approach. In addition, SNI is ineffective at removing hard and soft tissue remnants and debriding the apical third region.13,14 Approximately 30% (or more) of pulpal tissue and debris is left behind in the canal system, which raises the question: Can innovations in cleaning and disinfection technology bridge this gap?
PASSIVE ULTRASONIC ACTIVATION
Passive ultrasonic irrigation (PUI) has been available for decades and was one of the first proposed means by which a clinician can improve chemical debridement.15 This modality activates irrigants inside the canal system by utilizing a single frequency ultrasonic energy, which causes acoustic streaming.16 This occurs when a clinician places a noncutting ultrasonic tip into a shaped canal containing sodium hypochlorite, EDTA, chlorhexidine or other irrigant, and activates the tip for a short period (e.g., 30 seconds to 2 minutes per canal). When compared with hand instrumentation alone, studies show significant reduction in debris removal, even when applied for as little as 30 seconds per canal.17,18 Although PUI is effective at achieving cleaner canals, there has been concern that it is not always passive and is not as effective in curved canals.19
SONIC ACTIVATION
In order to avoid the possible detrimental effects of PUI (such as dampening of the metal file and change in canal morphology through wall contact),19 sonic activation has been proposed as an alternative modality. One type of passive sonic irrigation (PSI) employs a polyamide tip to deliver approximately 6000 Hz of sonic energy. These devices, including the EndoActivator (Dentsply Sirona, Tulsa, Okla.), have been shown to remove significantly more smear layer than passive ultrasonic activation.20 Another study showed that PSI reduced microorganism colony forming units significantly better than PUI.21 The EndoActivator was also superior to SNI, PUI and a negative apical pressure device in removing calcium hydroxide. However, none of these methods removed all the calcium hydroxide from the canal walls.22 Furthermore, because sonic activation works at lower frequencies, evidence generally suggests it is less effective at removing debris than ultrasonic activation.23–26
NEGATIVE APICAL PRESSURE
The EndoVac (Kerr Dental, Orange, Calif.) is another innovative irrigation technique that has shown the ability to provide cleaner canals, particularly in the apical one-third.27 Studies show that SNI only delivers irrigant 0 to 1.1 mm beyond the needle tip;28 SNI is also associated with vapor lock, or trapped air, in the apical one-third, which can hinder the effectiveness of cleaning debris from the apex and in complex, deep anatomy.29 To address these problems, the EndoVac was developed to deliver irrigants to the apex and prevent or bypass vapor lock. This device employs negative apical pressure using a macrocannula/microcannula that, when placed at or near the apex, can deliver irrigant into the apical one-third, thus avoiding apical vapor lock. Not only does this device deliver irrigant directly to the apex, it also evacuates the solution along with apical debris.30 This concept was demonstrated by Mancini et al,20 who reported the EndoVac was more effective than PUI and a PSI device in removing smear layer in the apical one-third. It should be noted, however, that EndoVac generally requires instrumentation to size #35 or greater.31–33 In addition, the EndoVac is not effective in complex anatomy.34
PHOTON-INDUCED PHOTOACOUSTIC STREAMING
In endodontics, PIPS technology (Fotona, Dallas, Texas) is used to improve chemical debridement of the root canal system. This approach is based on an erbium yttrium aluminum garnet (Er:YAG) laser that emits a wavelength of 2940 nanometers that is strongly absorbed by water. The Er:YAG laser provides an aggressive photoacoustic streaming of water, sodium hypochlorite, EDTA or chlorhexidine inside the root canal system by using laser-emitted photons from a quartz and radially firing tip that induces tens of thousands of microbubbles to form and quickly collapse. The collapse of thousands of microbubbles at any given time causes a sonic boom effect that loosens and removes pulpal tissue, removes the smear layer and biofilm, and lyses bacterial cell walls.35,36 This irrigation method also pushes antibacterial solution into isthmi, lateral and secondary canals, apical deltas and anastomosis, and can enhance disinfection of deep, complex apical anatomy or the apical red zone.37,38

Peters et al39 showed that using the PIPS proprietary technique (specifically, when used for 60 seconds for each tooth) was superior in removing bacteria when compared to SNI and PUI. Another study demonstrated 100% inhibition of Enterococcus faecalis growth after using the PIPS irrigation technique for 20 seconds with 6% sodium hypochlorite in 24 single-rooted teeth.40 However, other data indicate the efficacy of PIPS is comparable to standard techniques, such as conventional or ultrasonic irrigation and the EndoVac.41,42 In vitro studies also suggest PIPS extrudes more irrigant than SNI, EndoActivator and EndoVac.43
A NEW APPROACH
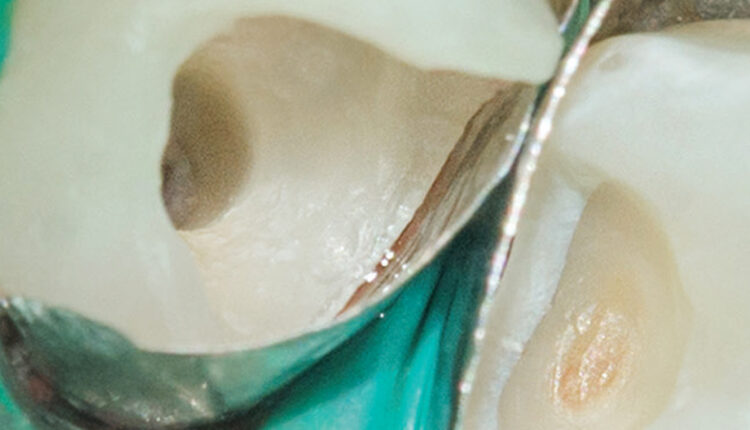
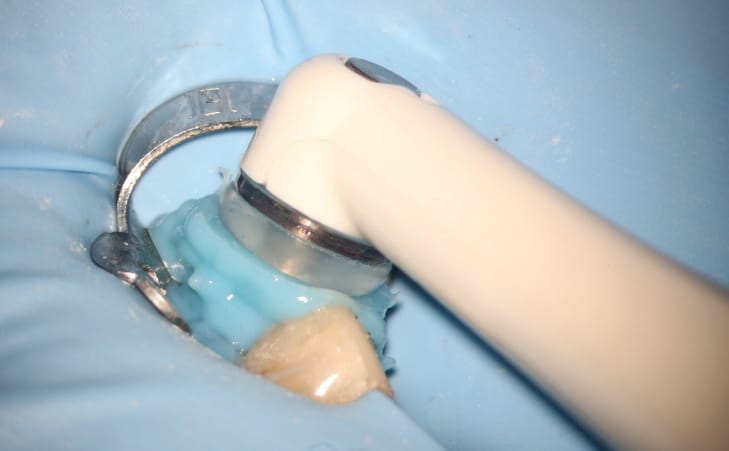
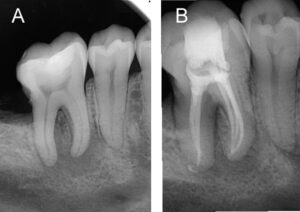
A novel technique, the GentleWave Procedure (Sonendo, Laguna Hills, Calif.) has been available since 2016. In this procedure, the GentleWave System (Figure 1) is used to dispense degassed fluids (e.g., distilled water, 3% sodium hypochlorite and 8% EDTA) by positioning a Procedure Instrument on top of the tooth to create a sealed coronal space, and placing the tip inside the pulp chamber (Figure 2). The stream of incoming fluid creates a shear force, resulting in the generation of a cavitation cloud and multisonic sound waves that remove organic and inorganic tissue, planktonic and biofilm microbes, and other debris within the canal system. This ~6- to 8-minute procedure not only removes pulpal tissue and microbes from the main canal, accessory canals and anastomoses, it is also effective at addressing clinical scenarios, such as cleaning isthmus tissue located in mesial roots of lower molars3 (Figure 3), removing separated files bound in the canal44 (Figure 4), and removing calcium hydroxide from the canal system.45 These data appear to translate into clinically relevant outcomes, as evidenced by clinical studies demonstrating > 97% healing rates after 12 months.46,47
The debridement properties of this approach were first reported by Molina et al3 in 2015. Histological analysis in freshly extracted molars showed 97.2% of pulp tissue was removed in maxillary molar mesiobuccal roots and mandibular molar mesial roots using the GentleWave Procedure with minimal instrumentation, compared to 67.8% using SNI and conventional shaping. Debris removal was also significantly improved using the GentleWave Procedure in the apical region of each tooth, which is a challenging region to clean.3 These findings underscore the procedure’s effectiveness in removing tissue and debris, and maximizing dentin preservation through minimal shaping (e.g., up to 20/.06). It also points to the limited efficacy of conventional endodontic irrigation in debris removal, even in canals with more aggressive shaping (e.g., 35/.04 or larger).
In vitro experiments also suggest that GentleWave technology is able to remove separated files lodged in the canal. Thirty-six extracted molars were subjected to the procedure after #10, #15 and #20 K files were purposefully separated in either the mid-root or apical region. Using this technology, researchers were able to remove 83% of the files in the mid-root and 61% in the apical region.44 In canals with curvature of less than 30 degrees, 91% of the files were removed. These data are consistent with the system’s cleaning effectiveness, as noted by Molina et al,3 particularly because there was no canal enlargement up to the separated file or ultrasonic engagement or manipulation of the separated file prior to the test procedure (Figure 4). It has also been hypothesized that if the GentleWave Procedure does not remove a separated file, it can clean around it, giving hope to cases in which a separated file lies in a canal with an apical lesion.48
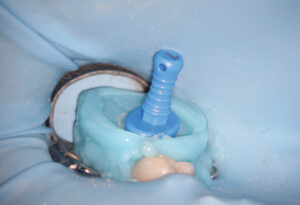
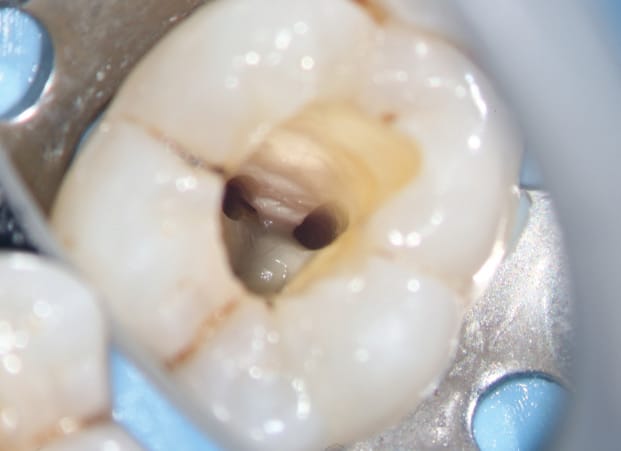
depth gauge is used to assess the depth of the pulpal floor.
In a common approach to root canal therapy, a clinician will place calcium hydroxide as an intracanal medicament and complete the root canal treatment at a future appointment. Research shows that it can be difficult to remove all calcium hydroxide at the next visit, however — as demonstrated by Lambrianidis et al,49 who reported that ~25% to 45% of calcium hydroxide remained attached to the canal wall after irrigation and mechanical filing with conventional techniques. In contrast, the GentleWave Procedure removed 99.85% and 99.97% in the mesial and distal canals, respectively.45
Does improved in vitro chemical debridement translate into greater clinical success? Prospective clinical studies by Sigurdsson et al47,50 reported 97.4% and 97.3% endodontic success after six and 12 months, respectively, in 75 patients treated with the GentleWave Procedure. By comparison, healing rates using standard techniques generally range from 65% to 90%.51–53 Furthermore, success rates were > 97% in patients with a nonvital pulp and large periodical lesions (as defined by an initial periodical index score ≥ 3) after 12 months, indicating efficacy in more severe cases.46 Although more research is needed, these prospective studies appear promising.
CLINICAL PROTOCOL
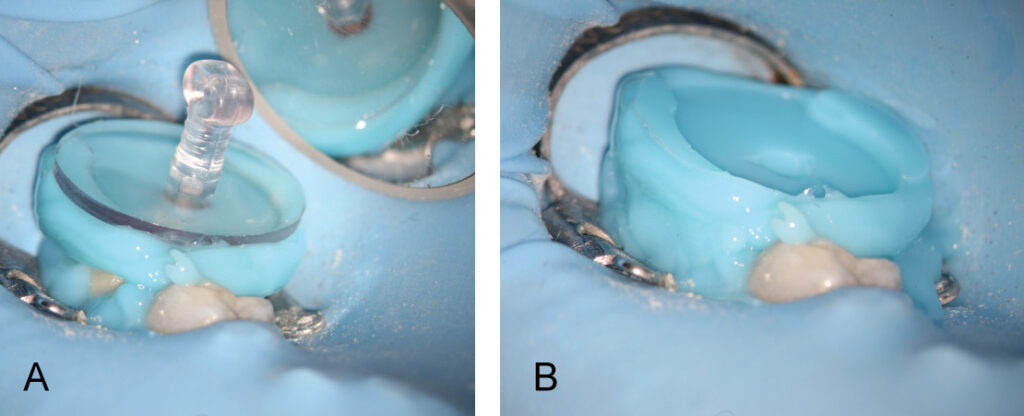
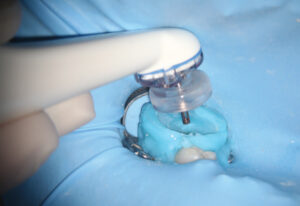
A fluid-tight seal over the endodontic access must be established prior to the GentleWave Procedure. This is accomplished by building a flat occlusal platform onto the tooth access with an occlusal matrix and a light-curable, conforming resin material (Figures 5A and 5B), which takes approximately 2 to 5 minutes. Once the occlusal platform is built, depth gauges are used to assess the depth of the pulpal floor (Figure 6). Either a molar or anterior/premolar Procedure Instrument is placed onto the occlusal platform with gentle pressure to ensure a fluid-tight seal. The procedure is initiated using the GentleWave console, which continually refreshes optimized procedure fluids to the tooth while the clinician monitors for a continuous seal (Figure 7). After ~6 to 8 minutes, the procedure is complete, and the Procedure Instrument and occlusal platform are removed. The canals are dried and cone fit radiographs are taken. After drying the canals, the clinician can obturate and complete the root canal treatment.
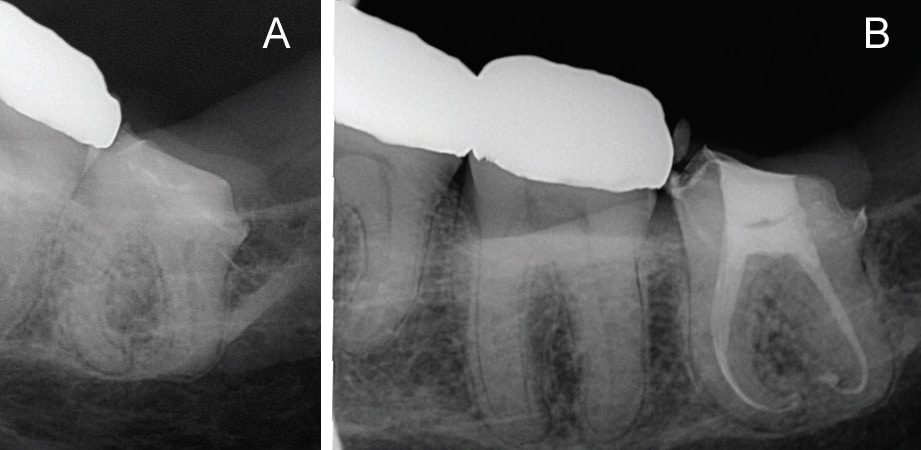
In addition to removing isthmus tissue, reports indicate that GentleWave technology can also help locate uninstrumented middle mesial canals that were previously unseen through the microscope (Figures 8A and 8B). Using this approach, clinicians can remove tissue in complex anatomies, such as C-shaped canals with minimal shaping (e.g., 20/.06 or smaller) during root canal debridement (Figures 9A and 9B).
CONCLUSION
Complex root anatomy makes endodontic treatment challenging, but innovative technologies have simplified canal cleaning and disinfection, improved efficiency, and increased clinical success. The modalities described here are designed to help clinicians effectively irrigate and chemically debride vital and necrotic pulp tissue, microorganisms and biofilm from the entire root canal system. In addition, empirical evidence suggests the GentleWave Procedure allows effective cleaning and disinfection with minimal shaping,3 thus preserving maximum tooth structure. In this respect, this is a promising new approach that warrants further clinical investigation.
REFERENCES
- Janner SF, Jeger FB, Lussi A, Bornstein MM.Precision of endodontic working length measurements: a pilot investigation comparing cone-beam computed tomography scanning with standard measurement techniques. J Endod. 2011;37:1046–1051.
- Topçuoğlu HS, Düzgün S, Aktı A, Topçuoğlu G.Laboratory comparison of cyclic fatigue resistance of WaveOne Gold, Reciproc and WaveOne files in canals with a double curvature. Int Endod J. 2017;50: 713–717.
- Molina B, Glickman G, Vandrangi P, Khakpour M.Evaluation of root canal debridement of human molars using the GentleWave System. J Endod. 2015;10:1701–1705.
- Mitchell RP, Baumgartner C, Sedgley C. Apical extrusion of sodium hypochlorite using different root canal irrigation systems. J Endod. 2011;37: 1677–1681.
- Miller WD. The Micro-Organisms of the Human Mouth. Philadelphia, Penn: The S.S. White Dental Mgf Co;1890.
- Kakehashi S, Stanley HR, Fitzgerald RJ. The effects of surgical exposures of dentral pulps in germ-free and conventional laboratory rats. Oral Surg Oral Med Oral Pathol. 1965; 20:340–349.
- Grossman LI, Meiman BW. Solution of pulp tissue by chemical agents. J Am Dent Assoc. 1941;28: 223–225.
- Paqué F, Balmer M, Attin T, Peters OA. Preparation of oval-shaped root canals in mandibular molars using nickel-titanium rotary instruments: a micro-computed tomography study. J Endod. 2010;36:703–707.
- Siqueira JF Jr, Alves FR, Versiani MA, et al. Correlative bacteriologic and micro-computed tomographic analysis of mandibular molar mesial canals prepared by self-adjusting file, reciproc, and twisted file systems. J Endod. 2013;39:1044–1050.
- El Ayouti A, Chu AL, Kimonis I, Klein C, Weiger R, Löst C. Efficacy of rotary instruments with greater taper in preparing oval root canals. Int Endod J. 2008; 41:1088–1092
- Peters OA, Arias A, Paqué F. A micro-computed tomographic assessment of root canal preparation with a novel instrument, TRUShape, in mesial roots of mandibular molars. J Endod. 2015;41:1545–1550.
- Siqueira JF Jr, Perez AR, Marceliano-Alves MF, et al. What happens to unprepared root canal walls: a correlative analysis using micro-computed tomography and histology/scanning electron microscopy. Int Endod J. 2017:1–8.
- Boutsioukis C, Lambrianidis T, Verhaagen B, et al. The effect of needle-insertion depth on the irrigant flow in the root canal: evaluation using an unsteady computational fluid dynamics model. J Endod. 2010; 36:1664–1668.
- Susin L, Liu Y, Yoon JC, et al. Canal and isthmus debridement efficacies of two irrigant agitation techniques in a closed system. Int Endod J. 2010;43: 1077–1090.
- Richman MJ. The use of ultrasonics in root canal therapy and root resection. Med Dent J. 1957; 12:12–18.
- Ahmad M, Pitt Ford TR, Crum LA. Ultrasonic debridement of root canals: an insight into the mechanisms involved. J Endod. 1987;13:93–101.
- Jensen SA, Walker TL, Hutter JW, Nicoll BK. Comparison of the cleaning efficacy of passive sonic activation and passive ultrasonic activation after hand instrumentation in molar root canals. J Endod. 1999;25:735–738.
- Sabins RA, Johnson JD, Hellstein JW. A comparison of the cleaning efficacy of short-term sonic and ultrasonic passive irrigation after hand instrumentation in molar root canals. J Endod. 2003;29:674–678.
- Boutsioukis C, Verhaagen B, Walmsley AD, Versluis M, van der Sluis LW. Measurement and visualization of file-to-wall contact during ultrasonically activated irrigation in simulated canals. Int Endod J. 2013; 46:1046–1055.
- Mancini M, Cerroni L, Iorio L, Armellin E, Conte G, Cianconi L. Smear layer removal and canal cleanliness using different irrigation systems (EndoActivator, EndoVac, and passive ultrasonic irrigation): field emission scanning electron microscopic evaluation in an in vitro study. J Endod. 2013;39:1456–1460.
- Neuhaus KW, Liebi M, Stauffacher S, Eick S, Lussi A. Antibacterial efficacy of a new sonic irrigation device for root canal disinfection. J Endod. 2016; 42:1799–1803.
- Alturaiki S, Lamphon H, Edrees H, Ahlquist M. Efficacy of three different irrigation systems on removal of calcium hydroxide from the root canal: a scanning electron microscopic study. J Endod. 2015;41:97–101.
- Paragliola R, Franco V, Fabiani C. et al. Final rinse optimization: influence of different agitation protocols. J Endod. 2010;36:282–285.
- Uroz-Torres D, González-Rodríguez MP, Ferrer-Luque CM. Effectiveness of the Endo Activator System in removing the smear layer after root canal instrumentation. J Endod. 2010;36:308–311.
- Sabins RA, Johnson JD, Hellstein JW. A comparison of the cleaning efficacy of short-term sonic and ultrasonic passive irrigation after hand instrumentation in molar root canals. J Endod. 2003;29:674–678.
- Johnson M, Sidow SJ, Looney SW, Lindsey K, Niu LN, Tay FR. Canal and isthmus debridement efficacy using a sonic irrigation technique in a closed-canal system. J Endod. 2012;38:1265–1268.
- Nielsen BA, Baumgartner JC. Comparison of the EndoVac System to needle irrigation of root canals. J Endod. 2007;33:611–615.
- Munoz HR, Camacho-Cuadra K. In vivo efficacy of three different endodontic irrigation systems for irrigant delivery to working length of mesial canals of mandibular molars. J Endod. 2012;38:445–448.
- Tay FR, Gu LS, Schoeffel G, et al. Effect of vapor lock on root canal debridement by using a side-vented needle for positive-pressure irrigant delivery. J Endod. 2010;36:745–750.
- Pawar R, Alqaied A, Safavi K, Boyko J, Kaufman B. Influence of an apical negative pressure irrigation system on bacterial elimination during endodontic therapy: a prospective randomized clinical study. J Endod. 2012;38:1177–1181.
- Dua A, Dua D. Comparative evaluation of efficacy of EndoVac irrigation system to Max-I probe in removing smear layer in apical 1 mm and 3 mm of root canal: An in vitro scanning electron microscope study. Dent Res J. 2015;12:38–43.
- Karade P, Chopade R, Patil S, et al. Efficiency of different endodontic irrigation and activation systems in removal of the smear layer: a scanning electron microscopy study. Iran Endod J. 2017;12:414–418.
- Suman S, Verma P, Prakash-Tikku A, Bains R, Kumar-Shakya V. A comparative evaluation of smear layer removal using apical negative pressure (EndoVac), sonic irrigation (EndoActivator) and Er:YAG laser — An In vitro SEM Study. J Clin Exp Dent. 2017;9:e981–e987.
- Versiani MA, Alves FR, Andrade-Junior CV, et al. Micro-CT evaluation of the efficacy of hard-tissue removal from the root canal and isthmus area by positive and negative pressure irrigation systems. Int Endod J. 2016;49:1079–1087.
- Zhu X, Yin X, Chang J, Wang Y, Cheung G, Zhang C. Comparison of the antibacterial effect and smear layer removal using photon-initiated photoacoustic streaming aided irrigation versus a conventional irrigation in single-rooted canals: an in vitro study. Photomed Laser Surg. 2013;31:371–377.
- Arslan H, Capar I, Saygili G, Gok T, Akcay M. Effect of photon-initiated photoacoustic streaming on removal of apically placed dentinal debris. Int Endod J. 2014;47:1072–1077.
- Lin S, Liu Q, Peng Q, Lin M, Zhan Z, Zhang Z. Ablation threshold of Er:YAG and Er,Cr:YSGG laser in dental dentin. Sci Res Essays. 2010;5:2128–2135.
- Majaron B, Lukac M, Sustercic D, Funduk N, Skaleric U. Threshold and efficiency analysis in Er:YAG laser ablation of hard dental tissue. Proc SPIE. 1996; 2922: 233–242.
- Peters OA, Bardsley S, Fong J, Pandher G, Divito E. Disinfection of root canals with photon-initiated photoacoustic streaming. J Endod. 2011;37: 1008–1012.
- Jaramillo DE, Aprecio R, Angelov N, Divito E, McClammy TV. Efficacy of photon induced photoacoustic streaming (PIPS) on root canals infected with Enterococcus faecalis: a pilot study. Endod Pract. 2012;7:28–32.
- Verstraeten J, Jacquet W, De Moor RJG, Meire MA. Hard tissue debris removal from the mesial root canal system of mandibular molars with ultrasonically and laser-activated irrigation: a micro-computed tomography study. Lasers Med Sci. 2017;32:1965–1970.
- Turkel E, Onay EO, Ungor M. Comparison of three final irrigation activation techniques: effects on canal cleanness, smear layer removal, and dentinal tubule penetration of two root canal sealers. Photomed Laser Surg. 2017;35:672–681.
- Azim AA, Aksel H, Margaret Jefferson M, Huang GT. Comparison of sodium hypochlorite extrusion by five irrigation systems using an artificial root socket model and a quantitative chemical method. Clin Oral Investig. 2018;22:1055–1061.
- Wohlgemuth P, Cuocolo D, Vandrangi P, Sigurdsson A. Effectiveness of the GentleWave System in removing separated instruments. J Endod. 2015; 41:1895–1898.
- Ma J, Shen Y, Yang Y, et al. In vitro study of calcium hydroxide removal from mandibular molar root canals. J Endod. 2015;41:553–558.
- Sigurdsson A, Garland RW, Le KT, Rassoulian SA. Healing of periapical lesions after endodontic treatment with the GentleWave procedure: a prospective multicenter clinical study. J Endod. 2018;44:510–517.
- Sigurdsson A, Garland RW, Le KT, Woo SM. 12-month healing rates after endodontic therapy using the novel GentleWave System: a prospective multicenter clinical study. J Endod. 2016;42:1040–1048.
- Haapasalo M. Lecture given in New Orleans, April 2017.
- Lambrianidis T, Margelos J, Beltes P. Removal efficiency of calcium hydroxide dressing from the root canal. J Endod. 1999;25:85–88.
- Sigurdsson A, Le KT, Woo SM, et al. Six-month healing success rates after endodontic treatment using the novel GentleWave System: the pure prospective multi-center clinical study. J Clin Exp Dent. 2016;8:290–298.
- Dammaschke T, Steven D, Kaup M, Ott KH. Longterm survival of root-canal–treated teeth: a retrospective study over 10 years. J Endod. 2003;29:638–643.
- Friedman S, Abitbol S, Lawrence HP. Treatment outcome in endodontics: the Toronto Study. Phase 1: initial treatment. J Endod. 2003;29:787–793.
- Farzaneh M, Abitbol, Freideman S. Treatment outcome in endodontics: The Toronto Study. Phases I and II: orthograde retreatment. J Endod. 2004;30:627–633.
Featured image by CREDIT/ISTOCK/GETTY IMAGES PLUS
From Decisions in Dentistry. April 2018;4(4):37-42.




