Oral Care for Patients With Gastroparesis
Dental professionals should be prepared to help patients address the oral signs and symptoms of gastroparesis.
This course was published in the May 2021 issue and expires May 2024. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Explain gastroparesis (GP), its prevalence, and the signs and symptoms of this condition.
- Discuss oral conditions that may result from GP, and related oral health risk factors.
- Describe the management of GP symptoms.
Gastroparesis (GP) is a rare and debilitating gastrointestinal motility disorder that results in the complete or partial paralysis of stomach muscles. In health, the stomach chemically and mechanically digests food, then propels it into the small intestines (gastric emptying) where further digestion and nutrient absorption occur. In the case of GP, impaired muscles impact the stomach’s ability to mechanically digest and propel food into the small intestines, leading to malnourishment and recurrent symptoms of nausea, vomiting, severe upper abdominal pain, early satiety and bloating.1–4
The diagnosis of GP is determined by the presence of symptoms, lack of stomach obstruction, and delayed gastric emptying — however, the etiology of this disorder is multifaceted.1,2 The stomach muscle impairments associated with GP have been linked to the diagnoses of postoperative GP and several underlying health conditions, including Parkinson disease, neurological disorders, scleroderma, post-viral infections and, most frequently, diabetes. While many known factors contribute to the development of GP, more than half of all cases are idiopathic.1–7
Due to its subclinical nature, the prevalence of GP is unknown. However, recent data suggest that GP impacts approximately 0.16% of the United States population.8 This disorder is predominantly observed in women (66% to 80%), and is more prevalent among white people compared to other racial groups.3,8 Though GP can occur at any age, its etiology is often related to the age of onset:8
- Idiopathic (age < 39)
- Type 1 diabetes (between ages 40 and 59)
- Type 2 diabetes (between ages 60 and 80)
Studies have shown that in addition to high mortality rates, patients with GP experience a reduced quality of life compared to the general public. Upward of 10% of individuals with GP become disabled due to the disorder’s effects.3,9,10
Currently, there is no known cure for GP. Medical management of the disorder begins by identifying and treating underlying conditions. Other GP treatment strategies include managing symptoms and increasing gastric emptying.4,11 Symptoms and their severity may vary from day to day. This unpredictability makes it imperative for patients and healthcare providers to work closely while managing the disorder. Treatment strategies include, but are not limited to, dietary modifications (Table 1), fluid and electrolyte restoration, liquid nutrients (protein shakes and smoothies), administration of antiemetics, prokinetics (gastric stimulating drugs), gastric electrical stimulators, and feeding tubes.2,11–13
ORAL SIDE EFFECTS OF GASTROPARESIS
Oral and systemic health are intrinsically related. The oral cavity is often referred to as the mirror to the body because it can reveal signs of disease and nutritional deficiencies, as well as symptoms of poor diet and recreational drug use. The oral cavity is also the start of the gastrointestinal tract and is responsible for initiating the chemical digestion through salivary enzymes and mechanical digestion through mastication. As such, patients with GP need to ensure their tooth structures and salivary glands are intact and functioning properly to aid their already compromised digestive processes.
Oral health professionals are well positioned to recognize the oral signs of systemic diseases. Because GP is underdiagnosed, dental teams should not only be aware of the disorder’s signs and symptoms, but also its oral manifestations. Dentists and dental hygienists can assist undiagnosed patients in seeking appropriate medical care and collaborate interprofessionally as part of the patient’s healthcare team.
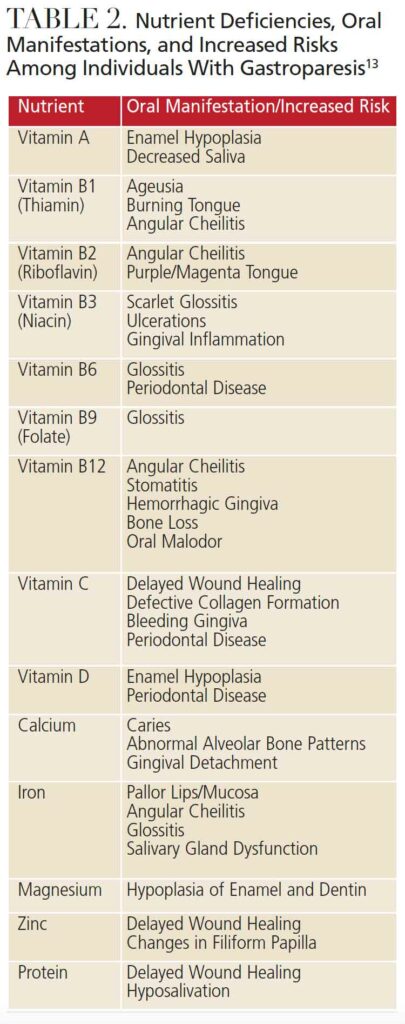
Due to the multifaceted nature of GP, patients are at high risk of oral side effects. Hyposalivation is observed in severe cases of malnutrition or can be caused by antiemetic drugs. Decreased salivary flow, along with frequent nausea and vomiting and strict dietary modifications, increase patients’ risk of developing erosive tooth wear (ETW), dentinal hypersensitivity and caries.14–16 Vomiting, dietary restrictions, early satiety, and/or slow gastric emptying rates contribute to malnutrition, which further impacts periodontal health and development of oral conditions related to vitamin and nutrient deficiencies, including, but not limited to, glossitis, angular cheilitis, pallor gingiva, stomatitis and oral malodor (Table 2).
![TABLE 2. Nutrient Deficiencies, Oral Manifestations, and Increased Risks Among Individuals With Gastroparesis13]() DENTAL CONSIDERATIONS
DENTAL CONSIDERATIONS
Because patients with GP are susceptible to ETW, dentinal hypersensitivity and caries, providers need to understand the similarities and differences of these calcified tooth structure disease processes. All three conditions result from an equilibrium imbalance of tooth mineralization processes and the dissolution of the organic matrix of tooth structures. Demineralization occurs when mineral ions (calcium and phosphate) are removed from hard tissue tooth structures. Conversely, remineralization is the process of moving minerals, including hydrogen fluoride, back to tooth surfaces. Saliva acts to facilitate the continuous process of demineralization and remineralization. It also protects tooth structure by buffering destructive acidic pH levels that break down and dissolve tooth surfaces.14,17–19 When patients experience hyposalivation, an imbalance occurs in the demineralization and remineralization processes, increasing the rate of demineralization, often leading to tooth erosion, hypersensitivity and/or caries.
Providers must further recognize how the etiologies of these oral conditions differ, as this understanding — together with a comprehensive assessment of patients’ oral health and GP status — will help in treating and preventing dental issues in patients with GP.
Of particular concern in this patient population, ETW is a result of nonbacterial acids (stomach acids) caused by pH fluctuations in the throat and oral cavity, as well as other factors — including nutrition and patient behaviors — that sensitize teeth to stimuli.18,19 Patients with GP who frequently vomit may present with smooth, saucer-like depressions on the lingual surfaces of their anterior teeth. In severe cases, ETW may extend into the dentin and/or compromise the integrity of dental restorations. Clinical signs of ETW must be diagnosed as early as possible so appropriate, individualized risk management and treatment can be swiftly implemented. Individuals who report vomiting should be instructed to avoid brushing immediately after vomiting and instead swish with water or sodium bicarbonate to help neutralize acids. Additionally, products containing fluoride, casein phosphopeptide-amorphous calcium phosphate (CPP-ACP), and xylitol should be recommended, as they effectively buffer salivary pH levels.14 Because patients with GP may be unaware of their actual acid intake, best practice dictates using a dietary and behavior diary over four days to help adjust eating and drinking habits that can prevent further destruction from ETW.15,20–22
Erosion is also a significant risk factor for dentinal hypersensitivity, as the destructive nature of ETW breaks down enamel, exposing the nerve fibers within dentinal tubules. Once the tubules are open, stimuli from food, beverages, toothbrushing and dental care can elicit a painful response that, in severe cases, can negatively impact quality of life.23 Determining which stimuli is causing the pain, as well as noting its frequency and severity, is key when caring for patients with sensitivity. Glass ionomer and resin-based composites can be placed to treat localized sensitivity. Varnishes, toothpastes and mouthrinses containing sodium fluoride, stannous fluoride or strontium salts can help prevent generalized symptoms by occluding dentinal tubules, thus protecting nerve endings from painful stimuli. In-office products containing calcium sodium phosphosilicate, arginine with calcium carbonate, glutaraldehyde hydroxyethyl methacrylate, or potassium binoxalate with nitric acid are also available. Products with oxalates act by decreasing dentinal permeability while occluding dentinal tubules, and potassium nitrate-based products decrease sensitivity by actively depolarizing nerve endings.24 Regardless of treatment modality, oral health professionals should work closely with the patient to reduce or eliminate the pain associated with sensitivity.
Poor control of oral biofilm25 and diminished salivary flow rates contribute to the development of caries lesions, as do other forces, such as malnutrition and/or systemic risk factors. Particularly for patients with GP, caries risk must be carefully assessed prior to developing a management strategy. One approach is caries management by risk assessment (CAMBRA), a criteria that rates caries risk as low, medium or high. The use of CAMBRA involves collecting information from the patient’s medical and dental history, conducting a clinical examination, caries preventive efforts, identifying lesions that may be reversed or treated, and developing a customized care plan.26 Products containing fluoride, CPP-ACP, and/or xylitol inhibit acidogenic bacterial growth, buffer salivary pH, and stimulate salivary flow rates. When present in saliva, fluoride and CPP-ACP aid in the remineralization process. Fluoride and ACP both act by creating a crystal matrix on tooth surfaces that attract calcium and phosphate ions. The use of CPP helps saturate saliva and plaque biofilm with calcium and phosphate, increasing tooth remineralization.14 As patients with GP often experience malnutrition, they are more likely to be deficient in calcium and phosphorous, making casein-based products important for the prevention and maintenance of caries.
CARING FOR THIS PATIENT POPULATION
While little is known about providing dental care specifically for patients with GP, dental teams can effectively address the disorder’s common oral side effects. Patient care should center on interprofessional collaboration, oral health risk assessment, and individualized treatment and management plans. Oral health professionals should be ready to collaborate with members of the patient’s medical team, as well as dietitians. Understanding the full extent of the patient’s diagnosis and/or underlying health condition, current GP symptoms and status, dietary modifications, and medications will help when developing dental treatment and maintenance plans.
Professional interventions should first focus on treating any underlying health conditions. For example, if a patient presents with type 2 diabetes-associated GP, dental diabetes standards of care would be followed first and documented in the patient’s chart. Any diabetes-associated nutritional counseling should uphold the treatment plan of the patient’s nutritionist and/or medical team. Treatment plans, which should be updated at each appointment, should offer preventive care and manage risk factors for oral disease.
For patients experiencing hyposalivation, salivary gland function should be assessed routinely at dental visits.15,27 Treatment of hyposalivation should include increased frequency of water consumption, saliva substitutes (such as gels, sprays or mouthrinses), and xylitol chewing gum. Research shows that saliva substitutes can increase remineralization due to the increased viscosity of the substitute product, which increases protection of the tooth surface.28
Patients at high risk for ETW, sensitivity, caries and/or hyposalivation should use extra-soft toothbrushes, fluoride- and/or casein-based products, saliva substitutes, and products containing xylitol. These individuals, including patients with GP, should avoid brushing their teeth immediately after vomiting and/or eating and drinking, and instead swish with water or hydrogen peroxide to neutralize oral pH levels. Concerns surrounding nutrient-related oral manifestations should be addressed collectively with patients and their medical teams.
SUMMARY
In conclusion, GP is a rare and often debilitating disorder with no known cure. Due to the complex nature of GP, patients are at high risk of experiencing adverse oral conditions, including, but not limited to, hyposalivation, ETW, dentinal hypersensitivity, caries, and oral manifestations specifically related to malnutrition.
Though evidence-based dental protocols are developing on this topic, oral health professionals should be prepared to critically assess and comprehensively manage the common oral side effects of GP. Treatment and maintenance planning should be conducted in collaboration with the patient and his or her extended healthcare team to help improve the patient’s oral health and overall quality of life.
REFERENCES
- American College of Gastroenterology. Gastroparesis. Available at: gi.org/topics/gastroparesis. Accessed March 25, 2021.
- Camilleri MD, Parkman HP, Mehnaz SA, et al. Clinical guideline: management of gastroparesis. Am J Gastroenterol. 2013;108:18–38.
- Lacy BE, Crowell MD, Mathis C, et al. Gastroparesis: quality of life and health care utilization. J Clin Gastroenterol. 2018;52:20–24.
- Mayo Clinic. Gastroparesis Symptoms and Causes. Available at: mayoclinic.org/diseases-conditions/gastroparesis/symptoms-causes/syc-20355787. Accessed March 25, 2021.
- Rey E, Choung RS, Schleck CD, et al. Prevalence of hidden gastroparesis in the community: the gastroparesis “iceberg.” J Neurogastroenterol Motil. 2012;18:34–42.
- Hasler ML. Gastroparesis: symptoms, evaluation, and treatment. Gastroenterol Clin N. 2007;36:619–647.
- Grover M, Farrugia G, Stanghellini V. Gastroparesis: a turning point in understanding and treatment. Gut. 2019;68:2238–2250.
- Syed AR, Wolfe MM, Calles-Escandon J. Epidemiology and diagnosis of gastroparesis in the United States: a population-based study. J Clin Gastroenterol. 2020;54:50–54.
- Hyett B, Martinez FJ, Gill BM, et al. Delayed radionucleotide gastric emptying studies predict morbidity in diabetics with symptoms of gastroparesis. Gastroenterol. 2009;137:445–452.
- National Organization for Rare Disorders. Gastroparesis. Available at: rarediseases.org/rare-diseases/gastroparesis. Accessed March 25, 2021.
- Guideline Central. Clinical Guideline: Management of Gastroparesis Clinical Practice Guidelines. Available at: guidelinecentral.com/summaries/clinical-guideline-management-of-gastroparesis/#section-society. Accessed March 25, 2021.
- Parkman HP, Hasler WL, Fisher RS. American Gastroenterological Association medical position statement: diagnosis and treatment of gastroparesis. Gastroenterol. 2004;127:1589–1591.
- U.S. Department of Agriculture. Methods and Application of Food Composition Laboratory. Available at: ars.usda.gov/nutrientdata. Accessed March 25, 2021.
- Farooq I, Moheet IA, Imran Z, Farooq U. A review of novel dental caries preventive material: casein phosphopeptide-amorphous calcium phosphate (CPP-ACP) complex. King Saud University Journal of Dental Sciences. 2013;4:47–51.
- Yoskikawa H, Furuta K, Ueno M, et al. Oral symptoms including dental erosion in gastroesophageal reflux disease are associated with decreased salivary flow volume and swallowing function. J Gastroenterol. 2012;47:412–420.
- Buzalaf M, Magalhaes A, Rios D. Prevention of erosive tooth wear: targeting nutritional and patient-related risks factors. Br Dent J. 2018;224:371–378.
- Pitts EI, Furgeson D. Saliva, remineralization and dental caries. Decisions in Dentistry. 2019;5(4):38–41.
- Frese C, Frese F, Kuhlman S, et al. Effect of endurance training on dental erosion, caries, and saliva. Scand J Med Sci Sports. 2015;25:319–326.
- Mulic A, Bjorg Tveit A, Songe D, Silvertsen H, Skaare A. Dental erosive wear and salivary flow rate in physically active young adults. BMC Oral Health. 2012;12:8.
- Lussi A, Carvalho T. Erosive tooth wear: a multifactorial condition of growing concern and increasing knowledge. Monogr Oral Sci. 2014;25:1–15.
- Lussi A, Hellwig E. Risk assessment and causal preventive measures. Monogr Oral Sci. 2014;25:220–229.
- Alaraudanjoki V, Laitala M, Tjaderhane L, et al. Influence of intrinsic factors on erosive tooth wear in a large-scale epidemiological study. Caries Res. 2016;50:508–516.
- O’Toole S, Bartlett D. The relationship between dentine hypersensitivity, dietary acid intake and erosive toothwear. J Dent. 2017;67:84–87.
- Miglani S, Aggarwal V, Ahuja B. Dentin hypersensitivity: recent trends in management. J Conserv Dent. 2010;13:218–224.
- Bowen W, Burne R, Wu H, Koo H. Oral biofilms: pathogens, matrix and polymicrobial interactions in microenvironments. Trends Microbiol. 2018;26:229–242.
- Featherstone J, Chaffee B. The evidence for caries management by risk assessment (CAMBRA). Adv Dent Res. 2018;29:9–14.
- Navazesh M, Kumar S, USC School of Dentistry. Measuring salivary flow: challenges and opportunities. J Am Dent Assoc. 2008;139:35S–40S.
- Aykut-Yetkiner A, Wiegand A, Attin T. The effect of saliva substitutes on enamel erosion in vitro. J Dent. 2014;42:720–725.
From Decisions in Dentistry. May 2021;7(5)40-43.