Oral Health Maintenance for Patients With Breast Cancer
Helping patients with breast cancer maintain optimal oral health is a key component in the overall continuum of care.
The authors have no commercial conflicts of interest to disclose.
This 2 credit hour self-study activity is electronically mediated.
OBJECTIVES
After reading this course, the participant should be able to:
- Identify the risk factors for breast cancer.
- Discuss the treatments for breast cancer and their effects on oral health.
- Explain the appropriate oral care regimen that should be provided prior
to initiating breast cancer treatment and during chemotherapy. - List the implications of antiestrogen therapy on oral health.
The American Cancer Society reports that, except for skin cancer, breast cancer is the most common cancer among American women.1 Most women diagnosed with breast cancer can expect an excellent outcome, with a five-year survival rate greater than 80%.2 Because cancer treatments can affect the oral tissues, dental care is an important component of breast cancer care and follow up. This review will cover how dental professionals can play a vital role in helping patients with breast cancer maintain their oral health, as well as prevent oral complications related to breast cancer therapies.
At present, there are more than 2.8 million breast cancers survivors in the United States, with nearly 232,000 new cases reported annually.1 The risk of developing breast cancer increases with age, with approximately two out of three cases of invasive breast cancer diagnosed in women age 55 or older.3 The average woman has a one in eight lifetime risk of developing breast cancer.1 The majority of cases will be diagnosed in post-menopausal women, and approximately 70% of these cancers express the estrogen receptor (ER) and/or the progesterone receptor (PR), and can be treated with antiestrogen therapy.4
Established risk factors for breast cancer include gender, increasing age, family history of breast cancer, early menarche, late menopause, long-term hormone replacement therapy, breast density, ethnicity and genetics.5 Research suggests that consuming one alcoholic drink a day increases the risk of breast cancer by 7% and consuming two or three drinks per day raises the risk by 20%.6
Breast cancer treatment is based on prognostic and predictive factors, including the histological grading (tumor severity); staging (how far cancer cells have spread); tumor cell responsiveness to various hormones or drugs; and patient’s age, overall health and preferences.7,8 Breast cancer surgery provides tissue for the purposes of grading, staging and assessing biomarkers. Depending on the cancer stage and histologic and molecular profile of the tumor, systemic adjuvant therapy may be indicated to decrease the risk of developing distant metastases.9,10 Systemic therapies may include chemotherapy, targeted therapies (such as trastuzumab), or antiestrogen therapy.7,11 These approaches may be considered either before or after surgery, based on the patient’s needs. Radiation therapy to the breast, chest wall and/or local lymph node regions may be administered, but it does not replace surgery, which is the foundation of early stage breast cancer management.
Approximately 75% of breast cancers express ER and/or PR.12,13 Breast cancer can depend on ER/PR signaling for tumor growth and survival.14 Targeting these receptors with antiestrogen therapies decreases the risk of breast cancer recurrence.10 In premenopausal women, surgery, radiation or chemical therapies with luteinizing hormone-releasing hormone inhibitors (e.g., goserelin, leuprolide or triptorelin) may ablate ovarian estrogen. The use of oral adjuvant systemic antiestrogens, such as tamoxifen online, is common. Post-menopausal women may be prescribed either tamoxifen or an aromatase inhibitor (e.g., anastrozole, exemestane or letrozole).15 Although male breast cancer accounts for approximately 1% of breast cancer diagnoses, nearly 90% of these tumors are ER positive. Treatment for male breast cancer patients is similar to female patients, and includes surgery followed by systemic therapy (chemotherapy and/or antiestrogen therapy). The need for radiation treatment is based on the tumor stage and biomarkers.16
EFFECTS OF TREAMENT
Lymphedema may result during or following breast cancer treatment. It manifests as a swelling that occurs when protein-rich lymph fluid accumulates in the interstitial tissue. Both the arm and the breast area may be affected. Although the etiology is unknown, the removal of lymph nodes and radiation of the breast may contribute to this condition. Patients with breast cancer should take preventive steps to reduce their risk of developing or exacerbating lymphedema, such as vigilant skin care, including avoiding needle punctures or blood draws, taking blood pressure readings on the unaffected arm, and using compression garments.17,18 Patients who have had a mastectomy and their lymph nodes removed should have their blood pressure monitored on the unaffected side. Patients who have undergone a bilateral axillary node dissection may need to have a blood pressure reading on their wrist or calf. Leg readings, however, can be lower than those taken on the arm due to peripheral vascular disease.
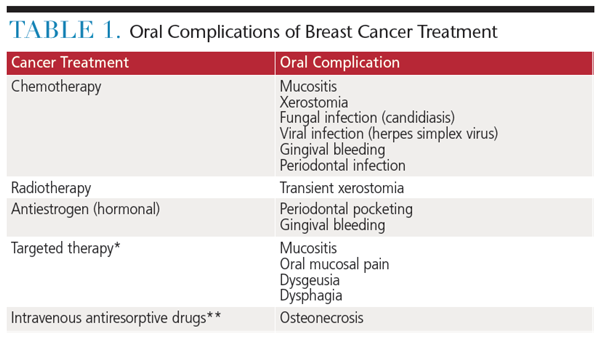
**Rare condition that generally has been related to dentoalveolar surgery
Oral complications resulting from breast cancer therapies may cause acute and late dental problems that often go unrecognized and untreated.19 Chronic complication include neurosensory changes; salivary, taste and functional changes; and oral maladies, such as caries and focal bony necroses of the jaw (Table 1). These complications may impact the patient’s quality of life.
PRETREATMENT DENTAL CARE
The National Institute of Dental and Craniofacial Research has developed guidelines for the provision of oral care to patients with cancer.20 These suggest that patients receive a dental visit prior to beginning cancer treatment. They also provide suggestions for care during chemotherapy and radiation20 — however, there is no universally accepted precancer therapy dental protocol at this time. Regardless, poor oral health is associated with increased incidence and severity of oral complications among patients with cancer.21 As such, the dental team’s involvement may reduce the risk of oral complications.
 The pretreatment dental examination enables clinicians to determine a patient’s oral health status and to decide whether care should be initiated. If possible, this visit should take place as far in advance as possible (at least one month) before the start of cancer treatment. Patients requiring oral surgery must have at least two weeks for healing before radiation therapy begins. Healing of oral surgical sites requires seven days to 10 days prior to beginning myelosuppressive chemotherapy. Medical consultation is always indicated before performing invasive procedures.22
The pretreatment dental examination enables clinicians to determine a patient’s oral health status and to decide whether care should be initiated. If possible, this visit should take place as far in advance as possible (at least one month) before the start of cancer treatment. Patients requiring oral surgery must have at least two weeks for healing before radiation therapy begins. Healing of oral surgical sites requires seven days to 10 days prior to beginning myelosuppressive chemotherapy. Medical consultation is always indicated before performing invasive procedures.22
The dental team should develop a comprehensive oral care plan that eliminates potential sites of infection that could produce complications during cancer treatment. Specific areas to address include necrotic teeth, mucosal lesions, dental caries, periodontal diseases, ill-fitting dentures or orthodontic appliances, temporomandibular dysfunction and xerostomia.
A comprehensive oral care plan must include oral hygiene strategies that may change throughout the stages of treatment. Patients should be counseled on tobacco cessation and the importance of limiting alcohol intake. Advise patients that some types of chemotherapy may cause temporary neuropathy with resulting numbness, tingling, pain, muscle weakness or even swelling. These symptoms may impact a patient’s ability to perform oral hygiene. Dental teams play an important role by educating patients about appropriate nutritional intake, effective oral hygiene practices, and early detection of oral lesions during the pretreatment visit (Table 2).20
CHEMOTHERAPY PHASE
Chemotherapy drugs are used to destroy cancer cells by stopping them from growing or multiplying. Chemotherapy can decrease platelet numbers, which can increase bleeding and reduce clotting time. Furthermore, chemotherapy can lower white blood cell counts and increase the risk of infection. Bloodwork should be conducted 24 hours before dental treatment to determine whether the patient’s platelet count, clotting factors and absolute neutrophil count are sufficient to prevent hemorrhage and infection. Treatment may need to be postponed if the platelet count is less than 50,000/mm3, abnormal clotting factors are present, and/or neutrophil count is less than 1000/mm3.
 Patients undergoing chemotherapy may also present with an indwelling venous access line for the administration of chemotherapy. Concerns regarding the need for antibiotic prophylaxis should be discussed with patients’ oncologists prior to treatment. Current guidelines do not recommend prophylactic antibiotic therapy for these patients because there is no evidence demonstrating that microorganisms associated with dental procedures cause infection of nonvalvular devices after implantation.23 That said, clinical judgment should be used if the patient is undergoing an invasive dental procedure where Staphylococcus microbes may be present.
Patients undergoing chemotherapy may also present with an indwelling venous access line for the administration of chemotherapy. Concerns regarding the need for antibiotic prophylaxis should be discussed with patients’ oncologists prior to treatment. Current guidelines do not recommend prophylactic antibiotic therapy for these patients because there is no evidence demonstrating that microorganisms associated with dental procedures cause infection of nonvalvular devices after implantation.23 That said, clinical judgment should be used if the patient is undergoing an invasive dental procedure where Staphylococcus microbes may be present.
Oral mucositis is the inflammation of oral mucosa resulting from chemotherapeutic agents, and typically manifests as erythema or ulcerations.24 Oral mucositis presents in approximately 40% of patients receiving chemotherapy, with nearly half requiring medical intervention, including modification of the cytotoxic cancer therapy. Oral mucositis usually begins seven days to 14 days after initiation of cytotoxic therapy, and remains present for approximately two weeks after the therapy is complete. It often presents on the soft palate, buccal mucosa, tongue, floor of the mouth and throat.25
Although it is difficult to predict which class of drugs may induce mucositis, some traditional chemotherapy drugs — such as fluorouracil, methotrexate and doxorubicin — are known to cause acute mucositis. Mucositis resulting from targeted therapy may present differently from traditional chemotherapy, with isolated ulcerations and mucosal pain (even in the absence of mucosal lesions).19
Topical cryotherapy may reduce mucositis caused by agents, such as 5-fluorouracil and high-dose melphalan, by decreasing vascular delivery of these toxic agents.26 While it is out of the scope of this review to describe the management of oral mucositis, Table 3 provides the basic approach for the management of chemotherapy-mediated oral mucositis.27
Viral, fungal and bacterial infections may also arise, with incidence depending on the use of prophylactic regimens, oral status prior to chemotherapy, and duration/severity of neutropenia. The frequency of infection decreases once the mucositis is resolved and the neutrophils have regenerated. Patients may remain at risk for infection, depending on the degree of immune recovery.28
ANTIESTROGEN THERAPY
Currently, there is scant evidence regarding the treatment of oral symptoms related to radiation treatment of the breast. While guidelines for dental care have been given for the pretreatment and chemotherapy phases of breast cancer treatment, little information is available about care for patients undergoing antiestrogen or hormonal therapies. The decrease in estrogen levels among post-menopausal women has been associated with reduced salivary flow unrelated to medications or periodontal disease and tooth loss.29,30
Recently, aromatase inhibitors, which severely diminish estrogen levels, have been shown to impact periodontal health and increase levels of xerostomia among patients with breast cancer.31,32 Decreased salivary flow can result in gingival bleeding and dental caries, and may be responsible for an increased prevalence of oral dysesthesia and alterations in taste sensation.29,30,33,34 Androgen deprivation therapy, used to treat prostate cancer, has been shown to negatively affect the periodontal health of some men.35 In addition, women using antiestrogen therapies may experience greater levels of depression, musculoskeletal symptoms and fatigue — any of which may impact their ability to achieve optimal oral hygiene. Oral health professionals should be aware of the potential changes in periodontal tissues, and the risk of xerostomia and psychosocial implications in patients undergoing antiestrogen therapies.
CONCLUSION
Oral complications of breast cancer treatments greatly affect quality of life, both during the treatment and surveillance periods. Consequently, professional care and optimal self-care — ideally, based on a pretreatment oral health assessment — can help ensure the best outcomes for patients fighting breast cancer.
References
- American Cancer Society. What Is Breast Cancer? What are the key statistics about breast cancer? Available at: http://www.cancer.org/cancer/breastcancer/detailedguide/ breast-cancer-key-statistics. Accessed December 10, 2015.
- Ries LAG, Young JL, Keel GE, Eisner MP, Lin YD, Horner MJ, eds. SEER Survival Monograph: Cancer Survival Among Adults: U.S. SEER Program, 1988–2001, Patient and Tumor Characteristics. National Cancer Institute, SEER Program, NIH Pub. No. 07-6215, Bethesda, Maryland: 2007.
- American Cancer Society. Causes, Risk Factors, and Prevention Topics. What are the risk factors for breast cancer? Available at: http://www.cancer.org/cancer/breastcancer/ detailedguide/breast-cancer-risk-factors. Accessed December 10, 2015.
- Anderson WF, Chatterjee N, Ershler WB, Brawley OW. Estrogen receptor breast cancer phenotypes in the Surveillance, Epidemiology, and End Results database. Breast Cancer Res Treat. 2002;76:27–36.
- McPherson K, Steel CM, Dixon JM. ABC of breast diseases. Breast cancer-epidemiology, risk factors, and genetics. BMJ. 2000;321:624–628.
- Hamajima N, Hirose K, Tajima K, et al. Alcohol, tobacco and breast cancer — collaborative reanalysis of individual data from 53 epidemiological studies, including 58,515 women with breast cancer and 95,067 women without the disease. Br J Cancer. 2002;87:1234–1245.
- Carlson RW, Allred DC, Anderson BO, et al. Invasive breast cancer. J Natl Compr Canc Netw. 2011;9:136–222.
- Carlson RW, Moench S, Hurria A, et al. NCCN Task Force Report: breast cancer in the older woman. J Natl Compr Canc Netw. 2008;6(Suppl 4):S1–S25.
- Bedard PL, Cardoso F. Can some patients avoid adjuvant chemotherapy for early stage breast cancer? Nat Rev Clin Oncol. 2011;8:272–279.
- EBCTCG. Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: an overview of the randomised trials. Lancet. 2005;365:1687–1717.
- Piccart-Gebhart MJ. Adjuvant trastuzumab therapy for HER2-overexpressing breast cancer: what we know and what we still need to learn. Eur J Cancer. 2006;42:1715–1719.
- Harvey JM, Clark GM, Osborne CK, Allred DC. Estrogen receptor status by immunohistochemistry is superior to the ligand-binding assay for predicting response to adjuvant endocrine therapy in breast cancer. J Clin Oncol. 1999;17:1474–1481.
- Rastelli F, Crispino S. Factors predictive of response to hormone therapy in breast cancer. Tumori. 2008;94:370–383.
- Miller WR, O’Neill J. The importance of local synthesis of estrogen within the breast. Steroids. 1987;50:537–548.
- Burstein HJ, Griggs JJ. Adjuvant hormonal therapy for early-stage breast cancer. Surg Oncol Clin N Am. 2010;19:639–647.
- Korde LA, Zujewski JA, Kamin L, et al. Multidisciplinary meeting on male breast cancer: summary and research recommendations. J Clin Oncol. 2010;28:2114–2122.
- Hayes S, Cornish B, Newman B. Comparison of methods to diagnose lymphoedema among breast cancer survivors: 6-month follow-up. Breast Cancer Res Treat. 2005;89:221–226.
- Harris SR, Hugi MR, Olivotto IA, Levine M. Clinical practice guidelines for the care and treatment of breast cancer: 11. Lymphedema. CMAJ. 2001;164:191–199.
- Epstein JB, Thariat J, Bensadoun RJ, et al. Oral complications of cancer and cancer therapy: from cancer treatment to survivorship. CA Cancer J Clin. 2012;62:400–422.
- National Institute of Dental and Craniofacial Research. Three Good Reasons to See a Dentist BEFORE Cancer Treatment. Available at: http://www.nidcr.nih.gov/oralhealth/ Topics/CancerTreatment/ThreeGoodReasons.htm. Accessed December 10, 2015.
- Sonis ST, Woods PD, White BA. Oral complications of cancer therapies. Pretreatment oral assessment. NCI Monogr. 1990;9:29–32.
- National Institute of Dental and Craniofacial Research. Oral Complications of Cancer Treatment: What the Dental Team Can Do. Available at: nidcr.nih.gov/oralhealth/Topics/CancerTreatment/OralComplicationsCancerOral.htm. Accessed December 10, 2015.
- Hong CH, Allred R, Napenas JJ, et al. Antibiotic prophylaxis for dental procedures to prevent indwelling venous catheter-related infections. Am J Med. 2010;123:1128–1133.
- Sonis ST, Fey EG. Oral complications of cancer therapy. Oncology (Williston Park). 2002;16:680–686.
- Sonis ST. Oral mucositis in cancer therapy. J Support Oncol. 2004;2(Suppl 3):3–8.
- Rocke LK, Loprinzi CL, Lee JK, et al. A randomized clinical trial of two different durations of oral cryotherapy for prevention of 5-fluorouracil-related stomatitis. Cancer. 1993;72:2234–2238.
- National Institute of Dental and Craniofacial Research. Chemotherapy and Your Mouth. Available at: nidcr.nih.gov/oralhealth/Topics/CancerTreatment/ChemotherapyYourMouth.htm. Accessed December 10, 2015.
- Hong CH, Napenas JJ, Hodgson BD, et al. A systematic review of dental disease in patients undergoing cancer therapy. Support Care Cancer. 2010;18:1007–1021.
- Streckfus CF, Baur U, Brown LJ, et al. Effects of estrogen status and aging on salivary flow rates in healthy Caucasian women. Gerontology. 1998;44:32–39.
- Wactawski-Wende J. Periodontal diseases and osteoporosis: association and mechanisms. Ann Periodontol. 2001;6:197–208.
- Taichman LS, Gomez G, Inglehart MR. Oral health-related complications of breast cancer treatment: assessing dental hygienists’ knowledge and professional practice. J Dent Hyg. 2014;88:100–113.32.
- Taichman LS, Inglehart MR, Giannobile WV, et al. Periodontal health in women with early stage postmenopausal breast cancer newly on aromatase inhibitors: a pilot study. J Periodontol. 2015;86:906–916.
- Lopez-Marcos JF, Garcia-Valle S, Garcia-Iglesias AA. Periodontal aspects in menopausal women undergoing hormone replacement therapy. Med Oral Patol Oral Cir Bucal. 2005;10:132–141.
- Mott AE, Grushka M, Sessle BJ. Diagnosis and management of taste disorders and burning mouth syndrome. Dent Clin North Am. 1993;37:33–71.
- Famili P, Cauley JA, Greenspan SL. The effect of androgen deprivation therapy on periodontal disease in men with prostate cancer. J Urol. 2007;177:921–924.




