 YELENA RODRIGUEZ MENA/ISTOCK/GETTY IMAGES PLUS
YELENA RODRIGUEZ MENA/ISTOCK/GETTY IMAGES PLUS
Introduction to Regenerative Endodontics
Regenerative endodontics is a biologically based approach that induces bleeding from the periapical region into the canal, thus generating vital tissue that provides a natural defense against reinfection
 By definition, bioactive means having a biological effect. Bioactive endodontics is achieved by performing regenerative procedures.1 More specifically, regenerative endodontics is a biologically based technique designed to physiologically replace damaged tooth structures, including dentin and root structures, as well as the pulp-dentin complex.2 The endodontic literature uses regenerative, revascularization and revitalization endodontics interchangeably.3
By definition, bioactive means having a biological effect. Bioactive endodontics is achieved by performing regenerative procedures.1 More specifically, regenerative endodontics is a biologically based technique designed to physiologically replace damaged tooth structures, including dentin and root structures, as well as the pulp-dentin complex.2 The endodontic literature uses regenerative, revascularization and revitalization endodontics interchangeably.3
Clinically, regenerative endodontics was first developed for immature necrotic permanent teeth in order to achieve root end closure, with the additional goal of obtaining continued development of the root and thickening of the canal wall.4,5 Subsequently, regenerative endodontics has been performed on necrotic mature permanent teeth, vital mature permanent teeth, teeth that have resorption (i.e., history of trauma), and for retreatment of teeth obturated with gutta-percha and sealer.6–8
In conventional endodontic therapy, the canal is obturated with biocompatible foreign materials, such as gutta-percha and sealer. By comparison, in regenerative endodontics the canal is filled with the host’s own vital tissue (i.e., blood).9
REGENERATIVE METHODOLOGY
After taking radiographs — periapical, bitewing and/or cone beam computed tomography — and reaching a pretreatment pulpal and periradicular diagnosis that indicates the tooth is restorable without the need for a post and core, the treatment tooth is anesthetized and a rubber dam is placed.
Once canal access is obtained, hand files are used in combination with an apex locator and periapical radiograph to determine the canal working length. Following canal preparation (depending on the type of tooth: mature, immature, necrotic or vital pulp), the canal should be irrigated with 1.5% sodium hypochlorite (NaOCl); note that a lower concentration is used to avoid killing stem cells.10 When irrigating with NaOCl, it is clinically important to minimize the possibility of irrigant extrusion into the periapical region. After the canal is prepared and irrigated with NaOCl, it should be dried and irrigated with 17% ethylenediaminetetraacetic acid (EDTA) before placing calcium hydroxide in the canal.
 Research shows the survival of stem cells from the apical papilla and growth factor released from the dentin is improved with the use of EDTA and calcium hydroxide.10 By comparison, the use of high concentrations of NaOCl and antibiotic paste negatively impact stem cell and growth factor survival.11
Research shows the survival of stem cells from the apical papilla and growth factor released from the dentin is improved with the use of EDTA and calcium hydroxide.10 By comparison, the use of high concentrations of NaOCl and antibiotic paste negatively impact stem cell and growth factor survival.11
Next, the canal access is temporized with a sterile cotton pellet or sponge and a temporary filling material or glass ionomer. At this point, the patient should be asked to return in one to four weeks for follow-up treatment.9
SECOND APPOINTMENT
At the second regenerative endodontic treatment appointment, the clinician should assess the patient’s response to the initial regenerative therapy. If there are signs or symptoms of persistent infection, providers should consider repeating the first treatment.9
If the tooth is asymptomatic, 3% mepivacaine without a vasoconstrictor should be used for local anesthesia (the lack of a vasoconstrictor will allow for periapical bleeding). After a rubber dam is placed and access achieved, 17% EDTA is used to irrigate the canal. This is done in conjunction with the use of files to remove the calcium hydroxide and prepare the dentin. The canal is dried and bleeding is induced with a hand file inserted 1 to 2 mm beyond the apical foramen. Bleeding should be stopped at a level in the canal (cemento-enamel junction) that will allow for 3 to 4 mm of restorative material.9
The induction of periapical bleeding into the canal space is necessary in regenerative endodontics procedures. This induced bleeding brings scaffolding, stem cells and blood-derived bioactive growth factors. These growth factors are in addition to the growth factors embedded in the dentinal matrix that are released when 17% EDTA is used as an irrigant.10,12,13 This triad of stem cells, scaffolding and growth factors enables tissue regeneration to occur.14
After a resorbable collagen matrix is situated over the blood clot, a bioceramic material is placed directly over the collagen matrix and a glass ionomer is positioned over the bioceramic material. Next, a final composite or amalgam restoration is placed over the glass ionomer and a final radiograph is taken. The patient should be put on a six-month recall for up to three years, as healing dictates.15
 Clinically, it is important to know the primary goal of bioactive endodontic treatment is the elimination of symptoms. Secondary goals include radiographic healing and continued root development, and a tertiary goal is a positive response to sensibility testing (e.g., thermal testing or use of an electronic pulp tester).16,17
Clinically, it is important to know the primary goal of bioactive endodontic treatment is the elimination of symptoms. Secondary goals include radiographic healing and continued root development, and a tertiary goal is a positive response to sensibility testing (e.g., thermal testing or use of an electronic pulp tester).16,17
SPECIFIC CONSIDERATIONS
As noted, regenerative endodontics was first developed for immature necrotic permanent teeth.4,5 Prior to regenerative treatment, apexification procedures on immature necrotic permanent teeth were performed with either the use of calcium hydroxide or mineral trioxide aggregate.18 Unlike regenerative therapy, these procedures resulted in closing the apex, but did not help develop the root or thicken canal walls.4,5 Regenerative endodontics has subsequently been used for necrotic mature permanent teeth, vital mature permanent dentition, teeth with resorption, and for retreatment of gutta-percha- and sealer-obturated teeth presenting with persistent apical periodontitis.6–8
Although the basic regenerative endodontic clinical technique is similar when being performed on immature, mature, necrotic or vital permanent teeth, there are some clinical variations.1,9
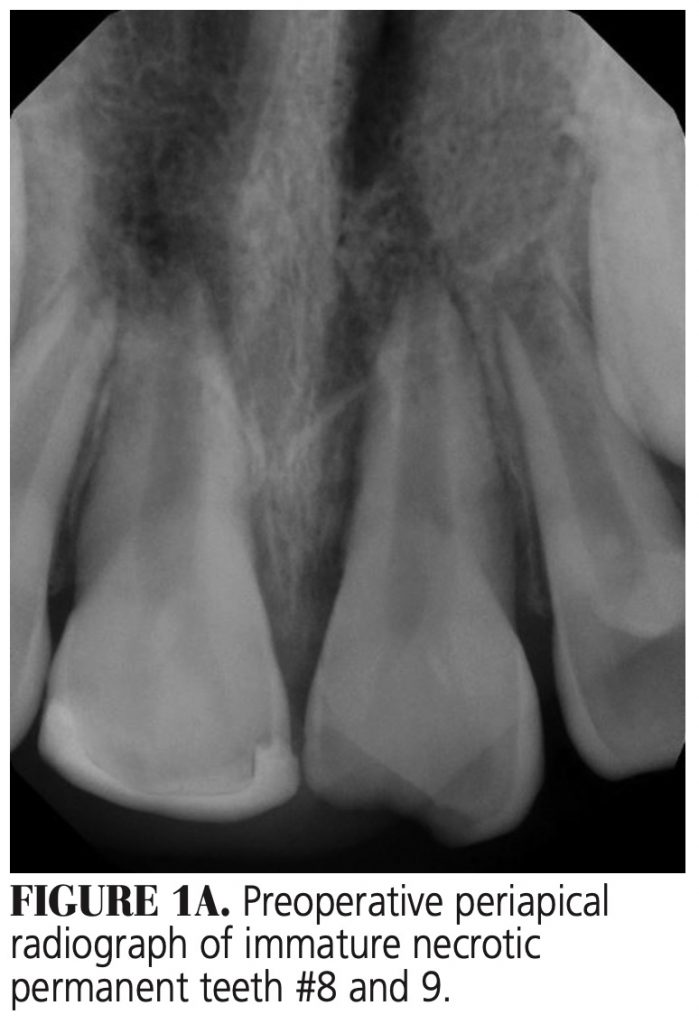
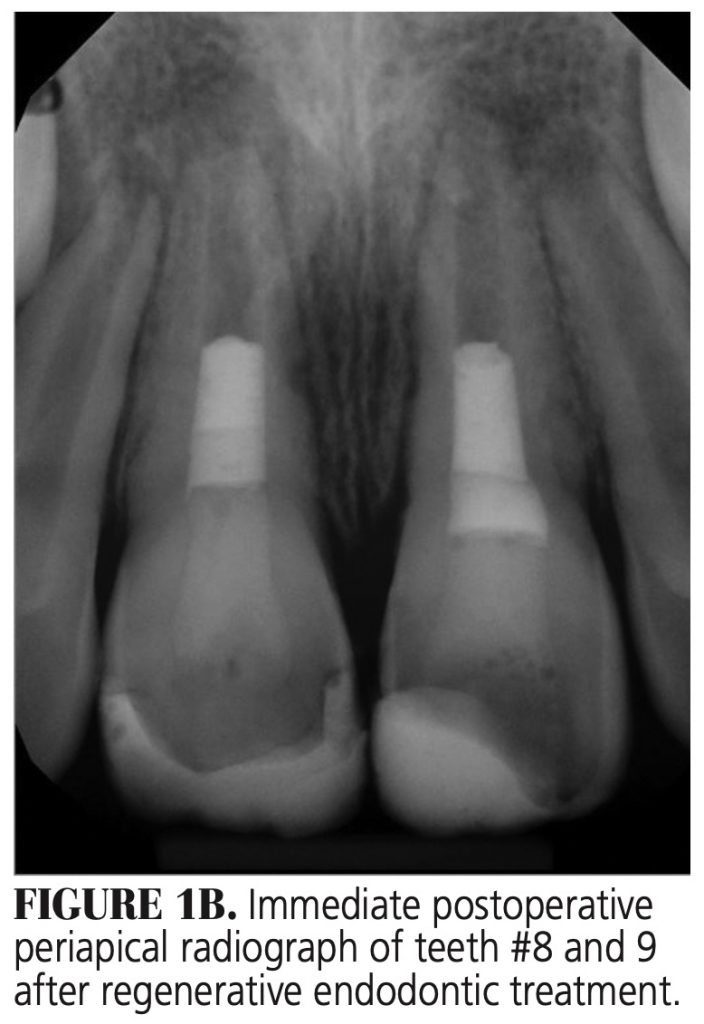
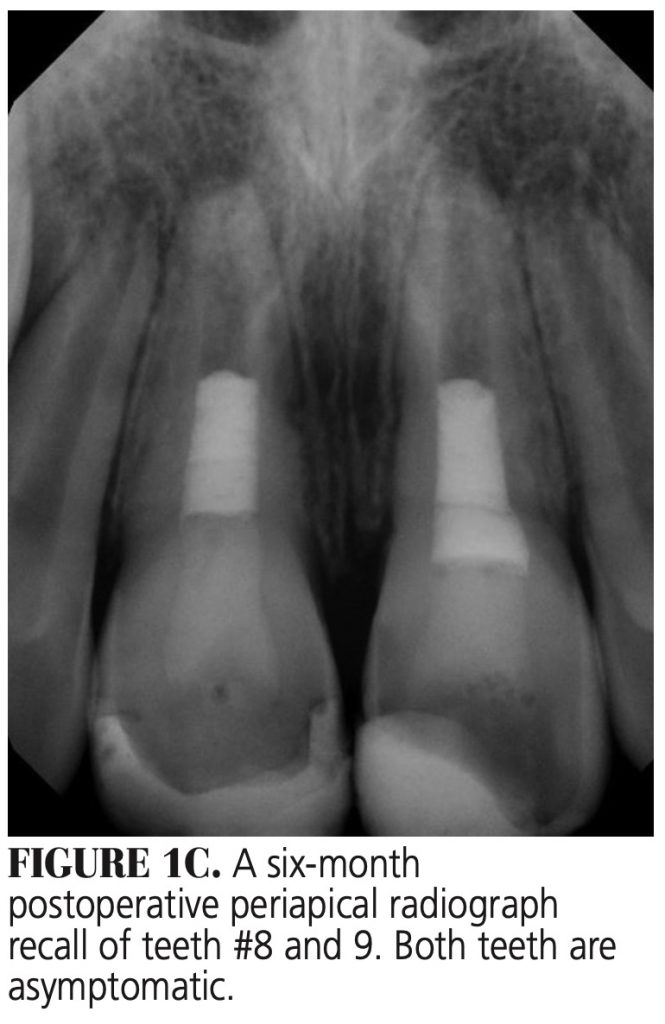
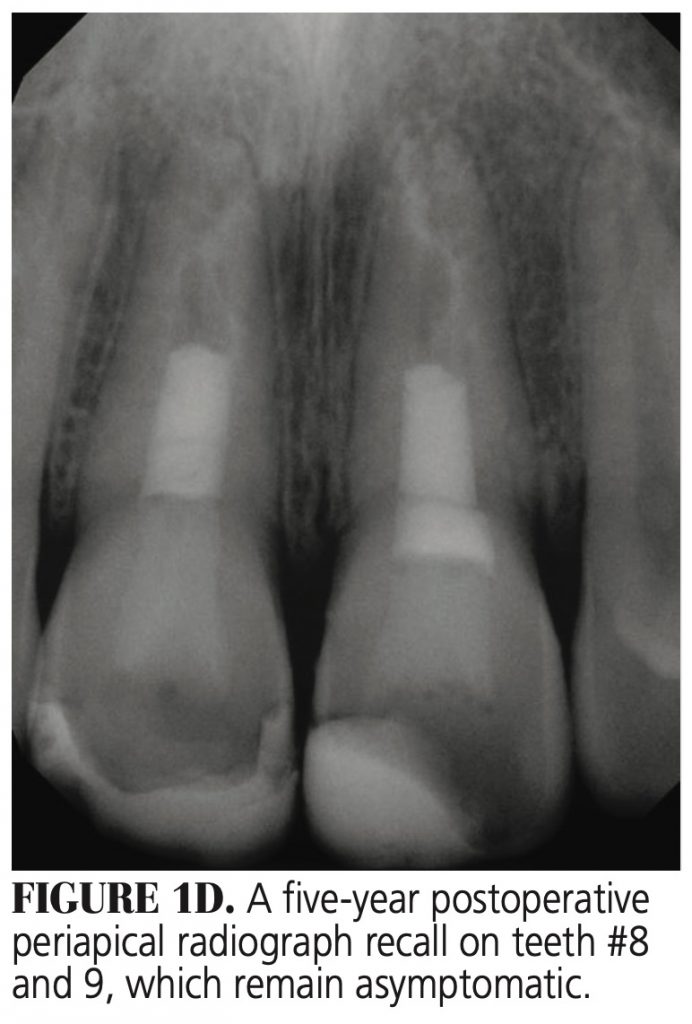
Immature Necrotic Permanent Teeth — Removal of the necrotic pulp tissue from the canal with minimum canal preparation is required because the canal walls are thin and over-preparation can weaken the root structure, making it susceptible to fracture (Figures 1A through 1D).19
Mature Necrotic Permanent Teeth — Unlike immature necrotic permanent teeth, regenerative endodontic procedures on mature necrotic permanent teeth require complete mechanical debridement to help eliminate necrotic pulpal tissue.9 While thickening of the canal walls or continued root development is not expected following regenerative treatment, apical closure can take place.20
 Mature Vital Permanent Teeth — Regenerative endodontic treatment of vital mature permanent teeth follows the same steps used for necrotic mature permanent teeth, except in vital teeth the bacterial infection component, as observed in necrotic pulp cases, is not the same (because little to no pulpal bacteria is present).21,22 Therefore, these teeth can be treated in a single visit, thus eliminating the need for placement of calcium hydroxide into the canal. In a study by Wang et al,23 the authors reported the existence of functional stem cells in clinically compromised dental pulp with irreversible pulpitis.
Mature Vital Permanent Teeth — Regenerative endodontic treatment of vital mature permanent teeth follows the same steps used for necrotic mature permanent teeth, except in vital teeth the bacterial infection component, as observed in necrotic pulp cases, is not the same (because little to no pulpal bacteria is present).21,22 Therefore, these teeth can be treated in a single visit, thus eliminating the need for placement of calcium hydroxide into the canal. In a study by Wang et al,23 the authors reported the existence of functional stem cells in clinically compromised dental pulp with irreversible pulpitis.
Traumatized Teeth With External Root Resorption — Regenerative endodontic procedures on teeth with external inflammatory root resection mirrors the technique used for immature, necrotic permanent teeth.8 Such treatment is performed with minimum canal preparation in order to prevent thinning of the canal walls and weakening of the root structure (Figures 2A through 2C).
It is important to note the older literature recommended regenerative endodontic treatment be used on root canals with an apical foramen at least 1 mm in diameter.24 The current literature states the final canal size of the apical foramen can be a master apical file size of 0.32 mm. This smaller size has been shown to still allow the blood cells from the periapical tissue to migrate into the canal space.9
BIOACTIVE EFFECTS
The tissue generated in a canal after a regenerative endodontic procedure resembles cementum, bone and periodontal ligaments; in other words, tissues with blood vessels and nerves.9 Although these are not true pulpal tissue, they are the host’s own vital tissue.
This is important because (as compared to the use of foreign obturation materials, such as gutta-percha and sealer) this vital tissue contains immune defense mechanisms to protect itself from foreign invaders.9,21
SUMMARY
As opposed to conventional endodontic treatment, regenerative endodontics offers clinicians and patients a more biologically based approach. This technique involves canal preparation, followed by the induction of bleeding from the periapical region into the canal. The vital tissues generated in the canals after a regenerative procedure provide a natural defense against reinfection by pathogenic bacteria.
Although this approach has distinct clinical benefits, additional study is needed to confirm the long-term prognosis when bioactive endodontic treatment is utilized.
The authors have no commercial conflicts of interest to disclose.
Key Takeaways
- Regenerative endodontics is a biologically based approach designed to physiologically replace damaged tooth structures, including dentin and root structures, as well as the pulp-dentin complex.2
- In conventional endodontic therapy, the canal is obturated with biocompatible foreign materials, such as gutta-percha and sealer, while in regenerative endodontics the canal is filled with the host’s own vital tissue (i.e., blood).9
- Induced bleeding brings scaffolding, stem cells and blood-derived bioactive growth factors into the canal, allowing regeneration to occur.14
- After the canal is prepared and irrigated with 1.5% sodium hypochlorite, it should be dried and irrigated with 17% ethylenediaminetetraacetic acid before placing calcium hydroxide in the canal. This combination improves survival of stem cells from the apical papilla and growth factor released from the dentin.
- Ultimately, the vital tissues generated in the canals after a regenerative procedure provide a natural defense against reinfection by pathogenic bacteria.
References
- Bahcall J, Xie Q, Baker M, Weeks S, Huliyar V. Bioactive endodontics for mature permanent teeth. Inside Dent. 2019;15:43–47.
- Murray P, Garcia-Godoy F, Hargreaves K. Regenerative endodontics: a review of current status and a call for actionJ J Endod. 2007;33:377–390.
- Kim SG, Malek M, Sigurdsson A, Lin LM, Kahler B. Regenerative endodontics: a comprehensive review. Int Endod J. 2018;51:1367–1388.
- Ostby BN. The role of the blood in endodontic therapy. An experimental histological study. Acta Odontol Scand. 1961;19:324–353.
- Banchs F, Trope M. Revascularization of immature permanent teeth with apical periodontitis: new treatment protocol. J Endod. 2004;30:196–200.
- Saoud TM, Martin G, Chen YH, et al. Treatment of mature permanent teeth with necrotic pulps and apical periodontitis using regenerative endodontic procedures: a case series. J Endod. 2016;42:57–64.
- Saoud TM, Huang G, Gibbs J, Sigurdsson A, Lin LM. Management of teeth with persistent apical periodontitis after root canal treatment using regenerative endodontic therapy. J Endod. 2015; 41:1743–1748.
- Santiago C, Pinto S, Sassone L, Hirata R, Fidel S. Revascularization technique for the treatment of external inflammatory root resorption: a report of 3 cases. J Endod. 2015;41:1560–1564.
- Saoud TM, Ricucci D, Lin LM, Gaengler P. Regeneration and repair in endodontics — a special issue of the regenerative endodontics — a new era in clinical endodontics. Dent J (Basel). 2016;4:3.
- Gallar KM, Buchalla W, Hiller KA, et al. Influence of root canal disinfectants on growth factor release from dentin. J Endod. 2015;41:363–368.
- Althumairy RI, Teixeira FB, Diogenes A. Effect of dentin conditioning with intracanal medicaments on survival of stem cells of apical papilla. J Endod. 2014;40 521–525.
- Casagrande L, Demarco FF, Zhang Z, Araujo FB, Shi S, Nör JE. Dentin-derived BMP-2 and odontoblast differentiation. J Dent Res. 2010;89:603–608.
- Cao Y, Song M, Kim E, et al. Pulp-dentin regeneration: current state and future prospects. J Dent Res. 2015;94:1544–1551.
- Langer R, Vacanti J. Tissue engineering. Science. 1993;260:920–926.
- Kahler B, Mistry S, Moule A, et al. Revascularization outcomes: a prospective analysis of 16 consecutive cases. J Endod. 2014;40:333–338.
- AAE Clinical Considerations for a Regenerative Procedure. Revised 2016. Available at: http/://www.aae.org/uploadedfiles/publicatio_s_and_research/research/ currentregenerativeendodonticconsiderations.pdf. Accessed September 2, 2020.
- Diogenes A, Ruparel N, Shiloah Y, Hargreaves KM. Regenerative endodontics. J Am Dent Assoc. 2016;147:372–380.
- Chala S, Abouqal R, Rida S. Apexification of immature teeth with calcium hydroxide or mineral trioxide aggregate: systemic review and meta-analysis. Oral Surg Oral Med Oral Pathol Radiol Endod. 2011;112:e36–e42.
- Cvek M. Prognosis of luxated non-vital maxillary incisors treated with calcium hydroxide and filled with gutta-percha: a retrospective clinical study. Endod Dent Traumatol. 1992;8:45–55.
- Saoud TM, Sigurdsson A, Rosenberg PA, Lin LM, Ricucci D. Treatment of a large cystlike inflammatory periapical lesion associated with mature necrotic teeth using regenerative endodontic therapy. J Endod. 2014;40:2081–2086.
- Nagaoka S, Miyazaki Y, Liu HJ, Iwamoto Y, Kitano M, Kawagoe M Bacterial invasion into dentinal tubules of human vital and nonvital teeth. J Endod. 1995;21:70–73.
- Ricucci D, Loghin S, Siqueira J. Correlation between clinical and histological diagnosis. J Endod. 2014;40:1932–1939.
- Wang Z, Pan J, Wright J, et al. Putative stem cells in human dental pulp with irreversible pulpitis: an exploratory study. J Endod. 2010;36:820–825.
- Kling M, Cvek M, Mejare I. Rate and predictability of pulp revascularization in therapeutically reimplanted permanent incisors. Endo Dent Traumatol. 1986;2:83–89.
From Decisions in Dentistry. October 2020;6(9):8, 10, 12-13.



