 DRAZEN ZIGIC/ISTOCK/GETTY IMAGES PLUS
DRAZEN ZIGIC/ISTOCK/GETTY IMAGES PLUS
Oral Health Management for Patients With Alzheimer’s Disease
Comprehensive, person-centered care for individuals with this neurodegenerative condition is key to maintaining quality of life.
This course was published in the February 2022 issue and expires February 2025. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Define Alzheimer’s disease (AD).
- Explain the similarities between the immune responses generated by AD and periodontitis.
- Identify oral health risk factors associated with AD.
- Discuss the role of oral health professionals in treating patients with AD.
Alzheimer’s disease (AD) is a progressive neuroinflammatory condition characterized by memory and communication loss, as well as cognitive decline. The most common form of dementia, AD affects more than 6 million Americans over the age of 65, and is the sixth leading cause of death nationwide.1 Dementia is an umbrella term used to describe a group of symptoms that affect intellectual and social abilities severely enough to interfere with daily function.2 There is currently no known cause or cure of AD; however, common risk factors include age, gender, genetics, brain injury, obesity, diabetes, and environmental factors, such as air pollution or geographic location.3
This condition encompasses several stages, including preclinical, mild cognitive impairment, and severe dementia. During the preclinical phase, patients do not show signs of AD, but neuropathologic changes and inflammatory biomarkers are still present. Biological changes of AD can begin years, possibly even decades, before the patient experiences clinical symptoms. Warning signs of mild cognitive impairment include difficulty finding words, diminished reasoning, visual or spatial awareness impediments, and memory loss. As the illness progresses, memory loss becomes more apparent. Some patients experience mood or behavioral changes, as well as difficulty with language, problem solving, and recognizing family members and friends. Signs of late-stage AD include loss of bowel and bladder control, weight loss, inability to communicate or swallow, seizures, and increased sleeping.4
ETIOLOGY OF NEUROINFLAMMATION
Though the initial cause is unknown, AD’s neuropathological hallmarks include the accumulation of ß-amyloid plaques and the formation of neurofibrillary tangles containing tau protein in brain tissue.1–5 The presence of ß-amyloid plaques and tau protein tangles activates a host immune response, resulting in the release of pro-inflammatory cells, which contribute to chronic neuroinflammation and, ultimately, destruction of brain tissue.5 The host immune response observed in AD is similar to that of periodontal disease. During periodontal disease, Gram-negative bacteria invade subgingival tissues in the periodontium, causing the release of pro-inflammatory cytokines and chemokines, resulting in the subsequent destruction of alveolar bone and tooth-supporting tissues.6,7 Periodontal therapy reduces levels of Gram-negative bacteria, inflammation, and disease destruction.8
Research suggests a bidirectional relationship between AD and periodontal disease.9 Elevated levels of pro-inflammatory mediators, interleukins, tumor necrosis factor-alpha, C-reactive protein, and matrix metalloproteinases are present in both AD and periodontal disease.7,9,10 Risk factors, such as age, tobacco use, genetics, obesity and socioeconomic status, may influence the pathogenesis of AD and periodontal disease, as well.9 Studies have demonstrated a reduction of pro-inflammatory mediators systemically after scaling and root planing;8 however, nonsurgical periodontal therapies are not widely used to treat most chronic conditions affecting other body systems.
Studies also suggest Porphyromonas gingivalis may be a contributing factor in the development of AD, as it has been found in the brain tissue of deceased patients with AD.11 Mice studies have shown that P. gingivalis can travel from the oral cavity, through the bloodstream, into the brain;12 however, this has yet to be demonstrated in human subjects. While the detrimental effects of periodontal disease on other major systemic diseases have been demonstrated, a causative relationship between periodontal diseases and AD has not been established.
MANAGEMENT STRATEGIES
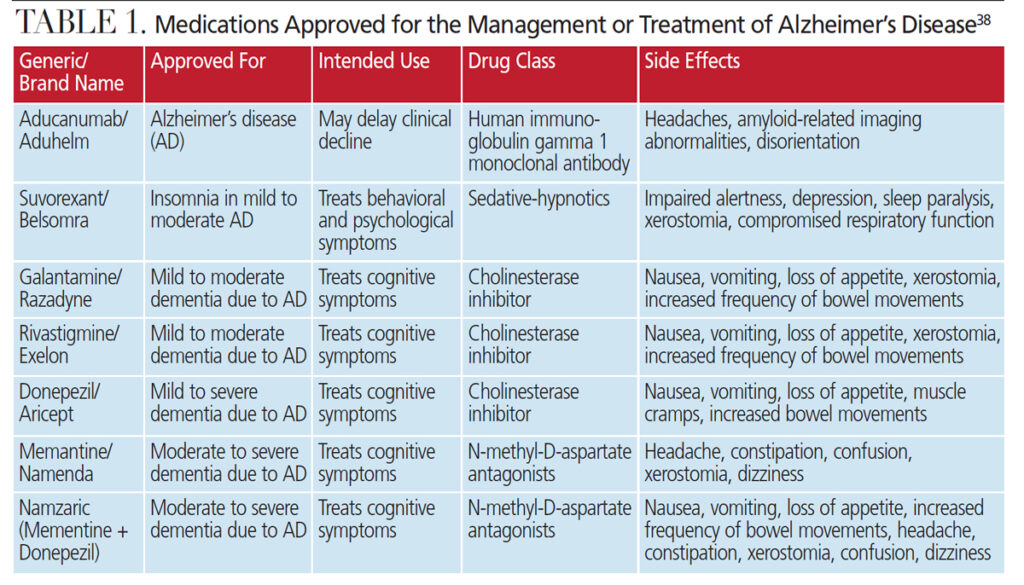
Therapies, including both pharmacological and nonpharmacological management, are available to reduce AD signs and symptoms, while improving overall quality of life. The U.S. Food and Drug Administration (FDA) has approved several cholinesterase inhibitors (Table 1) that may reduce or delay AD symptoms.13 The efficacy of these drugs is limited, however, and major side effects include nausea, fatigue, weight loss and hyposalivation.14 Aducanumab, a monoclonal antibody therapy for ß-amyloid, has received accelerated approval from the FDA for the treatment of AD.15 This is the first therapy that targets the fundamental pathophysiological changes seen in AD.16 The most common side effects are amyloid-related imaging abnormalities, headaches and disorientation.15,16 While aducanumab reduces the amount of ß-amyloid plaque in the brain,16 it has yet to show either reduced decline, stability or improvement of cognitive function following decreases in plaque.17
The emotional and behavioral changes linked with AD can be difficult to manage. Individuals with AD may experience depression, anxiety, sleep problems or irritability.4 These experiences may be triggered by environmental changes or when the person is prompted to perform routine hygiene tasks, such as toothbrushing or taking a bath.18 Managing triggers will help reduce the frequency and severity of these emotional and behavioral changes, while providing comfort and security for those with AD. In severe cases, medications — such as antidepressants, antianxiety or antipsychotic drugs — can be prescribed by a physician.19
Exercise is an effective nonpharmacological management strategy and improves cognitive function for all stages of AD.20 Physical activity also helps combat disease-modifying risk factors, such as depression, diabetes and obesity. A hallmark symptom of AD is the progressive shrinkage of the hippocampus, which is responsible for storing long-term memories, spatial processing and navigation.21 A meta-analysis by Wilckens et al21 found that mild-to-moderate exercise preserved hippocampal volume in at-risk older adults. Exercise has few side effects and better compliance when compared to medications. Thus, oral health professionals should consider discussing the benefits of physical activity on brain health during appointments with at-risk patients.
Diagnosis and therapeutic interventions in the preclinical stage of AD can preserve brain function and delay symptom onset.22 Early diagnosis can also help patients live independently and maintain a better quality of life. Patients may also preemptively make legal, financial and future care plans to avoid complications during the more severe stages of AD.22 Currently, diagnostic biomarkers for AD are tested via cerebral spinal fluid collection through painful lumber punctures; however, salivary diagnostics for ß-amyloid and tau protein are in development. Current research shows that salivary biomarkers can be used to diagnose AD,23 but further studies are necessary before this testing can be widely implemented.
ORAL HEALTH CONSIDERATIONS
Research suggests that severe periodontitis increases the risk of developing dementia by an odds ratio of 1.17, and patients with dementia are 69% more likely to be diagnosed with periodontal disease than those without.24 Additionally, patients diagnosed with AD are at increased risk of partial or complete edentulism compared to control groups.25 During early stages of AD, dental care should focus on disease prevention and oral health maintenance. Preventive oral healthcare is critical for the preservation of overall health. As AD progresses, the risk of oral disease increases because the ability to adequately and/or consistently perform oral hygiene declines. Common oral conditions associated with AD include dental caries, hyposalivation and periodontal disease.26
Due to the degenerative nature of AD, caries risk assessments should be performed regularly. Clinical assessments, along with current AD status and/or cognitive function, should be considered in the caries risk assessment process. Prescription and over-the-counter fluoride applications should be used for caries prevention. Fluoride, whether self-applied, professionally applied, or delivered through community water systems, reduces the incidence of caries by at least 25%.27 In advanced stages of AD, prescribed topical fluoride supplements, such as toothpastes or mouthrinses, should be stored in a secure location to avoid accidental ingestion.
Silver diamine fluoride (SDF) is ideal for this patient population, as it arrests active dentin caries and is an effective interim therapy.28 The application of this agent, however, is not a replacement for permanent restorative treatment. In addition to dentists, dental hygienists are permitted to apply SDF under the same authorization as other topical fluorides.29
Hyposalivation in patients with AD is often medication induced. To help address hyposalivation, clinicians should assess salivary gland function at each dental visit.30 Saliva substitutes, increased water consumption, and caries-reducing strategies should be considered.30 During advanced stages of AD, a humidifier in the patient’s home may help reduce the severity of xerostomia.
DENTAL PROFESSIONALS’ ROLE
Oral healthcare teams should be prepared to provide comprehensive, person-centered care for individuals with AD. Person-centered care requires a treatment plan for a person, not his or her disease. Collecting biographical information or interacting with family members can help personalize a care plan.31 Evaluating the patient’s cognitive function and physical ability can also facilitate an individualized therapeutic care plan. Clinicians can personalize preventive care appointments through collaborative efforts with other oral health professionals, medical providers and caregivers.
When providing person-centered care for patients with AD, dental teams should consider:32
- Working closely with the patient and/or caregiver to schedule appointments at a time that is most ideal for the patient
- Scheduling three-, four- or six-month preventive/periodontal recare intervals, depending on the patient’s oral health status
- Allowing for longer appointments
- Preventing falls by moving cords and adjusting the dental chair to an appropriate height
- Minimizing distractions and noise in the operatory. This may require seating the patient in a secluded room to avoid noise from surrounding treatment rooms
- Avoiding the use of dental terminology (when appropriate) to minimize confusion
- Using assistive devices, such as bite-wedges, metal finger guards, or a neck pillow and blanket to ensure comfort and safety
EMBRACE A COLLABORATIVE APPROACH
As a neurodegenerative condition, AD places significant burden on patients, caregivers, providers and healthcare delivery systems.33 As such, dental teams should collaborate with other dental and medical professionals in order to provide comprehensive, person-centered care and reduce the impact of disease.33 In the advanced stages of AD, the patient may require extensive treatment or accommodations. If the dental practice or clinician is unable to provide adequate and safe care, referrals to oral health professionals who have experience treating patients with AD should be considered.
Interprofessional collaboration improves person-centered care, reduces medical errors and healthcare costs, facilitates early diagnosis and intervention, and optimizes medication or treatment regimens.34 Unfortunately, communication between disciplines is one of the greatest barriers to providing comprehensive treatment.35 The dental practice should maintain a comprehensive list of all healthcare professionals and caregivers who care for the patient, as well as a complete list of medications and other treatment modalities. Additionally, many patients with AD reside in long-term care facilities and have limited access to care. Some states allow dental hygienists to practice in nontraditional settings, such as long-term care facilities or hospitals,36 which may improve care delivery to this patient population.
CAREGIVER SUPPORT AND ORAL HEALTH EDUCATION
Because caregivers play a vital role in maintaining the oral health of individuals with AD, effective communication between caregivers and the dental team is essential. Caregivers may require oral health training and education to properly care for these patients. Providing short and simple instructions on how to provide oral care is a good starting point.37 The mirroring technique is a strategy in which the caregiver demonstrates how to perform oral hygiene, while the patient mirrors those actions at the same time.37 This may be most effective when caring for patients who have lost the ability to follow step-by-step instructions. Encourage caregivers to experiment with different methods to find what works best for the patient. Training for effective handling and cleaning of removable dentures may be necessary in some cases.37 In the advanced stages of AD, the caregiver should continue to support and encourage good oral health to the best of his or her ability.
While the patient’s health is a caregiver’s chief concern, quality of life is equally important. Oral diseases can lead to other health disparities, including eating difficulties, digestive problems, malnutrition, and increased risk of infections.26 Loss of appetite in those with AD is a cardinal sign of oral discomfort.26 With the support and education provided by the dental team, caregivers and family members have the greatest opportunity to recognize oral diseases in their earliest stages.
CONCLUSION
As a progressive and debilitating neuroinflammatory condition with no known cause or cure, AD increases a patient’s risk for oral disease and poor outcomes. Dental teams should work closely with other healthcare professionals and caregivers to maintain patients’ oral health. Toward this goal, providing education to patients and caregivers on health promotion and disease prevention is an essential step in maintaining quality of life.
REFERENCES
- National Institute on Aging. Alzheimer’s Disease Fact Sheet. Available at: nia.nih.gov/health/alzheimers-disease-fact-sheet. Accessed January 5, 2022.
- Alzheimer’s Association. What Is Dementia? Available at: alz.org/alzheimers-dementia/what-is-dementia? Accessed January 5, 2022.
- Armstrong R. Risk factors for Alzheimer’s disease. Folia Neuropathol. 2019;57:87–105.
- National Institute on Aging. What Are the Signs of Alzheimer’s Disease? Available at: nia.nih.gov/health/what-are-signs-alzheimers-disease. Accessed January 5, 2022.
- Kametani F, Hasegawa M. Reconsideration of amyloid hypothesis and tau hypothesis in Alzheimer’s disease. Front Neurosci. 2018;12:25.
- Kinane DF. Causation and pathogenesis of periodontal disease. Periodontol 2000. 2001;25:8–20.
- Cekici A, Kantarci A, Hasturk H, et al. Inflammatory and immune pathways in the pathogenesis of periodontal disease. Periodontol 2000. 2014;64:57–80.
- Acharya AB, Thakur S, Muddapur MV. Effect of scaling and root planing on serum interleukin-10 levels and glycemic control in chronic periodontitis and type 2 diabetes mellitus. J Indian Soc Periodontol. 2015;19:188–193.
- Kamer AR, Craig RG, Niederman R, et al. Periodontal disease as a possible cause for Alzheimer’s disease. Periodontol 2000. 2020;83:242–271.
- Rempe RG, Hartz AMS, Bauer B. Matrix metalloproteinases in the brain and blood-brain barrier: versatile breakers and makers. J Cereb Blood Flow Metab. 2016;36:1481–1507.
- Dominy SS, Lynch C, Ermini F, et al. Porphyromonas gingivalis in Alzheimer’s disease brains: evidence for disease causation and treatment with small-molecule inhibitors. Sci Adv. 2019;5:1.
- Ishida N, Ishihara Y, Ishida K, et al. Periodontitis induced by bacterial infection exacerbates features of Alzheimer’s disease in transgenic mice. NPJ Aging Mech Dis. 2017;3:15.
- Sharma K. Cholinesterase inhibitors as Alzheimer’s therapeutics (Review). Mol Med Rep. 2019;20:1479–1487.
- Rewgenold W, Loreck D, Brandt N. Prescribing cholinesterase inhibitors for Alzheimer’s disease: timing matters. Am Fam Physician. 2018;97:700.
- U.S. Food and Drug Administration. Aducanumab (marketed as Aduhelm) Information. Available at: https://www.fda.gov/drugs/postmarket-drug-safety-information-patients-and-providers/aducanumab-marketed-aduhelm-information. Accessed January 5, 2022.
- Schneider L. A resurrection of aducanumab for Alzheimer’s disease. Lancet Neurol. 2020;19:111–112.
- Alzforum. Aducanumab Approved to Treat Alzheimer’s Disease. Available at: alzforum.org/news/research-news/aducanumab-approved-treat-alzheimers-disease. Accessed January 5, 2022.
- Gould E, Reed P. Alzheimer’s Association Quality Care Campaign and professional training initiatives: improving hands-on care for people with dementia in the USA. Int Psychogeriatr. 2009;21(Suppl 1):S25–S33.
- Calsolaro V, Antognoli R, Okoye C, et al. The use of antipsychotic drugs for treating behavioral symptoms in Alzheimer’s disease. Front Pharmacol. 2019;10:1465.
- Meng Q, Lin MS, Tzeng IS. Relationship between exercise and Alzheimer’s disease: a narrative literature review. Front Neurosci. 2020;14:131.
- Wilckens KA, Stillman CM, Waiwood AM, et al. Exercise interventions preserve hippocampal volume: a meta-analysis. Hippocampus. 2021;31:335–347.
- Chu LW. Alzheimer’s disease: early diagnosis and treatment. Hong Kong Med J. 2012;18:228–237.
- Pawlik P, Błochowiak K. The role of salivary biomarkers in the early diagnosis of Alzheimer’s disease and Parkinson’s disease. Diagnostics (Basel). 2021;11:371.
- Ju X, Kapellas K, Jamieson L, et al. The association between periodontal disease and dementia: a systematic review and meta-analysis. J Oral Biol Craniofac Res. 2019;2:2–11.
- Dioguardi M, Gioia GD, Caloro GA, et al. The association between tooth loss and Alzheimer’s disease: a systematic review with meta-analysis of case control studies. Dent J (Basel). 2019;7:49.
- Gao SS, Chu CH, Young FYF. Oral health and care for elderly people with Alzheimer’s disease. Int J Environ Res Public Health. 2020;17:5713.
- American Dental Association. Fluoridation Facts. Available at: https://www.ada.org/resources/community-initiatives/fluoride-in-water/fluoridation-facts. Accessed January 5, 2022.
- Subbiah GK, Gopinathan NM. Is silver diamine fluoride effective in preventing and arresting caries in elderly adults? A systematic review. J Int Soc Prev Community Dent. 2018;8:191–199.
- American Dental Hygienists’ Association. State Specific Information on Silver Diamine Fluoride. Available at: adha.org/resources-docs/Silver_Diamine_Fluoride_State_by_State_Information.pdf. Accessed January 5, 2022.
- Alabdullah J, Almuntashiri A. Strategies for safe and effective treatment of patients with Alzheimer disease. Dimensions of Dental Hygiene. 2020;18(7):32–35.
- Godfrey M, Young J, Shannon R, et al. Person-centred care: meaning and practice. In: Godfrey M, Young J, Shannon R, et al. The person, interactions and environment programme to improve care of people with dementia in hospital: a multisite study. NIHR Journals Library. 2018.
- Marchini L, Ettinger R, Caprio T, et al. Oral health care for patients with Alzheimer’s disease: an update. Spec Care Dentist. 2019;39:262–273.
- Galvin JE, Valois L, Zweig Y. Collaborative transdisciplinary team approach for dementia care. Neurodegener Dis Manag. 2014;4:455–469.
- Reeves S, Pelone F, Harrison R, et al. Interprofessional collaboration to improve professional practice and healthcare outcomes. Cochrane Database Syst Rev. 2017;6:CD000072.
- O’Daniel M, Rosenstein AH. Professional communication and team collaboration. In: Hughes RG. Patient Safety and Quality: an Evidence-Based Handbook for Nurses. Rockville, Md: Agency for Healthcare Research and Quality; 2008.
- Falcon CI, Coplen AE, Davis-Risen S, et al. Impact of an interprofessional education intervention and collaborative practice agreements of expanded practice dental hygienists in Oregon. J Dent Hyg. 2020;94:6–15.
- Manchery N, Subbiah GK, Nagappan N, et al. Are oral health education for carers effective in the oral hygiene management of elderly with dementia? A systematic review. Dent Res J (Isfahan). 2020;17:1–9.
- Alzheimer’s Association. FDA-Approved Treatment for Alzheimer’s. Available at: alz.org/media/documents/fda-approved-treatments-alzheimers-ts.pdf. Accessed January 5, 2022.
From Decisions in Dentistry. February 2022;8(2):40-43.