 VDCM IMAGE / ISTOCK / GETTY IMAGES PLUS
VDCM IMAGE / ISTOCK / GETTY IMAGES PLUS
Modern Perspectives on Endodontic Therapy
Craig A. Dunlap, DDS, from the University of the Pacific Arthur A. Dugoni School of Dentistry shares insights into the latest thinking in endodontic care.
Like every field in dentistry, endodontic therapy is subject to ongoing evolution, with new treatment modalities and therapeutic agents constantly entering the armamentarium. At times, these may replace more traditional treatment approaches; while, in other cases, they serve as clinical alternatives or adjuncts to care. To gain a better sense of the modern state of endodontics, we asked Craig A. Dunlap, DDS, an assistant professor and acting director of the graduate program in endodontology at the University of the Pacific Arthur A. Dugoni School of Dentistry in San Francisco, for his thoughts on today’s progressive endodontic practice.
The advent of new tools and technologies for endodontic therapy is leading to more predictable outcomes. Please briefly recap some of the more notable recent advances that support improved care.
Technological advancements in endodontic instruments and materials continue to evolve at a fast pace. Specifically, many of the newer nickel-titanium (NiTi) files are now made from heat-treated memory wire that makes them much more flexible than non-heat-treated instruments.1 Heat-treated memory wire is also more resistant to cyclic fatigue, and this makes them less prone to breaking.2 There have been advances in the physical design of rotary NiTi instruments as well. One such advancement is creating an instrument with a larger envelope of motion that has the ability to touch more of the canal walls when compared to previous rotary NiTi systems.3
In an attempt to conserve dentin and prevent root fractures, there is a trend toward more conservative endodontic access and canal preparations. However, it is difficult to adequately irrigate and disinfect these smaller preparations. In response to this challenge, irrigant activation has been advocated as an adjunct to allow the irrigant to reach the apical third of the canal system.4
There are many techniques to activate the irrigant, including sonic activation and ultrasonics. Among these two approaches, ultrasonics has been found to be more efficient than sonic activation5 — although both have the ability to improve the cleanliness of the canal walls and isthmuses.4 More recently, multisonic activation has been introduced. Preliminary research suggests that canals which have been treated with this technology are cleaner and have less debris, with root canal healing rates reported at 94% to 98% at 12 months.6,7
Photon-initiated photoacoustic streaming (PIPS) uses lasers to activate the irrigant. Research has shown that PIPS is able to reduce bacterial counts and eliminate biofilm better than ultrasonic activation.8 In addition, a novel laser irrigating system has been introduced that is so new that published data is not yet available. Taken as a whole, a recent systematic review showed that while irrigant activation removes more debris and cleans canals better, research to date has not demonstrated improved clinical outcomes.9
Endodontic biomaterials are another category with many recent advances. Specifically, innovations in bioceramic sealers allow clinicians to obturate canals with less tapered canal preparations. This is because gutta-percha condensation is not required with bioceramic sealers. Bioceramic putties and root repair materials are also readily available. These materials are biocompatible and seal extremely well. In turn, this has led to better outcomes, particularly in the area of endodontic microsurgery.10 Current investigations are underway with gutta-percha coated with silver or diamond nanoparticles, as these have shown promising results in reducing bacterial counts and the potential of bacterial reinfection.11
While important to endodontic therapy, conventional radiographs are only one facet of the diagnostic and treatment processes. What are the value and limitations of two-dimensional radiographs in endodontics?
Radiographs should be prescribed according to the as low as reasonably achievable (ALARA) concept. Accordingly, the joint position statement by the American Association of Endodontists (AAE) and American Association of Oral and Maxillofacial Radiologists states that intraoral radiographs, such as periapical and bitewing radiographs, should be considered the imaging modality of choice for endodontic evaluation.12 Periapical radiographs are still useful in endodontics for examining the entire tooth in question, including the periapical tissues. Bitewing radiographs are excellent for evaluating caries, existing restorations, previously initiated endodontic therapy, and periodontal attachment level. Additionally, bitewing radiographs are useful when determining the location and size of the pulp chamber, which can aid clinicians during endodontic access.13
THERE IS A TREND TOWARD MORE CONSERVATIVE ENDODONTIC ACCESS AND CANAL PREPARATIONS
Intraoral radiographs offer the added advantage of being readily available, easy to acquire, and inexpensive. Limitations of intraoral radiographs arise from the superimposition of surrounding structures and the inability to visualize the area of interest in all planes. Additionally, two-dimensional images do not accurately show the proximity of the roots to vital structures, such as the inferior alveolar nerve or maxillary sinus.
What role does three-dimensional imaging play in successful endodontic care? To what extent is the use of cone beam computed tomography (CBCT) case-dependent?
Consistent with the ALARA principle, intraoral radiographs should still be considered the imaging modality of choice for routine endodontic care because CBCT scans require approximately three times the amount of radiation than intraoral radiographs.14 That said, CBCT images are much more sensitive than intraoral radiographs at detecting periapical pathology and assessing root canal anatomy, including second mesiobuccal canals on maxillary molars. A CBCT volume is also recommended in trauma cases to evaluate whether there is an alveolar fracture. In addition, CBCT scans are essential in evaluating cases of internal and external resorption because they can accurately show the exact location and extent of the lesion.15 Because there is no distortion on CBCT images, accurate measurements are possible. In combination with its ability to accurately determine the location of the roots relative to vital anatomical structures (such as the inferior alveolar nerve, mental foramen and maxillary sinus), this makes CBCT the imaging modality of choice during presurgical planning.16
What’s more, the information from a CBCT scan has been found to influence the endodontic treatment plan. In a classic study by Ee et al,17 30 cases were diagnosed with periapical images by three board-certified endodontists. Three weeks later, the same three endodontists evaluated the same cases with CBCT images. When the CBCT images were available, there was a 63% difference in the recommended treatment plan.
There are some disadvantages to CBCT use, of course, including increased radiation and cost. Additionally, cone beam scans create image artifacts, such as beam hardening and scatter, when radiopaque objects are present, including root canal filling material, metal posts and restorations. When the radiation beam hits a radiopaque object, the lower energy photons are absorbed by the object and the higher energy photons can pass through, thereby “hardening” the beam. Beam hardening creates radiolucent artifacts which may be mistaken for untreated canals or fractures.18
PAIN FOLLOWING ENDODONTIC TREATMENT IS BEST MANAGED WITH A COMBINATION OF IBUPROFEN AND ACETAMINOPHEN
At present, CBCT is not yet recommended for the diagnosis of vertical root fractures. A recent systematic review and meta-analysis revealed that CBCT is only 78% accurate in diagnosing vertical root fractures.19 However, there are some common signs of vertical root fractures that can be seen on CBCT images. These may include: 1) A space between the buccal and/or lingual cortical plate; 2) a radiolucency at the terminus of a post; 3) missing the entire buccal cortical plate; 4) loss of bone midroot with intact bone coronal and apical to the defect; and 5) direct visualization of the fracture line.20
Please describe modern approaches to working length (WL). What role do electronic apex locators play in this phase of treatment?
Determining the correct apical limit of the canal system is critical during root canal treatment. An accurate WL allows for thorough mechanical and chemical disinfection of the canal system.21 The correct WL also protects the periodontal tissues from instrumentation beyond the canal terminus and helps prevent the extrusion of debris.22 A systematic review found that success rates are lower when root canal obturation extends beyond the radiographic apex, as well as when the root canal obturation terminates short of the radiographic apex by more than 2 mm.23
Historically, a periapical radiograph has been the primary method for determining WL,24 yet periapical radiographs pose several disadvantages. For example, they do not accurately show the location of the apical foramen.25 They are also susceptible to subjective interpretation.26 Other methods of WL determination include tactile feel and the paper point technique.27,28 The latter involves inserting a paper point at increasing lengths until a drop of tissue fluid appears on the tip. While there is research supporting the validity of the paper point technique, tactile feel has been shown to be unreliable.29
In 1942, Suzuki30 discovered that electrical resistance was a constant between the periodontal ligament and oral mucosa at 6.5 kiloohms. This finding was used to construct the first electronic apex locator. However, this primitive apex locator used direct current, which did not support readings in the presence of fluid. Today’s apex locators use alternating current and calculate the ratio of impedances at multiple frequencies, as this allows for accurate measurements in the presence of moisture, as well as pulp tissue.31 Modern electronic apex locators have been studied extensively and found to be accurate between 82% and 100% of the time.32
Research has shown that electronic apex locator readings are more accurate after coronal flaring has been performed.33 Additionally, the process of coronal flaring straightens the canal slightly which, in turn, shortens the WL. Therefore, clinicians are advised to perform coronal flaring prior to using an electronic apex locator. Once the electronic lengths are determined, a wire length radiograph should be taken. The wire length radiograph supplies invaluable information, such as whether canals join or canal curvatures are present that were not apparent on preoperative radiographs. Occasionally, an electronic WL may be inaccurate, so a wire shot is essential to avoid instrumenting short of the canal terminus and potentially creating a ledge, or instrumenting beyond the apical foramen and into the periapical tissues, which can cause inflammation or infection. Taken as a whole, it is recommended that WL be determined using a combination of electronic and radiographic information. Confirmation of WL can be obtained with the paper point test just prior to obturating the root canal system.
What is the current thinking regarding pain management and medications for endodontic procedures?
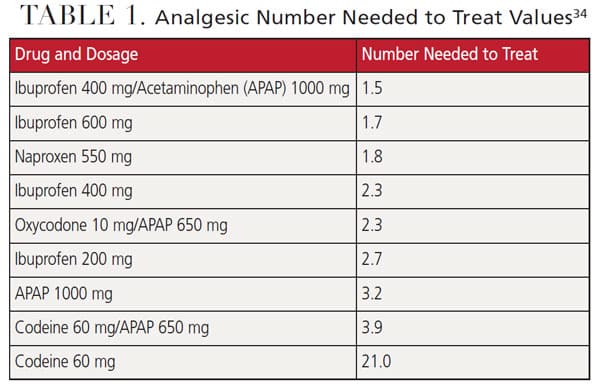
The effectiveness of a drug for pain management is measured by the number needed to treat (NNT), which is defined as the proportion of patients with at least 50% pain relief after four to six hours compared with a placebo in randomized, double-blind clinical trials. Low NNTs are better. For example, if you give a drug with an NNT of 2.0 to 100 patients, 50 of them will realize at least 50% pain relief. If you give a drug with an NNT of 1.0 to 100 patients, all of them will experience at least 50% pain relief.
A Cochrane review of 35 analyses consisting of 45,000 patients in 350 studies yielded the NNT values found in Table 1.34 Interpretation of the data shows the combination of ibuprofen and acetaminophen with an NNT of 1.5 is superior to all other drugs or drug combinations. When comparing the data in the last two rows of the table, it is clear that the pain relief comes from the acetaminophen in the codeine 60 mg/acetaminophen 650 mg combination (NNT of 3.9), not the codeine itself (which has an NNT of 21.0). As such, pain following endodontic treatment is best managed with a combination of ibuprofen and acetaminophen.
Antibiotics should only be prescribed when there is systemic involvement, such as trismus, dyspnea, dysphagia, vision problems, fever surpassing 102 degrees Fahrenheit, cellulitis or malaise. If a patient requires antibiotics for one of these indications, amoxicillin is the drug of choice because it is well absorbed and provides a broad antibacterial spectrum. Clindamycin is no longer recommended as the second drug of choice for penicillin-allergic patients due to the risk of pseudomembranous colitis caused by an overgrowth of Clostridoides difficile (formerly Clostridium difficile).35 Therefore, azithromycin (Z-pack) has become the second drug of choice for penicillin-allergic patients. Antibiotic guidelines have changed, and a loading dose is no longer recommended for any antibiotic except azithromycin.36
Please discuss the importance of endodontic case selection, especially for general practitioners. And what are the broad guidelines that would help dentists decide to treat or refer?
The decision to attempt root canal treatment or refer to an endodontist varies from clinician to clinician and depends on the individual’s skill and experience. The AAE has created a case assessment form and guidelines to aid in determining the relative complexity of a case. The assessment form is available at https://www.aae.org/specialty/wp-content/uploads/sites/䁰/떓/葎/葟AAE_CaseDifficultyAssessmentForm.pdf. In addition, the AAE has developed a free case assessment mobile app, called AAE EndoCase, that is available for download from app stores.
If complications arise during treatment, it is recommended the patient be referred to an endodontist before any iatrogenic errors occur which could jeopardize the tooth or compromise overall treatment success.
What do you see on the horizon for endodontic care?
The field of endodontics is changing rapidly and there is active research into using artificial intelligence (AI) to aid endodontic therapy and diagnosis. Specific applications being studied include assessing root canal anatomy, detecting periapical pathology, and diagnosing root fractures. Investigations are also underway to use AI for determining WL measurements, assessing the viability of dental pulp stem cells, and predicting the success of endodontic retreatment.37 In the future, it may be possible that AI software could “read” a CBCT scan and provide a radiology report. Future advances in AI might also include determining the best treatment options for a given patient, predicting drug interactions, and supporting robotic-assisted endodontic microsurgery.37 Clearly, this is an exciting time in endodontics.
REFERENCES
- Shen Y, Zhou HM, Zheng YF, Peng B, Haapasalso M. Current challenges and concepts of the thermomechanical treatment of nickel-titanium instruments. J Endod. 2013:39;163–172.
- Shen Y, Qian W, Abtin H, Gao Y, Haapasalo M. Fatigue testing of controlled memory wire nickel-titanium rotary instruments. J Endod. 2011;37:997–1001.
- Azim A, Piasecki L, da Silva Nieto UX, Timponi Goes Cruz A, Azim K. XP Shaper, a novel adaptive core rotary instrument: Micro-computerd tomographic analysis of its shaping abilities. J Endod. 2017;43:1532–1538.
- Gutarts R, Nusstein J, Reader A, Beck M. In vivo debridement efficacy of ultrasonic irrigation following hand-rotary instrumentation in human mandibular molars. J Endod. 2005;31:166–170.
- Jiang LM, Verhaagen B, Versluis M, van der Sluis LWM. Evaluation of a sonic device designed to activate irrigant in the root canal. J Endod. 2010;1:143–146.
- Molina B, Glickman G, Vandrangi P, Khakpour M. Evaluation of root canal debridement of human molars using the GentleWave system. J Endod. 2015;41:1701–1705.
- Sigurdsson A, Garland R, Le K, Woo SM. 12-month healing rates after endodontic therapy using the novel GentleWave system: a prospective multicenter clinical study. J Endod. 2016;42:1040–1048.
- Peters OA, Bardsley S, Fong J, Pandher G, DiVio E. Disinfection of root canals with photon-initiated photoacoustic streaming. J Endod. 2011;37:1008–1012.
- Caputa PE, Retsas A, Kuijk L, Chavez de Paz LE, Boutsioukis C. Ultrasonic irrigant activation during root canal treatment: A systematic review. J Endod. 2019;1:31–44.
- Von Arx T. Apical surgery: A review of current techniques and outcome. Saudi Dent J. 2011;23:9–15.
- Vishwanath V, Rao HM. Gutta-percha in endodontics — A comprehensive review of material science. J Conserv Dent. 2019;22:216–222.
- American Association of Endodontists. AAE and AAOMR Joint Position Statement. Use of Cone Beam Computed Tomography in Endodontics — 2015 Update. Available at: http://aaomr.org/wp-content/uploads/떕/葕/aae-aaomr-2015update.pdf. Accessed July 11, 2022.
- Robinson D, Goerig AC, Neaverth EJ. Endodontic access: An update, Part I. Compendium. 1989;10:290–298.
- Ludlow JB, Timothy R, Walker C, et al. Effective dose of dental CBCT — a meta analysis of published data and additional data for nine CBCT units. Dentomaxillofac Radiol. 2015;44:20140197.
- Patel S, Durack C, Abella F, Shemesh H, Roig M, Lemberg K. Cone beam computed tomography in endodontics — a review. Int Endod J. 2014;48:3–15.
- Bornstein MM, Luber R, Sendi P, von Arx T. Comparison of periapical radiography and limited cone-beam computed tomography in mandibular molars for analysis of anatomical landmarks before apical surgery. J Endod. 2011;37:151–157.
- Ee J, Fayad MI, Johnson BR. Comparison of endodontic diagnosis and treatment planning decisions using cone-beam volumetric tomography versus periapical radiography. J Endod. 2014;40:910–916.
- Fox A, Basrani B, Kishin A, Lam EWN. A novel method for characterizing beam hardening artifacts in cone-beam computed tomographic images. J Endod. 2018;44:869–874.
- PradeepKumar AR, Shemesh H, Nivedhitha S, et al. Diagnosis of vertical root fractures by cone-beam computed tomography in root filled teeth with confirmation by direct visualization: A systematic review and meta-analysis. J Endod. 2021;47:1198–1214.
- Fayad MI, Ashkkenaz PJ, Johnson BR. Different representations of vertical root fractures detected by cone-beam volumetric tomography: A case series report. J Endod. 2012;38:1435–1442.
- Sjogren U, Hagglund B, Sundqvist G, Wing K. Factors affecting the long-term results of endodontic treatment. J Endod. 1990;16:498–504.
- Ricucci D, Langeland K. Apical limit of root canal instrumentation and obturation, Part 2. A histological study. Int Endod J. 1998;31:394–409.
- Ng Y-L, Mann V, Rahbaran S, Lewsey J, Gulibivala K. Outcome of primary root canal treatment: systematic review of the literature — Part 2: Influence of clinical factors. Int Endo J. 2008;41;6–31.
- McDonald NJ, Hovland EJ. An evaluation of the apex locator Endocator. J Endod. 1990;16:5–8.
- Bramante CM, Berbert A. A critical evaluation of some methods of determining tooth length. Oral Surg. 1974;37:463–473.
- Goldman M, Pearson AH, Darzenta N. Endodontic success: who’s reading the radiograph? Oral Surg. 1972;33:432–439.
- Rosenberg DB. The paper point technique, Part 2. Dent Today. 2003:22(3):62–64, 66,67.
- Rosenberg DB. The paper point technique. Part 1. Dent Today. 2003:22(2):80–86.
- Seidberg BH, Alibrandi BV, Fine H, Logue B. Clinical investigation of measuring working lengths of root canals with an electronic device and with digital-tactile sense. J Am Dent Assoc. 1975;90:379–387.
- Suzuki K. Experimental study on iontophoresis. J Jap Stoma. 1942;16:411–417.
- Jenkins JA, Walker WA, Schindler WG, Flores CM. An in vitro evaluation of the accuracy of the root ZX in the presence of various irrigants. J Endod. 2001;27:209–211.
- Dunlap CA, Remeikis NA, BeGole EA, Rauschenberger CR. An in vivo evaluation of an electronic apex locator that uses the ratio method in vital and necrotic canals. J Endod. 1998;24:48–50.
- De Camargo EJ, Zapata RO, Medeiros PL, et al. Influence of preflaring on the accuracy of length determination with four electronic apex locators. J Endod. 2009;35:1300–1302.
- Moore RA, Derry S, McQuay HJ, Wiffen PJ. Single dose oral analgesics for acute postoperative pain in adults. Cochrane Database Syst Rev. 2011;9:CD008659.
- American Association of Endodontists. AAE guidance on the use of systemic antibiotics in endodontics: AAE position statement. Available at: https://www.aae.org/specialty/wp-content/uploads/sites/䁰/떑/葒/aae_systemic-antibiotics.pdf. Accessed July 11, 2022.
- Johnson M. Endodontics and antibiotic update. AAE Colleagues for Excellence. Fall 2019.
- Aminoshariae A, Kulild J, Nagendrababu V. Artificial intelligence in endodontics: Current applications and future directions. J Endod. 2021;47:1352–1357.
From Decisions in Dentistry. August 2022;8(8)16-19.


