
Examining Bioactive Restorative Materials
What the evidence says about their clinical application in restorative dentistry.
The term “bioactive” has become a trend in dental materials marketing, but many practicing dentists and researchers are struggling to understand what it truly means.1 By its most basic definition, bioactive refers to a material that has a biological effect on surrounding tissues. The desired effect will depend on the purpose of the material. For example, a bioactive liner may cause the release of growth factors from the dentinal collagen to induce the formation of reparative dentin in the pulp chamber. A desirable bioactive restorative material would create a bond with surrounding tooth structure, and release ions to allow remineralization at tooth margins following an acid challenge. A bioactive cement may have the additional benefit of attracting calcium phosphate precipitates to its surface to occlude an existing cement gap.
This article will describe several classes of restorative materials that have been termed bioactive and explore the evidence for their claims of bioactivity; in addition, while it provides product examples for clinical context, this is not meant to be an all-inclusive list. There are additional clinical applications for bioactive materials outside the scope of this article, including endodontic sealers, root end fillers, and root perforation repair materials. There are also emerging concepts for bioactive dental materials that will not be covered, such as the use of small molecules to inhibit biofilm (plaque) accumulation on restorative materials, or alkasite materials that will elevate the local pH to prevent demineralization of surrounding tooth structure.
Early studies of the compatibility between calcium hydroxide and vital pulp structure date back to the 1930s. Since then, calcium hydroxide liners have become the gold standard in pulp capping materials due to their antimicrobial activity (by increasing pH) and ability to stimulate reparative dentin (by initiating release of growth factors entombed in dentinal collagen).2 Calcium hydroxide liners, however, have limitations, including an inability to bond to tooth structure and high solubility. Clinically, inadequately sealed calcium hydroxide liners are susceptible to dissolution, exposing the underlying pulp tissue to bacterial penetration.3 For this reason, calcium hydroxide liners should be covered with a layer of resin-modified glass ionomer (RMGI). While calcium hydroxide liners incorporated in light-cured resins were introduced to reduce solubility and allow a bond to the tooth, this modification limited the material’s ion release.2
DEVELOPMENTS IN MATERIALS
In 1993, mineral trioxide aggregate (MTA) was introduced, which is a mix of Portland cement (tricalcium silicate, dicalcium silicate and tricalcium aluminate) and a radiopaquer (bismuth oxide). When calcium silicate in MTA is mixed with water, it forms calcium hydroxide. Therefore, MTA has the same mechanism of action as calcium hydroxide,4 but MTA does offer an advantage in that it forms a seal with tooth structure.5 Additionally, it will deposit a layer of calcium phosphate on its surface in the presence of a phosphate-containing solution (such as pulpal fluid). The deposited calcium phosphate may aid in the formation of reparative dentinal bridging when MTA is used for direct pulp capping.4 The disadvantages are that it requires a 2 hour and 45 minute final setting time and it is difficult to manipulate.6 Examples of MTA products include ProRoot MTA (Dentsply Sirona), MTA Angelus (Angelus), NuSmile NeoMTA (NuSmile) and NeoMTA Plus (Avalon Biomed).
Based on the components of MTA, newer materials have been fabricated with improved handling and setting properties. In 2008, a light-cured liner (Theracal LC, Bisco) was introduced that contains calcium silicate in a resin modified with hydrophilic components. This resin absorbs water, which allows hydration of the calcium silicate and release of calcium ions.7 The formulation was later modified to produce a cement (Theracem, Bisco). In 2009, a dental liner/temporary restorative (Biodentine, Septodont) was introduced with a tricalcium silicate powder mixed with calcium chloride liquid instead of water. This liquid allows the material to set in only 12 minutes.8,9
In addition to calcium silicate formulations, calcium aluminate materials have been developed for dental applications, and, similar to MTA, they are able to form a layer of precipitated calcium phosphate.10 Initially, a product was introduced as a restorative material with close adaptation to tooth structure that was interpreted as a chemical bond.10 In 2011, it was marketed as a dental cement (Ceramir C&B, Doxa) with the ability to attract calcium phosphate precipitates capable of occluding an existing cement gap.11
In 2014, a permanent restorative material (Activa BioActive Restorative, Pulpdent) was released that was described as an “RMGI preparation with a bioactive resin matrix and bioactive glass fillers.” In this case, bioactivity referred to the ability to release beneficial ions, such as calcium, fluoride and phosphate. The exact classification of this material is unclear, as it contains methacrylate resins with polyacrylic acid copolymers, suggesting it may be a compomer. This formulation was later modified to produce a liner and cement (Activa BioActive Base/Liner, Activa BioActive cement, Pulpdent) and a cement for pediatric crowns (BioCem, NuSmile). Another restorative material containing ion-releasing calcium fluorosilicate glass in a methacrylate resin was released in 2017 (Cention N, Ivoclar Vivadent).
In summary, materials for restorative procedures that are being marketed as bioactive include calcium silicate products based on Portland cement (MTA, tricalcium silicate mixed with calcium chloride, and calcium silicate in a resin matrix), calcium aluminate, and resin-based materials with ion-releasing fillers. The following sections will address the potential clinical advantages for bioactive materials as liners, restorative materials and cements.
BIOACTIVE LINERS
In a 2009 review, Hilton12 described evidence-based clinical recommendations for pulp capping. At that time, he concluded that if pulpal exposure occurred, calcium hydroxide and MTA liners performed equivalently, whereas glass ionomer, adhesive and zinc-oxide eugenol were shown to be poor pulp capping agents. In 2013, a large, practice-based study reported the probability of failure of a direct pulp cap was lower with MTA (19.7%) than calcium hydroxide (31.5%) at 24 months.13 A multicenter randomized controlled clinical trial also reported the probability of failure of a direct pulp cap with MTA (18%) was lower than calcium hydroxide (48%) at 36 months.14 These results were unexpected, as the bioactive component of MTA is, in fact, calcium hydroxide. The authors credited the improved clinical performance of MTA over calcium hydroxide to its ability to seal the dentin.13 In histological studies, MTA has shown more predictable dentinal bridging and less pulpal inflammation.15
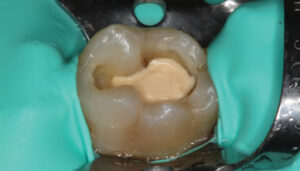
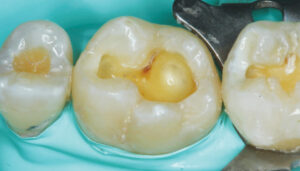
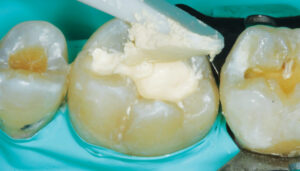
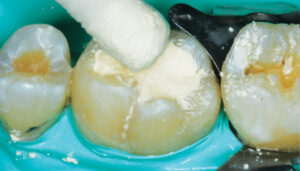
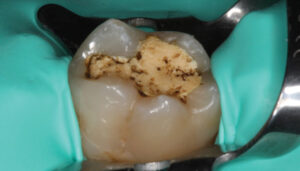
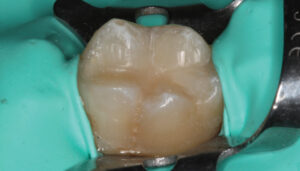
The limitation of a traditional MTA product remains its lengthy final setting time — although some materials have a faster initial (i.e., washout resistance) time. This slow setting time requires the clinician to either place an RMGI liner over the MTA while it sets, or place a temporary restoration over the MTA (that is later replaced once the MTA is set). In order to gain the benefits of using a calcium silicate-based liner (namely, MTA) without the slow setting time, the practitioner may choose to use a calcium silicate/calcium chloride temporary restorative material (Biodentine) with a 12-minute setting time (Figure 1 through Figure 6, page 25), or opt to place a light-cured calcium silicate liner (Theracal LC) immediately covered with a permanent restoration (Figure 7 through Figure 9). Both materials have demonstrated calcium release and the ability to form dentinal bridging.3,16,17 The calcium silicate/calcium chloride temporary material has been shown to maintain clinical function for up to six months before needing to be covered with a permanent restoration.18 Regarding the light-cured calcium silicate liner, there is some concern of the cytotoxicity of the resin component19 and its ability to adequately hydrate (and release calcium) with the limited fluid released from the pulp.20
BIOACTIVE RESTORATIVES
Unlike liners, bioactive restorative materials are a relatively new concept that lacks a universally accepted clinical definition. In the orthopedic field, bioactive implies that a material will form a layer of calcium phosphate apatite, over which a collagen matrix is deposited by osteoblasts. This process is how bioactive glasses or MTA may form a chemical bond with bone.21 For this reason, the International Standards Organization developed a test for bioactivity of implant surfaces requiring the formation of a calcium phosphate apatite layer (ISO 23317:2012). However, the absence of osteoblasts in dentin prevents this same bonding mechanism from occurring in tooth structure. Some bioactive restorative materials offer the option of forgoing application of an adhesive — the rationale being that calcium phosphate precipitates may occlude the gap between the restorative material and tooth, similar to corrosion products from amalgam. Some laboratory studies have shown calcium phosphate precipitates at the interface between a bioactive restorative material and dentin.22 Early case reports have shown acceptable clinical performance of Activa BioActive Restorative in primary teeth without the use of an adhesive.23 In addition, a 12-month clinical trial of Cention N showed acceptable performance without an adhesive, although the authors noted a tendency toward better margins when an adhesive was used.24 Long-term clinical evidence for using these materials without an adhesive is still lacking. Until the evidence emerges, the prudent clinician should continue to use an adhesive with these materials.
Another claim regarding bioactive restorative materials is the release of calcium, phosphate and fluoride ions. Some authors have suggested this ability is more accurately termed “biointeractivity.”7 The release of fluoride from glass ionomer restoratives has been credited for their decreased incidence of secondary caries.25 New bioactive materials have differentiated themselves from fluoride-releasing glass ionomer, RMGI and giomer (such as Beautifil, Shofu) restoratives by reporting the ability to release calcium and, in some cases, phosphate. If a material elutes the additional ions present in tooth structure (calcium and phosphate), it would, in theory, provide better protection against demineralization. A recent study reported that Cention N released fluoride at a level similar to an RMGI, and fivefold the level of calcium.26 Activa BioActive Restorative has the ability to release and recharge fluoride (albeit at a lower level than an RMGI), and releases low levels of calcium (slightly more than an RMGI).27 At this time, the ion-releasing ability of these materials has not been proven to protect the surrounding tooth structure from secondary caries.
BIOACTIVE CEMENTS
An ideal bioactive cement would offer properties similar to those required of a restorative material — namely, the abilities to form a bond with tooth structure and release ions — although a cement would not require the same mechanical properties. As previously noted, for example, a calcium aluminate material that was initially introduced as a restorative material demonstrated unacceptable clinical fracture rates28 and was later developed into a cement. A laboratory study showed that this calcium aluminate cement (Ceramir C&B) was able to occlude a 50-micron gap by attracting calcium phosphate precipitates.11 This may be beneficial clinically for occluding an open cement gap. A recent laboratory study demonstrated that several cements that were marketed as bioactive had varying levels of calcium release. All cements demonstrated less fluoride release than a reference RMGI cement. Additionally, all cements produced crown retention comparable to a reference RMGI cement.29 Therefore, these new bioactive cements may be capable of calcium release and occluding open margins with calcium phosphate precipitates — however, the clinician should not employ these cements for clinical situations that require adhesively bonded restorations.
CONCLUSION
In light of the foregoing discussion, the judicious use of bioactive materials may offer certain clinical advantages in restorative procedures — although further evidence is needed to substantiate any benefits in these indications. In addition, operators should consider these materials’ possible limitations. As with all care delivery, it is incumbent on the clinician to consider the evidence, as well as specific case demands, when planning treatment.
The author has received grants or research support from Ivoclar, 3M, Dentsply Sirona, GC America, Doxa, Bisco, Danville Materials and Pulpdent.
KEY TAKEAWAYS
- The use of mineral trioxide aggregate (calcium silicate) liners has shown higher success rates than calcium hydroxide liners for direct pulp capping.
- Bioactive calcium silicate liners with fast setting times show promising clinical and histological results, however, additional clinical studies are needed.
- Bioactive restorative materials do not have a proven bond with surrounding tooth structure or evidence of protection from acid challenges. Further evidence is needed to substantiate such claims.
- Bioactive cements may be capable of occluding open margins with calcium phosphate precipitates, but the retention strength of current materials should not be expected to match that of an adhesive resin cement.
References
- Jefferies SR. Bioactive dental materials: composition, properties, and indications for a new class of restorative materials. Inside Dent. 2016;12:58–64.
- Gandolfi MG, Siboni F, Botero T, Bossù M, Riccitiello F, Prati C. Calcium silicate and calcium hydroxide materials for pulp capping: biointeractivity, porosity, solubility and bioactivity of current formulations. J Appl Biomater Funct Mater. 2015;13:43–60.
- Tyas MJ. Pulp protection under restorations — do you need a liner? Aust Endod J. 1998;24:104–108.
- Prati C, Gandolfi MG. Calcium silicate bioactive cements: biological perspectives and clinical applications. Dent Mater. 2015;31:351–370.
- Ferk Luketić S, Malcić A, Jukić S, Anić I, Segović S, Kalenić S. Coronal microleakage of two root-end filling materials using a polymicrobial marker. J Endod. 2008;34:201–203.
- Torabinejad M, Hong CU, McDonald F, Pitt Ford TR. Physical and chemical properties of a new root-end filling material. J Endod. 1995;21:349–353.
- Gandolfi MG, Siboni F, Prati C. Chemical-physical properties of TheraCal, a novel light-curable MTA-like material for pulp capping. Int Endod J. 2012;45:571–579.
- Wang X, Sun H, Chang J. Characterization of Ca3SiO5/CaCl2 composite cement for dental application. Dent Mater. 2008;24:74–82.
- Simila HO, Karpukhina N, Hill RG. Bioactivity and fluoride release of strontium and fluoride modified Biodentine. Dent Mater. 2018;34:e1–e7.
- Engqvist H, Schultz-Walz JE, Loof J, et al. Chemical and biological integration of a mouldable bioactive ceramic material capable of forming apatite in vivo in teeth. Biomaterials. 2004;25:2781–2787.
- Jefferies SR, Fuller AE, Boston DW. Preliminary evidence that bioactive cements occlude artificial marginal gaps. J Esthet Restor Dent. 2015;27:155–166.
- Hilton TJ. Keys to clinical success with pulp capping: a review of the literature. Oper Dent. 2009;34:615–625.
- Hilton TJ, Ferracane JL, Mancl L; Northwest Practice-based Research Collaborative in Evidence-based Dentistry (NWP). Comparison of CaOH with MTA for direct pulp capping: a PBRN randomized clinical trial. J Dent Res. 2013;92 (Suppl 7):16S–22S.
- Kundzina R, Stangvaltaite L, Eriksen HM, Kerosuo E. Capping carious exposures in adults: a randomized controlled trial investigating mineral trioxide aggregate versus calcium hydroxide. Int Endod J. 2017;50:924–932.
- Nair PN, Duncan HF, Pitt Ford TR, Luder HU. Histological, ultrastructural and quantitative investigations on the response of healthy human pulps to experimental capping with mineral trioxide aggregate: a randomized controlled trial. Int Endod J. 2008;41:128–150.
- Nowicka A, Lipski M, Parafiniuk M, et al. Response of human dental pulp capped with biodentine and mineral trioxide aggregate. J Endod. 2013;39:743–747.
- Cannon M, Gerodias N, Viera A, Percinoto C, Jurado R. Primate pulpal healing after exposure and TheraCal application. J Clin Pediatr Dent. 2014;38:333–337.
- Koubi G, Colon P, Franquin JC, et al. Clinical evaluation of the performance and safety of a new dentine substitute, Biodentine, in the restoration of posterior teeth — a prospective study. Clin Oral Investig. 2013;17:243–249.
- Jeanneau C, Laurent P, Rombouts C, Giraud T, About I. Light-cured tricalcium silicate toxicity to the dental pulp. J Endod. 2017;43:2074–2080.
- Camilleri J, Laurent P, About I. Hydration of Biodentine, Theracal LC, and a prototype tricalcium silicate-based dentin replacement material after pulp capping in entire tooth cultures. J Endod. 2014;40:1846–1854.
- Niu LN, Jiao K, Wang TD, et al. A review of the bioactivity of hydraulic calcium silicate cements. J Dent. 2014;42:517–533.
- Garcia-Godoy F, Morrow BR. Profilometry bioactive dental materials analysis and evaluation of dentin integration. J Dent Res. 2016;95 (Spec Iss A):1828.
- Croll TP, Berg JH, Donly KJ. Dental repair material: a resin-modified glass-ionomer bioactive ionic resin-based composite. Compend Contin Educ Dent. 2015;36:60–65.
- Lawson NC, Burgess JO, Fu CC, Robles A, Givan DA. Clinical evaluation of an amalgam replacement restorative material: 12-month recall. J Dent Res. 2018;97 (Spec Iss A):0234.
- Mickenautsch S, Yengopal V. Absence of carious lesions at margins of glass-ionomer cement and amalgam restorations: an update of systematic review evidence. BMC Res Notes. 2011;4:58.
- Kulkarni P, Farheen F, Lawson NC, Burgess JO. Ion release and flexural strength of bioactive materials. J Dent Res. 2018;97 (Spec Iss A):1160.
- May E, Donly KJ. Fluoride release and re-release from a bioactive restorative material. Am J Dent. 2017;30:305–308.
- Van Dijken JW, Sunnegårdh-Grönberg K. A three year follow-up of posterior doxadent restorations. Swed Dent J. 2005;29:45–51.
- Reznik J, Kulkarni P, Shah S, et al. Crown retention strength and ion release of various bioactive cements. J Dent Res. 2018;97 (Spec Iss A):0656.
Featured image by MARKUS BRUNNER/GNU FREE DOCUMENTATION LICENSE
From Decisions in Dentistry. April 2018;4(4):24-27.


