A Review of How Photofunctionalization Restores Titanium Implants
This technique may be able to reverse titanium implant degradation, promote osseointegration, and shorten healing times
Over the years, researchers have attempted to improve the titanium implant surface through modification techniques such as sand blasting, laser etching, acid etching, and plasma spraying. The goals of these techniques are to increase the surface area and strengthen the bone implant interface. These modifications have improved the interface but had no impact on the osseointegration speed index (OSI), titanium degradation, or loss of bioactivity. A surface modification that could reverse degradation, restore bioactivity, strengthen the interface, and increase the osseointegration speed index could change implant dentistry. This paper looks at the surface modification technique, photofunctionalization with ultraviolet (UV) light, and how it improves osseointegration.
For a dental implant to achieve osseointegration, a labyrinth of physiologic processes must take place. The success of these processes leads to the completion of peri-implant bone remodeling. This remodeling should lead to 50% to 80% of the implant being in contact with bone, depending on the research.1–4 The percentage of bone-to-implant contact (BIC) in this range is believed to be necessary to achieve osseointegration and is used by researchers to determine osseointegration.
Over time, researchers have determined that numerous variables can impact the percentage of BIC and, consequently, the success or failure of osseointegration. These variables include: surgical technique, implant composition, bone quality and density, macroscopic design of the implant, microscopic design of the implant, an unloaded healing phase, medical and dental disease, certain prescription medications, and biomechanical considerations after loading.5–9
If these variables are managed properly when planning and executing the surgical and restorative phases of a dental implant, then osseointegration is expected in 3 to 6 months. Changes in the implant design, most notably the architecture of the implant surface (eg, roughening of the surface on a micron scale), have been made over the years to increase BIC, strengthen implant anchorage, and permit earlier loading of the implant.8–10
Even with these technological improvements, BIC does not approach 100%. This begs the question why this has not been accomplished thus far.
Once titanium is exposed to ambient air, a surface oxide layer is formed that is both protective and passive in nature and provides a noncytotoxic and corrosion resistance quality. With this oxide layer, titanium dioxide (TiO2), being crucial in osseointegration, researchers began to wonder, is it possible that the effects on this layer are responsible for BIC not approaching 100%?
Att et al11 were some of the first researchers to explore this possibility. They hypothesized that the bioactivity of titanium changes over time, and their initial research investigated the effects of age on titanium and how osteoblasts and extracellular proteins behave in response to this aging. They looked at whether the aging negatively impacts the TiO2 layer which, in turn, prevented BIC from approaching 100%. Four different ages of acid-etched titanium were included: new, 3-day-old, 2-week-old, and 4-week-old titanium surfaces.
This in vitro part of the study evaluated albumin adsorption and the number, size, and activity of osteoblasts attached to the surfaces between 2 and 24 hours of incubation time. Findings showed a significant difference between the new and aged titanium in all three categories (Figure 1A, B, and C).11 The new titanium showed better osteoblast migration onto the surface as the osteoblasts were larger in size and greater in number.
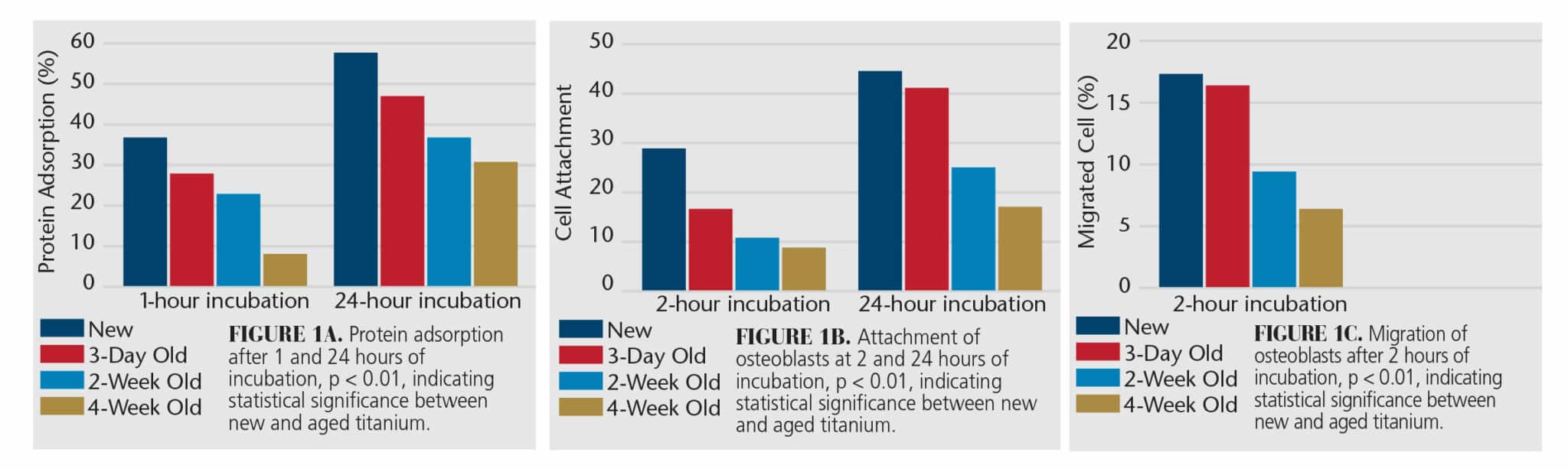 The amount of albumin adsorbed to the surface significantly decreased on the aged titanium. Mineralizing cultures showed alkaline phosphatase was diminished on the aged titanium,12 which is an important biomarker of mineralization around dental implants.13 Its diminished presence on aging titanium indicates a reduction in primary mineralization at the implant surface. The reduction in adhesion of extracellular proteins, specifically albumin and fibronectin, adversely affect cellular attachment and proliferation to the titanium surface, especially osteoblasts.14 This evidence strongly indicates that the attachment of these proteins is vital to the biocompatibility of the dental implant surface. These data demonstrate that age clearly influences titanium and its bioactivity. A new titanium surface is more bioactive than aged surfaces. The question then must be asked: What impact does age have on titanium to diminish its bioactivity?
The amount of albumin adsorbed to the surface significantly decreased on the aged titanium. Mineralizing cultures showed alkaline phosphatase was diminished on the aged titanium,12 which is an important biomarker of mineralization around dental implants.13 Its diminished presence on aging titanium indicates a reduction in primary mineralization at the implant surface. The reduction in adhesion of extracellular proteins, specifically albumin and fibronectin, adversely affect cellular attachment and proliferation to the titanium surface, especially osteoblasts.14 This evidence strongly indicates that the attachment of these proteins is vital to the biocompatibility of the dental implant surface. These data demonstrate that age clearly influences titanium and its bioactivity. A new titanium surface is more bioactive than aged surfaces. The question then must be asked: What impact does age have on titanium to diminish its bioactivity?
Impact of Age
The action of osseointegration depends heavily on the number and the constitution of cell attachment. The amount of osteogenic cells in contact with the implant surface affects the quantity of peri-implant bone and, consequently, the percentage of BIC. As titanium ages, it loses its ability to attract proteins and cells and therefore its ability to promote osseointegration. This biological aging seems to be unique to titanium and correlates to diminishing hydrophilicity.12
The loss of hydrophilicity and bioactivity is attributed to the formation of hydrocarbons on the TiO2 layer following manufacturing when the implant is introduced to ambient air. The hydrocarbons increase over time during transportation and eventual storage. Depending on the extent of carbon accumulated on the implant surface, cell proliferation, osteoblast attachment, and calcification may be adversely impacted. As a result, BIC is roughly 60% compared to a new clean titanium surface, which can achieve a BIC exceeding 90%.11,15 This would infer that hydrocarbons gathering on the titanium implant surface may be diminishing osseointegration.16
The difference in the strength of the interface between new titanium and aged titanium is significant. The bond between new titanium and bone is significantly stronger than that of aged titanium (Figure 2, page 13).11,15 If the hydrocarbons could be removed, then perhaps the hydrophilicity and bioactivity could be restored.
![]() Ultraviolet Light
Ultraviolet Light
In 1997, Wang et al17 discovered the effects of UV light on the superhydrophilicity of TiO2. The photochemical reaction gained considerable interest and is used in anti-fogging, stain resistance, pollutant degrading, and antibacterial treatments.15 Using UV light to modify a titanium surface to remove hydrocarbons, restore hydrophilicity, alter physiochemical properties, and improve bioactivity is referred to as photofunctionalization (PFZ).18,19
The surface charge of new titanium is positive, which is attractive to the biological components of osseointegration. The accumulation of hydrocarbons reverses the charge to negative, which significantly reduces its bioactivity.18 This leads to the negative effects on osseointegration and BIC.
The first clinical application of PFZ to modify dental implant surfaces prior to placement was studied by Funato and Ogawa20 in 2013. In this study, seven implants with micro-roughened surfaces were photofunctionalized via UV light exposure for 15 minutes chairside. They found that the implant stability quotient increased variably from 48 to 75 at insertion to 68 to 81 at loading. At the 1-year follow-up, all implants demonstrated successful osseointegration with early loading.
 Funato et al21 continued to look at the effects of PFZ through a retrospective study. They found that the implant healing time can be shortened using PFZ. The healing times were shortened from 6.6 months to 3.2 months compared to the control, which showed that PFZ will increase the OSI.22 While these results are encouraging, a 15-minute PFZ time is not practical in everyday practice. Fortunately, research has been ongoing to shorten this time.
Funato et al21 continued to look at the effects of PFZ through a retrospective study. They found that the implant healing time can be shortened using PFZ. The healing times were shortened from 6.6 months to 3.2 months compared to the control, which showed that PFZ will increase the OSI.22 While these results are encouraging, a 15-minute PFZ time is not practical in everyday practice. Fortunately, research has been ongoing to shorten this time.
A chairside UV activator has been introduced that photofunctionalizes dental implants with a 172 nm xenon excimer-generated UV vacuum light and stores and irradiates them in quartz ampules. Suzumura et al23 compared this device to other commercially available products and found that the UV vacuum light device achieved > 90% organic decomposition (removal of hydrocarbons) in 1 minute. Earlier devices would take anywhere from 15 minutes (clinically) to 48 hours (laboratory).19,23
Conclusion
PFZ makes it possible to reverse titanium degradation, eliminate organic matter, restore hydrophilicity, revert the surface charge to a positive state, and regain bioactivity in the surface TiO2. The proteins (albumin and fibronectin) are attracted to the surface and, in turn, will promote the migration, proliferation, and attachment of osteoblasts to the implant. All of this leads to greater biomechanical strength at the bone implant interface (Figure 3) with BIC of > 90% for cortical and marginal bone (Figure 4).2
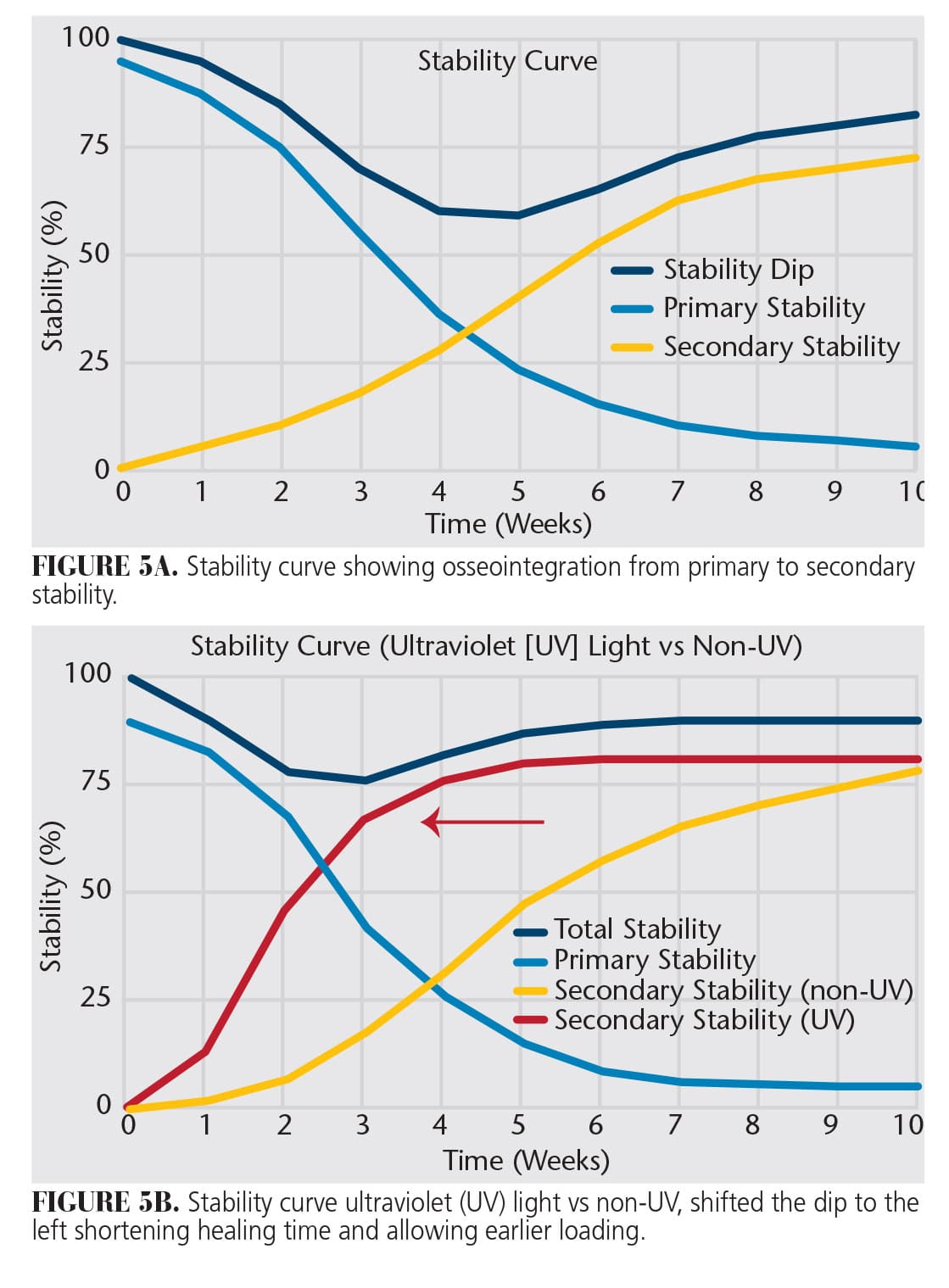
Perhaps what is most important to patients is the increase in the OSI as determined by sequential monthly implant stability quotient values.28 This means that PFZ has shifted the stability curve to the left, allowing earlier loading of dental implants. (Figure 5 A and B). UV vacuum lights make it possible to PFZ dental implants chairside, improve the biomechanical bond between the bone and implant, and shorten treatment time with predictable results.
Key Takeaways
- Various techniques have improved implant surfaces, but failed to enhance osseointegration and reverse titanium degradation.
- Aging titanium implants lose bioactivity and hydrophilicity due to the accumulation of hydrocarbons on the titanium dioxide layer, affecting osseointegration.
- Photofunctionalization restores hydrophilicity, reverses surface charge to positive, and removes hydrocarbons.
- Studies show that photofunctionalization can shorten healing times, increase the osseointegration speed index, and improve the biomechanical bond between bone and implant.
References
- Lian Z, Guan H, Ivanovski S, Loo YC, Johnson NW, Zhang H. Effect of bone to implant contact percentage on bone remodeling surrounding a dental implant. Int J Oral Maxillofac Surg. 2010;39:690–698.
- Pyo SW, Park YB, Moon HS, Lee, JH, Ogawa T. Photofunctionalization enhances bone-implant contact, dynamics of interfacial osteogenesis, marginal bone seal, and removal torque value of implants: a dog jawbone study. Implant Dent. 2013;22:666–675.
- Xing XJ, Liu BL, Liu L. The influence of osseointegration percentage on implant-bone interfacial stresses. J Xi’an Jiaotong Univ (Med Sci). 2002;23:395–397.
- Berglundh T, Abrahamsson I, Albouy JP, Lindh J. Bone healing at implants with a fluoride-modified surface: an experimental study in dogs. Clin Oral Implants Res. 2007;18:147–152.
- Park NI, Kerr M. Terminology in implant dentistry. In: Resnick R, ed. Misch’s Contemporary Implant Dentistry. 4th ed. New York: Elsevier; 2021:20-22.
- Parithimarkalaignan S, Padmanabhan TV. Osseointegration: an update. J Indian Prosthodont Soc. 2013;13:2–6.
- Albrektsson T, Brånemark PI, Hansson HA, Lindström J. Osseointegrated titanium implants. Requirements for ensuring a long-lasting, direct bone-to-implant anchorage in man. Acta Orthop Scand. 1981;52:155-170.
- Carr AB, Beals DW, Larsen PE. Reverse-torque failure of screw-shaped implants in baboons after 6 months of healing. Int J Oral Maxillofac Implants. 1997;12:598-620.
- Elias CN, Meirelles L. Improving the osseointegration of dental implants. Expert Rev Med Devices. 2010;7:241–256.
- Albrektsson T, Wennerberg A. On osseointegration in relation to implant surfaces. Clin Implant Dent Relat Res. 2019;21:4–7.
- Att W, Hori N, Takeuchi M, et al. Time-dependent degradation of titanium osteoconductivity: an implication of biological aging of implant materials. Biomaterials. 2009;30:5352–5353.
- Hori N, Att W, Ueno T, Sato N, Yamada M, Saruwatari L, et al. Age-dependent degradation of the protein adsorption capacity of titanium. J Dent Res. 2009;88:663–667.
- Sela, J, Gross UM, Kohavi D, et al. Primary mineralization at the surfaces of implants. Crit Rev Oral Biol Med. 2000:11:423-436.
- Lee J, Ogawa T. The biological aging of titanium implants. Implant Dent. 2012;21:415-420.
- Aita H, Hori N, Takeuchi M, et al. The effect of ultraviolet functionalization of titanium on integration with bone. Biomaterials. 2009;30:1015-1025.
- Hirota M, Ozawa T, Iwai T, Mitsudo K. UV-mediated photofunctionalization of dental implant: a seven-year results of a prospective study. J Clin Med. 2020;9:2733.
- Wang R, Hashimoto K, Fujishima A. Light-induced amphiphilic surfaces. Nature. 1997;388:431-432.
- Sugita Y, Saruta J, Taniyama T, et al. UV-pre-treated and protein adsorbed titanium implants exhibit enhanced osteoconductivity. Int J Sci. 2020;21:4194.
- Ogawa T. Ultraviolet photofunctionalization of titanium implants. Int J Oral Maxillfac Implants. 2014;29:95-102.
- Funato A, Ogawa T. Photofunctionalized dental implants: a case series in compromised bone. Int J Oral Maxillofac. 2013;28:1589-1601.
- Funato A, Yamada M, Ogawa T. Success rate, healing time, and implant stability of photofunctionalized dental implants. Int J Oral Maxillofac Implants. 2013;28:1261-1271.
- Suzuki S, Kobayashi H, Ogawa T. Implant stability change and osseointegration speed of immediately loaded photofunctionalized implants. Implant Dent. 2013;22:481-490.
- Suzumura T, Matsuura T, Komatsu K, Ogawa T. A novel high-energy vacuum ultraviolet light photofunctionalization approach for decomposing organic molecules around titanium. Int J Mol Sci. 2023;24:1978.
From Decisions of Dentistry. October 2023; 9(9):8,10,13


 Ultraviolet Light
Ultraviolet Light