PORNPAK KHUNATORN/ISTOCK/GETTY IMAGES PLUS
PORNPAK KHUNATORN/ISTOCK/GETTY IMAGES PLUS
Consequences of Tooth Clenching
This common parafunctional behavior may have long-term consequences on the central nervous system.
Part 2 of a two-part series
The first article in this two-part series, “The Problem With Bruxism” (available here), discusses how terminology related to grinding and clenching can lead to misunderstandings in clinical practice. This concluding installment focuses on the consequences of persistent temporomandibular disorders on the central nervous system, and explores the concept that tooth clenching during sleep may have both beneficial and harmful consequences.
Published in the September 2020 issue, the initial installment of this two-part series (available here) described the misunderstandings that arise when the word bruxism is used because different authors define bruxism in different ways. In 2000, Christenson1 separated tooth clenching from bruxism by asserting bruxism meant tooth grinding, but not tooth clenching. The common oral parafunctions of grinding and clenching are sufficient to describe the consequences that result from their actions. Clenching is most important in studies of both non-temporomandibular disorder (TMD) subjects, as well as patients with acute and chronic TMD.
This concluding installment will highlight the central nervous system consequences of persistent peripheral myofascial pain. In addition, tooth clenching during sleep appears to have both beneficial and harmful consequences. Moreover, low level electromyography (EMG) background muscle activity offers evidence of the nature of clenching during sleep, as well as the intraoral soft tissue formations of linea alba and scalloped tongue.
Central Pain Matrix and Peripheral. Dysfunction
The practicing dentist may benefit from this reminder for care: Pain is not a peripheral phenomenon. Pain happens in the brain. Nociception is the neural activity initiated by intense mechanical, chemical or thermal stimulation to peripheral tissue. These stimuli are transduced to nerve impulses in peripheral nociceptors that propagate to the central nervous system by action potentials, which, in turn, modulate second and third order transmission nociceptors. The ultimate target of this sequence of nociceptive activity is the perception areas located in higher centers of the brain.
In the trigeminal system, there are two pathways to the perception areas of the brain. First is the sensory-discriminative pathway that terminates in the posterior central gyrus of the brain, where intensity and location of the intense peripheral stimuli are perceived. Second is the affective-motivational pathway that terminates in various limbic and neocortical nuclei, which influence the emotion and stress of the perceived threatening stimulus. Together, the trigeminal sensory-discriminative and affective-motivational pathways are integral to the neuromatrix model of trigeminal pain.
Melzack2 put forth a genetically determined neuromatrix model by proposing that sensory experience of persistent myofascial pain initiated by muscular nociceptors converges on, and modulates, the central neuromatrix, which is the primary neural network that generates neural patterns which produce pain. What follows are dysfunctional changes within the motor centers of the neuromatrix that evoke excitatory changes in the peripheral musculature — seen as muscle splinting, a reduction in normal resting muscle length, and the maintenance of a dysfunctional muscle condition.3–5
Particular interest lies in the medial pain system of the trigeminal nerve, otherwise called the affective-motivational pain system, which includes, in part, ascending trigeminal input to the pain matrix network.6 The affective-motivational system modulates neurophysiological responses to long-standing biopsychosocial activity.7 The neuroplasticity of the pain matrix network modulated by motor-evoked jaw pain may aid in understanding the relationship between chronic TMD pain and physical disability.8
Recent findings from the orofacial pain prospective evaluation and risk assessment (OPPERA) project indicate the state of chronic TMD varies over time.9 Individuals experiencing chronic TMD before and following initial examination show a tendency to improve in Axis II disabilities of psychosocial variables, such as affective trait disposition. In addition, improvement was observed in regard to Axis I clinical variables, such as jaw function and somatic symptoms. Likewise, in a 5-year study, 49% of the individuals with a history of TMD symptoms before the study reported an absence of TMD pain in the final evaluation at the end of the study, and the maximal mouth opening was essentially unchanged.10 On the other hand, what about the remaining 51% of patients with TMD who may have a significant parafunctional tooth clenching habit? If myofascial pain associated with clenching becomes chronic, increasing pain can be produced throughout the day11 and may persist for years.12 Utilizing an elegant chronic jaw pain model, Wang et al8 demonstrated that submaximal tooth clenching is associated with motor-evoked jaw pain for patients with chronic TMD, as compared to a healthy non-TMD control group. Moreover, long-term pain duration in patients with TMD increases the probability of developing comorbidities, including central sensitization.13,14
Tooth Clenching and Sleep Background Activity
Awake tooth-together positioning of the mandible may be a function of tooth clenching during sleep. The persistence of low level EMG background muscle activity (as when a muscle is in a particular static contracted position, which occurs during clenching) while asleep is, in part, responsible for motor-evoked myofascial pain during wakefulness and sleep.15 Clenching during sleep has been cited as an important comorbidity among sleep-time oral parafunctions.16 The elevator muscles of the mandible that are active during sleep (even at a minimal submaximal EMG level of 10 mv) may maintain that level during the day.17 Clinically, many patients with TMD report a tooth-together jaw position during waking hours without being aware of forcefully clenching their teeth.
Even though background silent nonrhythmic EMG activity of the masticatory musculature during sleep has not been studied in detail, clenching while asleep may play a role in the perpetuation of TMD, and denote a risk factor for its development.18 Background EMG recordings made during clenching episodes while asleep may represent jaw overuse activity that leads to myofascial pain. Patients who clench during sleep and who have not been observed to grind their teeth complain of awakening in the morning, or sometimes awakening during sleeping hours, with jaw stiffness.19 Raphael et al20 found that a low hypertonic state of background EMG masticatory muscle activity during sleep in patients with myofascial TMD was elevated compared to non-TMD control subjects.
Elevated EMG activity of the closure muscles of the mandible during sleep background activity which occurs not during, but in between rhythmic masticatory muscle activity (RMMA) may have more clinical relevance to the development and perpetuation of TMD than elevated EMG activity found during RMMA events. Lavigne et al21 postulate that RMMA is associated with a series of motor and autonomic-cardiac interactions in the setting of sleep disordered breathing. The background masticatory muscle activity occurring separately from RMMA events during sleep appears to be much the same as low level bite forces produced by clenching or a teeth-together jaw position during waking hours.18 Thus, high levels of masticatory muscle activity in patients with TMD may not be necessary to produce persistent trigeminal nociceptive potentials at the segmental level that ultimately progress to central sensitization and chronic pain.
Beneficial Versus Harmful Effects of Clenching
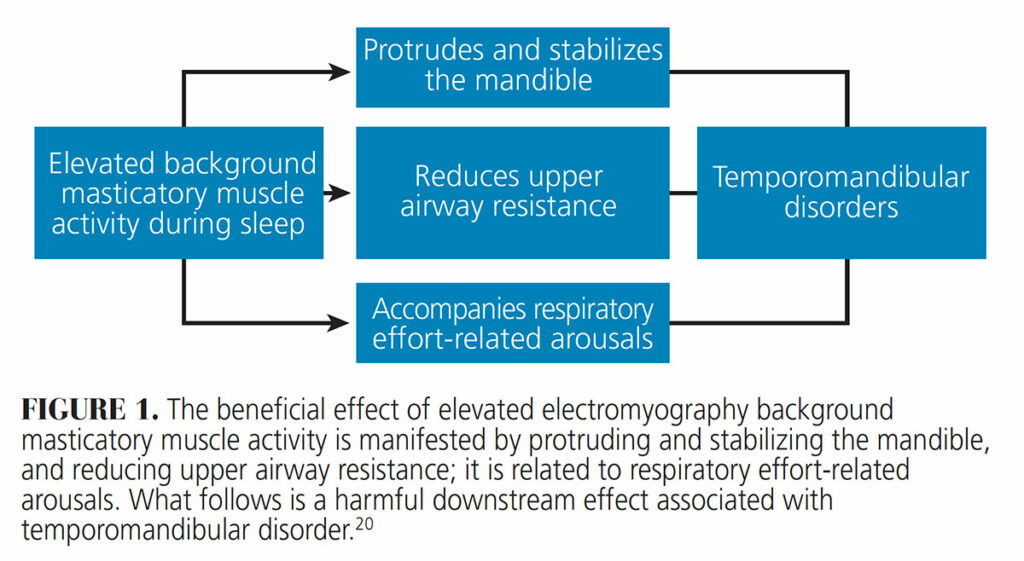
A hypothesis has developed in sleep physiology positing that low level EMG background activity of the masticatory muscles during sleep (analogous to low intensity EMG activity of clenching or tooth-to-tooth contact), even in the absence of RMMA events, may be beneficial by reducing upper airway resistance, thus contributing to the restoration of a patent upper airway.18 Moreover, contraction of the masseter muscle coincident with the activation of the muscles of respiration during arousals or awakenings is said to play a role in reestablishing a patent upper airway in the setting of obstructive sleep apnea.22 In addition, Yoshida23 demonstrated the masseter muscle exhibited lower EMG levels during obstructive apnea events, and higher EMG levels between apneic events. The higher EMG levels following apneic events may play a role in reestablishing patency of the upper airway.
 Raphael et al20 suggested that background masticatory muscle activity during sleep has the beneficial effect of reducing upper airway resistance — though linked to a harmful downstream effect of TMD (Figure 1). Clenching during sleep may be beneficial in regard to sleep disordered breathing, whereas clenching while asleep may be harmful by acting to initiate and/or perpetuate TMD. The mechanism(s) by which a beneficial role of low level background activity of the masticatory muscles leads to the harmful role associated with TMD pain onset has yet to be clarified.
Raphael et al20 suggested that background masticatory muscle activity during sleep has the beneficial effect of reducing upper airway resistance — though linked to a harmful downstream effect of TMD (Figure 1). Clenching during sleep may be beneficial in regard to sleep disordered breathing, whereas clenching while asleep may be harmful by acting to initiate and/or perpetuate TMD. The mechanism(s) by which a beneficial role of low level background activity of the masticatory muscles leads to the harmful role associated with TMD pain onset has yet to be clarified.
In some cases, clenching-induced background masticatory muscle activity may not be sufficient to protrude and stabilize the mandible or reduce upper airway resistance, yet still be sufficient to perpetuate, and denote a risk factor for, TMD. Other factors may dominate as risks for upper airway resistance, such as a high Mallampati score or other upper airway anatomical problems. The clinical correlate for the presence of overwhelming negative factors is an initial examination protocol that includes a thorough sleep assessment for every patient with TMD.
Muscle guarding may play a role in generating background EMG activity during sleep. In the setting of acute jaw pain, muscle guarding occurs as a result of sustained involuntary muscle contraction that limits jaw movement, thus acting as a protective mechanism in order to avoid noxious or harmful mechanical stimuli during movement and promote healing. Muscle guarding with the teeth separated or the mandible held rigid produces more activity in the suprahyoid or depressor muscles of the mandible compared to the masseter and temporalis or elevator muscles.24 On the other hand, in the setting of chronic jaw pain, the protective aspect of muscle guarding is often no longer operative, thus creating and perpetuating jaw discomfort in its struggle to maintain diminished upper airway resistance.25 Persistent muscle guarding has the potential to produce sufficient elevator muscle activity to create a tooth-together position that results in low bite force levels similar to non-RMMA background activity during sleep. In this respect, when the mouth is closed during persistent muscle guarding, the pain experienced by patients with TMD may be accentuated by continuous isometric activity of the elevator and depressor muscles of the mandible.
Linea Alba and Scalloped Tongue
The etiologies of linea alba and scalloped tongue are controversial. Though the preponderance of evidence suggests the diagnosis of clenching during sleep or while awake is associated with the presence of linea alba and/or scalloped tongue, a cause-and-effect relationship has not been established.26 Several studies have suggested that linea alba and scalloped tongue are the result of oral parafunctional habits, such as grinding or clenching.19,27,28
Clinical experience gives evidence that grinding and clenching are distinct conditions with different consequences.26,28 Paesani29 stated that scalloped tongue may be related to “force that the tongue exerts against the dental arches simultaneously with clenching.” Grinding is a condition manifested as a jaw movement behavior, whereas clenching is an isometric non-movement jaw condition. Neither linea alba nor scalloped tongue have a causal relationship to grinding because the jaw is moving during tooth grinding. A moving mandible does not produce the static oral environment necessary to form the stereotyped linear architecture of hyperkeratosis on the buccal mucous membrane juxtaposed to the occlusal plane, or the curvilinear scalloped appearance of the tongue juxtaposed to the palatal and lingual surfaces of the dental arches.
It should not be disturbing to consider that clenching during sleep may have a beneficial effect on sleep disordered breathing. Raphael et al20 theorized that elevated background masticatory muscle activity during sleep protrudes and stabilizes the mandible and reduces upper airway resistance (Figure 1). In addition, a beneficial effect may also manifest during brief episodes of phasic tooth grinding in the context of RMMA. In a recent retrospective polysomnographic laboratory study, approximately one-third of adult patients with obstructive sleep apnea demonstrated phasic grinding during RMMA, leading the authors to suggest “a possible protective role of RMMA in respiratory-related arousals.”30
In the absence of a protruded and stabilized mandible, one of the most important biomechanical events in the setting of obstructive sleep apnea is the collapse of the tongue posteriorly, thus increasing upper airway resistance. The presence of scalloped tongue indicates the tongue is advanced as far forward as possible, or, at the very least, demonstrates the tongue has stabilized itself in a tonic position against the palatal and lingual surfaces of the posterior teeth. This would prevent the posterior portion of the tongue from collapsing into the oral pharynx. Any stable forward positioning of the tongue would be an advantage in maintaining a patent airway. What keeps the tongue stable and forward? The genioglossus muscle is the only extrinsic muscle of the tongue that holds or advances the tongue. Yet the genioglossus muscle weakens with age in concert with all other skeletal muscles. Consequently, the tongue is more apt to collapse posteriorly over time. Strengthening the genioglossus muscle provides increased muscle tone, thus aiding in unconsciously holding the tongue forward during sleep. The authors of this article have developed a home-care exercise program designed to strengthen the genioglossus muscle.31 Every one of our patients whom we treat for obstructive sleep apnea or upper airway resistance syndrome is trained and encouraged to perform this daily exercise program.
Additional Clinical Concerns
There may be common concerns of the practicing dentist related to clenching and grinding, including the etiology of abfractions and relationship between malocclusion and TMD. Abfractions are caries-free lesions or defects located at the cementoenamel junction on the buccal aspect of either the maxillary and/or mandibular teeth. While the etiology is unclear,32 the causal antecedent of abfractions appears to be excessive occlusal loading.33–35 Tooth clenching is a logical cause of excessive occlusal loading. Grinding may be a factor in the formation of abfractions if there is evidence of associated tooth wear facets on the affected teeth.
The general agreement in the dental community that malocclusion is not a primary etiological factor for TMD was established approximately 30 years ago36 and reinforced in recent years.33 Evidence supporting a causal relationship between malocclusion and TMD pain is unconvincing.37
Conclusion
The meaning of bruxism is problematic given that various authors render bruxism to include only tooth grinding, only clenching, or both grinding and clenching.38,39 Assuming that bruxism is an adequately explicit, indicatively descriptive word, the term itself violates the law of identity. The reader is led to confusion as to whether a particular author means grinding and/or clenching since these two oral parafunctions have different identities.28,40 In scientific language, a descriptive term should not have two identities.
In clinically applied patient language, use of the terms tooth clenching and tooth grinding are sufficiently explicit. By avoiding the ambiguous term bruxing, and separating clenching from grinding, patients diagnosed with TMD have a better understanding when receiving an explanation of the cause(s) and appropriate treatment(s). Further study on clenching should include a rigorously conducted statistical meta-analysis. This application of the power of statistical methodology to the entire volume of tooth clenching literature may lead to alternative conclusions.
Key Takeaways
- Tooth clenching during sleep appears to have both beneficial and harmful consequences.
- Wang et al8 demonstrated that submaximal tooth clenching is associated with motor-evoked jaw pain for patients with chronic temporomandibular disorder (TMD).
- Tooth clenching while asleep may play a role in the perpetuation of TMD, and denote a risk factor for its development.18
- Raphael et al20 theorized that elevated background masticatory muscle activity during sleep protrudes and stabilizes the mandible and reduces upper airway resistance.
- Although the preponderance of evidence suggests the diagnosis of clenching during sleep or while awake is associated with the presence of linea alba and/or scalloped tongue, a cause-and-effect relationship has not been established.26
References
- Christenson GJ. Treating bruxism and clenching. J Am Dent Assoc. 2000;131:233–235.
- Melzack R. Pain and the neuromatrix in the brain. J Dent Educ. 2001;65:1378–1382.
- Hodges PW, Tucker K. Moving differently in pain: A new theory to explain the adaptation to pain. Pain. 2011;152:590–598.
- Farina D, Negro F, Gizzi L, Falla D. Low-frequency osscillations of the neural drive to the muscle are increased with experimental muscle pain. J Neurophysiol. 2012;107:958–965.
- Falla D, Farina D. Neuromuscular adaptation to experimental and clinical neck pain. J Electromyogr Kinesiol. 2008;18:255–261.
- Apkarian AV, Bushnell MC, Treede RD, Zubieta JK. Human brain mechanisms of pain perception and regulation in health and disease. Eur J Pain. 2005;9:463–484.
- Dworkin SF. The case for incorporating bio-behavioral treatment into TMD management. J Am Dent Assoc. 1996;127:1607–1610.
- Wang W-E, Roy A, Misra G, et al. Motor-evoked pain increases force variability in chronic jaw pain. J Pain. 2018;19:636–648.
- Fillingim RB, Slade GD, Greenspan JD, et al. Long-term changes in biopsychosocial characteristics related to temporomandibular disorder: findings from the OPPERA study. Pain. 2018;159:2403–2413.
- Ohrbach R, Dworkin SF. Five-year outcomes in TMD: relationship of changes in pain to changes in physical and psychological variables. Pain. 1998;74:315–326.
- Annino DJ, Goguen LA. Pain from the oral cavity. Otolaryngol Clin N Am. 2003;36:1127–1135.
- Hublin C, Kaprio J, Partinen M, Koskenvuo M. Sleep bruxism based on self-report in a nationwide twin cohort. J Sleep Res. 1998;7:61–67.
- Dahan H, Shir Y, Velly A, Allison P. Specific and number of comorbidities are associated with increased levels of temporomandibular pain intensity and duration. J Headache Pain. 2015;16:528.
- Nguyen TT, Vanichanon P, Bhalang K, Vongthongsri S. Pain duration and intensity are related to coexisting pain and comorbidities present in temporomandibular disorder pain patients. J Oral Facial Pain Headache. 2019;33:205–212.
- Ohrbach R, McCall WD Jr. The stress-hyperactivity-pain theory of myogenic pain. Proposal for a revised theory. Pain Forum. 1996;5:51–66.
- Glaros AG, Tabacchi KN, Glass EG. Effect of parafunctional clenching on TMD pain. J Orofac Pain. 1998;12:145–152.
- Carra MC, Huynh N, Lavigne G. Sleep bruxism: a comprehensive overview for the dental clinician interested in sleep medicine. Dent Clin N Am. 2012;56:387–413.
- Essick GK, Raphael KG, Sanders AE, Lavigne G. Orofacial pain and temporomandibular disorders in relation to sleep disorder breathing and sleep bruxism. In: Kryger M, Roth T, Dement WC, eds. Principles and Practice of Sleep Medicine. 6th ed. Philadelphia: Elsevier; 2017:1435–1444.
- Huynh N, Lavigne GJ, Okura K, et al. Sleep bruxism. In: Montagna P, Chokroverty S, eds. Handbook of Clinical Neurology. Vol. 99 (3rd series) Sleep Disorders Part 2. Philadelphia: Elsevier; 2011.
- Raphael KG, Janal MN, Sirois DA, et al. Masticatory muscle sleep background electromyographic activity is elevated in myofacial TMD patients. J Oral Rehabil. 2013;40:883–891.
- Lavigne GJ, Rompre PH, Poirier G, et al. Rhythmic masticatory muscle activity during sleep in humans. J Dent Res. 2001;80:443–448.
- Kato T, Katase T, Yamashita S, et al. Responsiveness of jaw motor activation to arousals during sleep in patients with obstructive sleep apnea. J Clin Sleep Med. 2013;9:759–765.
- Yoshida KA. A polysomnographic study on masticatory and tongue muscle activity during obstructive and central sleep apnea. J Oral Rehabil. 1998;25:603–609.
- Ohrbach R, Markiewicz MR, McCall WD Jr. Waking-state oral parafunctional behaviors: specificity and validity as assessed by electromyography. Eur J Oral Sci. 2008;116:438–444.
- Woolf CJ. What is this thing called pain? J Clin Invest. 2010;120:3742–3744.
- Klasser GD, Rei N, Lavigne GJ. Sleep bruxism etiology: The evolution of a changing paradigm. J Can Dent Assoc. 2015;81:f2.
- Koyano K, Tsukiyama Y, Ichiki R, Kuwata T. Assessment of bruxism in the clinic. J Oral Rehabil. 2008;35:495–508.
- Lobbezoo F, Koyano K, Paesani DA, Manfredini D. Sleep bruxism: diagnostic considerations. In: Kryger M, Roth T, Dement WC, eds. Principles and Practice of Sleep Medicine. 6th ed. Philadelphia: Elsevier; 2017:1427–1434.
- Paesani DA. Diagnosis of bruxism. In: Bruxism: Theory and Practice. Chicago: Quintessence Publishing Co; 2010:28.
- Tan MW, Yap AU-J, Chua AP, Wong JC, Parot MV. Prevalence of sleep bruxism and its association with obstructive sleep apnea in adult patients: a retrospective polysomnographic investigation. J Oral Facial Pain Headache. 2019;33:269–277.
- Fuselier B, Loughner B. The Einstein Method. Available at: https://issuu.com/orlandomedicalnews/docs/orl_oct2017_digitaledition. Accessed November 13, 2020.
- Sarode GS, Sarode SC. Abfraction: A review. J Oral Maxillofac Pathol. 2013;17:222–227.
- Okeson JP. Management of Temporomandibular Disorders and Occlusion. 6th ed. St. Louis: Mosby; 2008:143.
- Bernhardt O, Gesch D, Schwahn C, et al. Epidemiological evaluation of the multifactorial aetiology of abfractions. J Oral Rehabil. 2006;33:17–25.
- Miller N, Penaud J, Ambrosini P, Bisson-Boutelliez C, Briancon S. Analysis of etiologic factors and periodontal conditions involved with 309 abfractions. J Clin Periodontol. 2003;30:828–932.
- Gaynor G. Orthodontic Therapy. In: Kaplan AS, Assael LA, eds. Temporomandibular Disorders: Diagnosis and Treatment. Philadelphia: Sanders Co; 1991:579–580.
- Lobbezoo F, Lavigne G. Do bruxism and temporomandibular disorders have a cause and effect relationship? J Orofac Pain. 1997:11:15–23.
- American Academy of Sleep Medicine. International classification of sleep disorders. AASM Sleep Related Bruxism. 3rd ed. Darien, Ill: AASM; 2014:303–311.
- Rugh JD, Harlan J. Nocturnal bruxism and temporomandibular disorders. In: Jankovic J, Tolosa E, eds. Advances in Neurology. New York, NY: Raven Press; 1988;49:329–341.
- Carlsson GE, Egermark I, Magnusson T. Predictors of bruxism, other oral parafunctions, and tooth wear over a 20-year follow-up period. J Orofac Pain. 2003;17:50–57.
From Decisions in Dentistry. December 2020;6(11): 16-18, 21.