Clinical Insights Into Managing Pediatric Pain
Successful procedural outcomes depend on effective pain management — and this is especially true when treating children.
This course was published in the April 2016 issue and expires 04/30/19. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
OBJECTIVES
After reading this course, the participant should be able to:
- Identify strategies for assessing pain levels in children.
- Discuss behavior management techniques when administering
local anesthesia to pediatric patients. - Explain strategies for the effective administration of local
anesthesia to children. - List the roles of nitrous oxide and analgesics in pain
management for this patient group.
Pain is the prototypical psychological response to nociceptive stimuli. Response to a stimulus varies greatly from one person to another and the characterization of pain in an individual can vary over time. The delivery of quality oral health care relies heavily on the ability to prevent and relieve pain. Controlling pain is the key to successful dental treatment of nearly all patients, particularly children. It wasn’t long ago that many in the medical community doubted that children, particularly infants, were susceptible to pain.1 This notion has been disproven, and recent studies suggest that children begin to show developmental changes in response to painful stimuli early in infancy.2
When treating adult patients, dentists rely heavily on reported symptoms and detailed descriptions of pain quality and intensity. Pediatric patients — particularly preschool-age children and those with special health care needs — may not be able to provide such descriptions. Fortunately, there are several pain scales that can be used by children to report pain, such as the Wong-Baker scale. This tool uses a series of faces that children can choose from to reflect the level of pain felt at the time.3 When a visual scale such as this cannot be employed due to lack of comprehension, oral health professionals must rely on behavior and voice intonation. When it is determined that a child is experiencing orofacial pain, a detailed history should be obtained and documented in the patient record.
The potential effects of emotion on pain perception are sometimes overlooked. Anxiety and emotional distress can decrease an individual’s pain threshold, thereby increasing the magnitude of a patient’s reaction to a painful stimulus.4 Oral health professionals are more likely to provide pain-free dentistry when the patient is calm. Other factors are also involved in preparing for a dental visit, including the presence of parents/caregivers —as well as their behavior during treatment.
The presence of parents/caregivers during the dental visit may affect a child’s demeanor and behavior in positive or negative ways. Consider a young child’s reaction to a minor injury, such as a fall resulting in an abrasion. If the parent/caregiver responds in a concerned, over-excited or scared fashion, it is likely the child will react in kind. If the parent/caregiver remains calm, the child may brush off this minor injury and avoid an emotional breakdown. A skinned knee is not exactly analogous to a dental visit, but the idea is similar. Children look to parents/caregivers for direction and take emotional cues from them. This is not to say that parent/caregiver presence is always negative during dental appointments.5 Many children do better with parents/caregivers present, but it is important that parents/caregivers remain supportive and keep their own anxiety in check. Anxious behavior may accentuate children’s perceptions of pain.6
BEHAVIOR MANAGEMENT
The use of basic behavior management techniques when treating young patients is key to pain control in the dental office. While myriad behavior management options exist, most children respond well to the use of simple requests and commands. “Tell-Show-Do” is the bedrock approach for behavior management in children.7 Explaining what is about to happen (using age-appropriate terms), showing the child what will be used, and then proceeding with treatment as described will allow practitioners to treat most children with a high level of success. These simple interactions also allow a calm rapport to be built. This is important because, as noted, children who are anxious are more likely to have painful responses to stimuli.
Another simple and vital part of behavior management is word choice. Children, particularly preschoolers, are very sensitive to the words used to describe an instrument or procedure. While using words like “Mr. Whistle” and “tooth counter” may seem silly, these terms are much more benign to a timid child than “ultrasonic scaler” and “explorer.” Using words that conjure positive thoughts and happy emotions can go a long way toward decreasing anxiety.
When treating children, it seems that every appointment has one or more “make or break” moments. The injection of local anesthetic and the initiation of a cavity preparation are two common hurdles for successful dental appointments. Both of these procedures have the potential to cause significant anxiety due to the noxious stimuli that often accompany them. Anticipating the moments in the treatment interval that may be difficult for children is important so the provider can anticipate and prepare for unwanted behaviors.
Unfortunately, many children view a visit to the dentist as a frightening experience.8 Numerous items in a dental office may prove to be challenging for pediatric patients. A startling noise from a scaler, the sharp hook of an explorer, and a bright glare from the overhead light are all potential pitfalls for an anxious child. As such, providers need to be aware of how these objects and stimuli may be viewed in order to minimize patient anxiety.
LOCAL ANESTHESIA
Many of the tasks performed in dentistry have the potential to cause discomfort. Local anesthesia blocks nerve impulses, thereby potentially altering the perception of pain. To review, a noxious stimulus (such as an injection through the skin or mucus membranes) activates A-delta and C fibers. Such fibers may be nociceptive, and the impulses transmitted along these fibers, when integrated with other sensory input and psychic phenomena in the central nervous system, may cause pain. When a local anesthetic is delivered, it diffuses into the extensions of nerve cells (axons and/or dendrites) and temporarily interrupts the conduction of action potentials by preventing the inflow of sodium ions.9 The smaller fibers are shut down before the larger fibers, so the sensation of pain is blocked first, with the sensation of pressure being blocked last, if at all, as this is carried by a larger fiber.
The administration of local anesthesia can be a frightening experience for patients of all ages. Many individuals have a fear of needles; as a result, fear of local anesthesia is legitimate and should be treated as such.10,11
The first step to providing good local anesthesia is to use a topical anesthetic, usually benzocaine, to dry mucosa in the area where the injection will take place. It is important that the tissue be dry to prevent the benzocaine from washing away rather than being absorbed by the tissue. The benzocaine should be kept in contact with the tissue for at least 1 minute prior to injection. The second step is to ensure that patients, especially young children, do not see the syringe, which can look quite frightening. Oral health professionals should also try to distract pediatric patients so they are not focused on the impending injection.
The prevention of adverse events during an injection starts with stabilizing the patient’s head. Anticipation of unwanted movements by the oral health professional and the dental assistant can help prevent injury, as well. In order to avoid needle separation, the needle must not be placed too deeply into the soft tissue upon injection. The provider should always be able to see a segment of the needle outside of the soft tissue, that way if the needle were to separate (most likely at the “hub” of the needle), it could be retrieved. Controlling the patient’s head movement and remaining aware of needle placement will help prevent needle separation or other soft tissue trauma that can be caused by sudden movement.
The anesthetic technique used for children is different from that indicated for adults. The main difference is in the landmarks used when performing inferior alveolar nerve (IAN) blocks, which provide pulpal anesthesia to a mandibular quadrant. The position of the mandibular foramen in children is relatively lower than in adults, located just above the occlusal plane in the primary dentition. This position will change as the child ages due to mandibular growth. Upon maturity, it will be located more than 5 mm above the occlusal plane. If a block is unsuccessful, the provider should insert the needle higher on the ramus than the previous attempt, keeping in mind maximum dosages (based on patient’s weight) to prevent an overdose.12
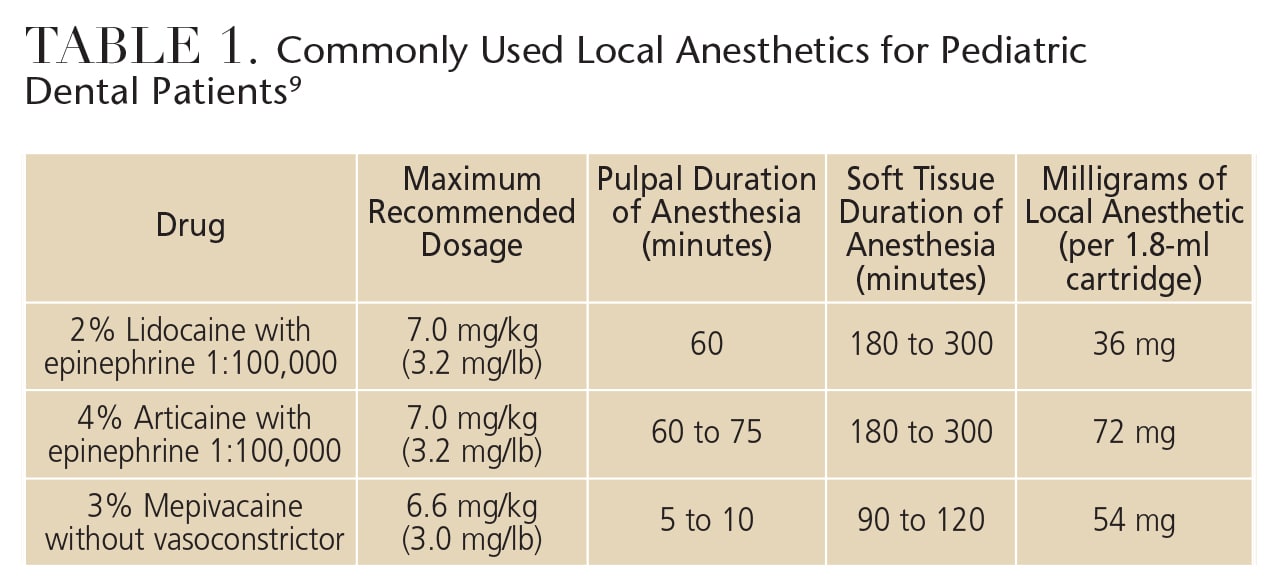
Table 1 provides an overview of the anesthetics used in pediatric dentistry.9 The most commonly used local anesthetic agent in children is 2% lidocaine with epinephrine 1:100,000.13 This anesthetic has a long track record of safe and effective use in pediatric dental populations. The vasoconstrictor, epinephrine, is added to cause constriction of the blood vessels in the area where the injection occurs, thereby keeping more of the anesthetic solution localized and slowing the systemic uptake of the solution. This provides longer anesthesia in the area while reducing the risk of overdose.14
Other agents used for local anesthesia in children include 4% articaine with epinephrine and 3% mepivacaine without vasoconstrictor. Mepivacaine (without vasoconstrictor) is not recommended when treating multiple areas in pediatric patients because the lack of a vasoconstrictor increases the likelihood of overdose and the duration of pulpal anesthesia is very brief (five to 10 minutes).
In recent years, 4% articaine — a local anesthetic with a unique chemical makeup that manifests characteristics of both ester and amides — has grown in popularity. Many dental professionals claim this drug works faster than lidocaine and provides more profound anesthesia. There are concerns, however, because articaine has a higher rate of persistent paresthesia when used for IAN blocks.15 Avoiding block anesthesia with 4% articaine may be prudent, but there are other uses for the drug in pediatric dentistry.9
The use of 4% articaine may eliminate the need to provide block anesthesia in some instances, reducing the risk of soft tissue trauma. Sharaf16 reported that mandibular buccal infiltration in children (ages 3 to 9) was as effective as IAN blocks for all restorative situations with the exception of a pulpotomy on a second primary molar. This anesthetic technique should be used with caution because the likelihood of successful local anesthesia decreases as a child grows older. Studies have also shown that local infiltration of 4% articaine can improve the effectiveness of IAN blocks.17
The administration of local anesthesia is not without risk — the most serious being the possibility of overdose in pediatric patients. Knowing each patient’s weight and calculating a maximum dosage of local anesthetic prior to treatment are important considerations. The risk of overdose or other adverse reactions can also be diminished by aspirating prior to injection, which verifies that an intravascular injection is not going to be administered. While an adult’s large body mass reduces the risk of overdose in this population, it is much easier to reach local anesthetic maximums in children.
Another major concern regarding local anesthetic use in children is postoperative complications, such as lip biting. Children are often not used to the feeling of being numb and may respond to this strange sensation by chewing or biting on the lip. This can result in significant swelling and trauma to the area. When children traumatize their lip following a dental procedure, the swelling and irritation can be misinterpreted by parents and medical providers as an infection or allergic reaction to local anesthetic, leading to inappropriate treatment.18 Proactively preparing parents/caregivers for the possibility of lip biting, as well as discussing prevention methods, are of paramount importance. Unfortunately, it can be very difficult to prevent lip biting among preschool-age children, and they require close surveillance for as long as the soft tissue anesthesia persists.
Recently, a drug (phentolamine mesylate) that shortens the duration of soft tissue anesthesia was approved for use in the United States.19 Early studies have shown that it is successful in reversing the effects of anesthesia and shortening the duration of time that lips, tongue and cheeks are numb following the administration of local anesthesia.19 This is a potentially useful drug that can aid in patient comfort and limit the possibility of trauma caused by involuntary lip biting. The administration of phentolamine mesylate seems to be a great fit for individuals with special health care needs and young children who may be more prone to self-injurious behavior and soft tissue trauma following a procedure. At this time, however, it is not approved for use on children younger than 6 due to a lack of research in this age group.
NITROUS OXIDE SEDATION
Along with the basic behavior management techniques, many oral health professionals use pharmacological methods of managing behavior and preventing pain. The most commonly used anti-anxiety medication for pediatric dental patients is nitrous oxide. Nitrous oxide is safe, easy to administer, and provides some anti-anxiety effects. Its use, however, can cause nausea and vomiting.20 This risk can be reduced by instructing patients to avoid eating large meals before the procedure, keeping the concentration of nitrous oxide as low as possible, and avoiding large fluctuations in nitrous oxide dose rates. The desired effect is that the patient is content, comfortable, and has somewhat blunted sensation, making the injection easier to accomplish.21
MANY DENTAL PROFESSIONALS CLAIM THIS DRUG WORKS FASTER THAN LIDOCAINE AND PROVIDES MORE PROFOUND ANESTHESIA
The key to the successful use of nitrous oxide is patient selection. Patients must be willing to wear the nasal hood and breathe through their nose to achieve the desired effect. If patients are unable to breathe freely through the nose or they are sobbing uncontrollably, nitrous oxide will not be effective. Also, patients with claustrophobia or those with a history of abuse may find the nasal hood uncomfortable, resulting in increased agitation and anxiety.
The ideal pediatric patient for nitrous oxide is a slightly anxious child who is old enough to comfortably wear the nasal hood (which can be too large for children younger than 5), who can breathe freely through his or her nose, and who requires a mild level of anxiety relief to complete the desired treatment. Given proper patient selection, nitrous oxide can be a very effective and safe drug that reduces anxiety and increases a patient’s pain threshold.
ANALGESICS FOR CHILDREN
Discomfort following a dental visit is relatively common; as a result, parents are likely to administer analgesics to their child.22 A 2011 study found that one-third of pediatric dental patients reported pain following a restorative visit and most were given over-the-counter pain medications by their parent/caregiver.23
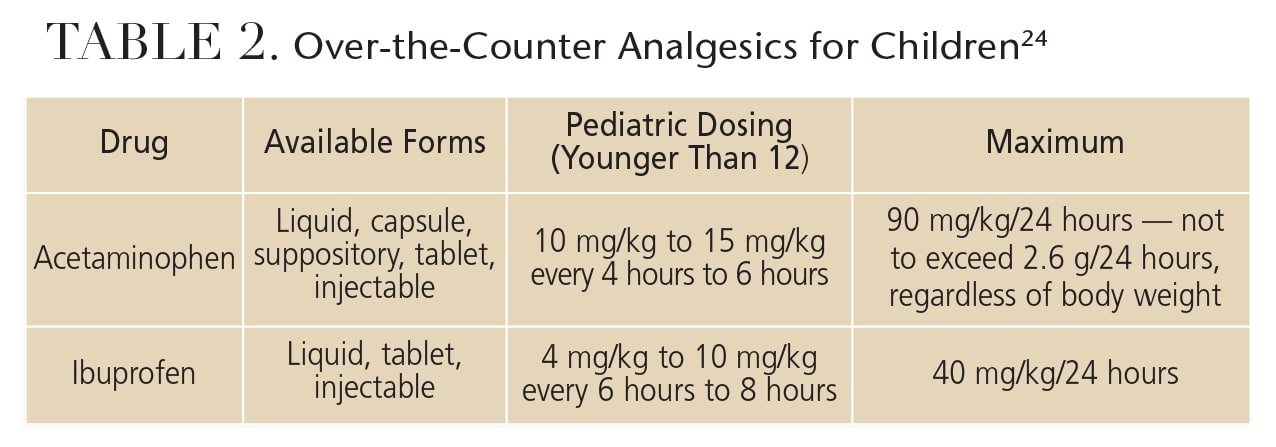
The drugs prescribed for pain control in children are generally limited to acetaminophen and ibuprofen (Table 2).24 Acetaminophen is classified as an analgesic and an antipyretic (fever-reducing) agent. It is effective for mild pain. Ibuprofen is an analgesic and antipyretic, as well, but it is also an effective anti-inflammatory agent in higher doses. This drug is proven to be effective at relieving moderate to severe pain. Generally, for most routine restorative and hygienic dental procedures (e.g., restorations, simple extractions and deep scaling), alternating between ibuprofen and acetaminophen is sufficient to control pain. It is important to be aware of the daily maximum dosages for these agents — 90 mg/kg for acetaminophen and 40 mg/kg for ibuprofen — because these maximums can be easily exceeded in small children.
Aspirin is not recommended for use in children because of its association with viral illnesses and the development of Reye syndrome. Narcotics, such as acetaminophen with codeine, are of limited value for pediatric patients and should be reserved for the rare circumstances where a child is in moderate to severe pain, such as following an extensive surgical procedure. If acetaminophen with codeine is utilized, the maximum amount of acetaminophen should never be exceeded due to the risk of liver damage. Codeine is also a respiratory depressant and its value for children should be carefully weighed against its risks.
SUMMARY
Pediatric pain control is a broad term that encompasses many aspects of patient care. Pretreatment pain mitigation begins at home and continues once the patient enters the dental office because anxiety and fear can affect pain perception during treatment. Ensuring a welcoming, bright, and positive experience in the dental office goes a long way toward producing positive outcomes and preventing pain.
Once a child is in the dental chair, the use of simple behavior management techniques, such as “Tell-Show-Do” and positive word choice, will help put the patient at ease. During treatment, establishing high-quality local anesthesia without causing pain is often the biggest hurdle to ensuring a child’s visit is positive. This can only be accomplished with a team approach, using the proper techniques, and ensuring patient safety.
Pain prevention and management — two skills that all oral health professionals must master — will make a major difference in the ability to treat children and may well lead some pediatric dental patients to pleasurably anticipate subsequent visits to the dental office.
References
- Schechter NL. The under-treatment of pain in children: An overview. Pediatr Clin North Am. 1989;36:781–794.
- Walker SM. Biological and neurodevelopmental implications of neonatal pain. Clin Perinatol. 2013;40:471–491.
- Barretto EPR, Ferreira EF, Pordeus IA. Evaluation of toothache severity in children using a visual analog scale of faces. Pediatr Dent. 2004:26:485–491.
- Peters ML. Emotional and cognitive influences on pain experience. Mod Trends Pharmacopsychiatri. 2015;30:138–152.
- Kim JS, Boynton JR, Inglehart MR. Parents’ presence in the operatory during their child’s dental visit: a person-environmental fit analysis of parents’ responses. Pediatr Dent. 2012;34:407–413.
- Karibe H, Aoyagi-Naka K, Koda A. Maternal anxiety and child fear during dental procedures: a preliminary study. J Dent Child. 2014;81:72–77.
- Law CS, Blain S. Approaching the pediatric dental patient: A review of nonpharmacologic behavior management strategies. J Calif Dent Assoc. 2003;31:703–713.
- Doerr PA, Lang WP, Nyquist LV, Ronis DL. Factors associated with dental anxiety. J Am Dent Assoc. 1998;129:1111–1119.
- Malamed SF. Handbook of Local Anesthesia. 6th ed. St. Louis: Mosby; 2013.
- Leal AM, Serra KG, Queiroz RC, Araújo MA, Maia Filho EM. Fear and/or anxiety of children and parents associated with the dental environment. Eur J Paediatr Dent. 2013;14:269–272.
- Milgrom P, Coldwell SE, Getz T, Weinstein P, Ramsay DS. Four dimensions of fear of dental injections. J Am Dent Assoc. 1997;128:756–762.
- Benham NR. The cephalometric position of the mandibular foramen with age. J Dent Child. 1976;43:233.
- Cheatham BD, Primosch RE, Courts FJ. A survey of local anesthetic usage in pediatric patients by Florida dentists. J Dent Child. 1992;59:401–407.
- Malamed SF. Pharmacology of vasoconstrictors. In: Handbook of Local Anesthesia. 6th ed. St. Louis: Mosby; 2013:124–156.
- Garisto GA, Gaffen AS, Lawrence HP, Tenenbaum HC, Haas DA. Occurrence of paresthesia after dental local anesthetic administration in the United States. J Am Dent Assoc. 2010;141:836–844.
- Sharaf AA. Evaluation of mandibular infiltration versus block anesthesia in pediatric dentistry. ASDC J Dent Child. 1997;64:276–281.
- Kanaa MD, Whitworth JM, Corbett IP, et al. Articaine buccal infiltration enhances the effectiveness of lidocaine inferior alveolar nerve block. Int Endod J. 2009;42:238–246.
- Chi D, Kanellis M, Himadi E, Asselin M. Lip biting in a pediatric dental patient after dental local anesthesia: a case report. J Ped Nurs. 2008:23:490–493.
- Prados-Frutos JC, Rojo R, González-Serrano J, et al. Phentolamine mesylate to reverse oral soft-tissue local anesthesia: A systematic review and meta-analysis. J Am Dent Assoc. 2015;146:751–759.
- Kupietzky A, Tal E, Shapira J, Ram D. Fasting state and episodes of vomiting in children receiving nitrous oxide for dental treatment. Pediatr Dent. 2008;30:414–419.
- Levering NJ, Welie JVM. Current status of nitrous oxide as a behavior management practice routine in pediatric dentistry. J Dent Child. 2011;78:24–30.
- Vernacchio L, Kelly JP, Kaufman DW, Mitchell AA. Medication use among children younger than 12 years of age in the United States: Results from the Slone Survey. Pediatrics. 2009;124:446–454.
- Staman NM, Townsend JA, Hagan JL. Discomfort following dental procedures for children. Pediatr Dent. 2013;35:52–54.
- Jaske AH. Mosby’s Dental Drug Reference. 11th ed. St. Louis: Elsevier/Mosby; 2014.




