Impact of Keratinized Mucosa in Dental Implant Treatment
Learning to readily identify and treat potential soft tissue deficiencies can improve outcomes in dental implant treatment.
This course was published in the September 2020 issue and expires September 2023. The author has no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Explain the role of keratinized mucosa in dental implant treatment outcomes, and the controversy surrounding this topic.
- Describe techniques used to augment keratinized tissue.
- Discuss clinical considerations when probing peri-implant tissues to assess attachment and inflammation.
Dental implants do not function entirely like teeth. In large part, this is due to the unique organization of epithelium, connective tissue and alveolar bone that forms during wound healing and surrounds the implant-abutment complex. In health, bone intimately contacts and supports the implant body, while the soft tissue (i.e., peri-implant mucosa) protects and maintains the overall tissue architecture. A band of keratinized mucosa runs from the peri-implant mucosa to the elastic, oral mucosa along most implants.1 The amount and thickness of keratinized mucosa needed around dental implants to prevent the destructive sequelae of inflammation is controversial.
Clinicians have adopted techniques to augment this band of attached tissue. These include the use of various autograft, allograft or xenograft materials, with varying success rates and predictability.2 Procedures to enhance inherently thin periodontal phenotypes have also proven successful in preventing mucosal recession and/or bone loss around implants, especially for those placed in fresh extraction sockets.3 Learning to readily identify and treat potential soft tissue deficiencies can improve implant outcomes and patient satisfaction.
In implant treatment, it is important to understand the structural differences between teeth and implants. In the healthy periodontium, the periodontal ligament and supracrestal attached tissues anchor the tooth in place. A thin, protective layer of junctional epithelium is found more coronally. The peri-implant mucosa, however, is the result of tissue injury that takes months to fully mature.4 In fact, while a junctional epithelium similar to teeth forms by two weeks, the barrier epithelium or implant-mucosal seal is not well established until at least six weeks after implant placement.5 Between this epithelial layer and the peri-implant bone, there is a connective tissue component that tightly adapts, but does not attach, to the implant surface. Most of this connective tissue consists of collagen matrix (85%), but it also includes fibroblasts (3%) and vascular tissue (5%). An inner connective tissue layer, more abundant in fibroblasts, has been identified immediately around the implant surface. Inflammatory cells, such as neutrophils, lymphocytes and macrophages, are also present in small amounts. In general, the peri-implant mucosa is approximately 3 to 4 mm in thickness.1
Keratinized mucosa is found at the vast majority of implant sites. It includes a highly collagenous connective tissue layer with an orthokeratinized epithelium.1 In a comparative study, the width of keratinized mucosa was about 1 mm less at implant sites compared to the contralateral natural teeth. The thickness of keratinized mucosa at the depth of the sulcus was 1 mm greater at implant sites than in natural teeth.6 Since healthy peri-implant soft tissues are important for implant success, it is essential to monitor soft tissue parameters around implants during initial healing and after loading. This begins with a visual examination of the implant site, while probing helps detect changes in tissue attachment and inflammation by identifying bleeding tendency. It is important to note the peri-implant apparatus is more delicate and sensitive to probing than teeth, so use of a lighter probing force is advised to improve diagnostic accuracy.7
CONTROVERSIAL ROLE OF KERATINIZED MUCOSA
Unlike peri-implant disease, soft tissue deficiencies can exist both before and after implant placement. The 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions recognized that the amount of keratinized tissue needs to support all stages of implant treatment, from surgery to the final prosthesis. This includes tension-free closure of a ridge augmentation procedure, complete coverage of the implant after placement, or passive flap adaptation around the implant collar to achieve optimal form and esthetics. The workshop, however, could neither quantify an “ideal” amount of peri-implant keratinized mucosa, nor determine its effect on implant health and maintenance over time.8
There are numerous examples of how a narrow or absent band of keratinized tissue negatively affects implant treatment outcomes. In a four-year retrospective study, implants with < 2 mm of keratinized mucosa had more bleeding sites and higher plaque scores. Mucosal width and time in function also had a significant effect on marginal bone loss in a multilevel model.9 A systematic review found that implants with wider bands of keratinized mucosa had significantly less attachment loss, plaque accumulation, and mucosal recession and inflammation, with trends toward shallower probing depths and decreased radiographic bone loss.10 When evaluating outcomes of mandibular implant-assisted overdentures, greater plaque and gingival index scores were noted at implant sites without keratinized mucosa.11 A six-month follow-up study found that patients without keratinized mucosa were less satisfied with the implant soft tissue esthetics and initially had more recession.12 As a possible indicator for peri-implant disease progression, < 2 mm of keratinized mucosa was significantly associated with the severity of peri-implant mucositis.13
While most studies have confirmed the importance of a healthy band of keratinized tissue around implants, others contrarily found no association with implant health. In a five-year retrospective study, the width of keratinized mucosa did not correlate to any clinical parameters of peri-implant health, including probing depth, bleeding on probing, or marginal bone position. Furthermore, this study was unable to define a necessary threshold for the width of mid-buccal keratinized mucosa.14 A separate longitudinal study that stratified keratinized mucosa by width, implants by surface configuration and location, as well as type of prosthesis, found no association between width of keratinized tissue and bone loss after three years.15
Despite these controversial findings, a defined role for keratinized mucosa around implants has recently been proposed. Implants without keratinized mucosa in the posterior mandible had higher plaque accumulation, soft tissue recession, and sites requiring intervention over a 10-year period.16 In implant therapy, the negative effects of poor plaque control and irregular maintenance on peri-implant health are well documented.17
SOFT TISSUE AUGMENTATION
Various autograft, allograft and xenograft materials to increase the amount and thickness of keratinized mucosa have been identified. In a recent dog study, implants and healing abutments were placed after two months of healing, with either partial or total excision of the buccal keratinized tissue. At implant sites, free soft tissue autografts resulted in the greatest gain of keratinized tissue height (5.0 to 5.5 mm) compared to an apically positioned flap or xenogenic collagen matrix. Interestingly, keratinized tissue regenerated to a lesser extent around implants than teeth by about 1 mm after 60 days.18
An additional benefit of free soft tissue autografts is the increase in vestibular depth during preparation of the recipient bed. This is important since a shallow vestibule often contributes to poorer implant parameters. Vestibular depths ≤ 4 mm led to significantly increased mucosal recession, attachment loss, and peri-implant bone loss in a six-year retrospective study. Slightly greater bleeding and gingival indices were also noted.19 The free soft tissue autograft technique may be particularly helpful in patients planned for implant-assisted prostheses, such as overdentures, that are still reliant on intraoral hard and soft tissues for support and stability.
Other grafting techniques can be used to augment keratinized mucosa. Connective tissue grafting has been shown to increase soft tissue thickness and mucosal width around dental implants by 2.5 mm on average within the first three months.20 The use of an acellular dermal matrix allograft at implant sites increased the width of keratinized mucosa from approximately 0.8 mm to 3.2 mm at three months, with mild soft tissue relapse at six months.21 Xenogenic collagen matrix soft tissue augmentation of implant sites led to a mean keratinized tissue width of 2.8 mm; although this value was not statistically different from that achieved by connective tissue grafting at six months. In addition, patients reported less pain during the perioperative period.22 A separate investigation found that buccal mucosal thickness increased with use of a xenogenic collagen matrix to approximately 0.9 mm, although connective tissue grafting had a more favorable result of nearly 1.2 mm.23 Overall, both allografts and xenografts are sound alternatives when there is concern for discomfort from a second surgical site, or when treatment of multiple defects does not lend itself to palatal harvesting alone.
In addition to the width of the keratinized tissue band, tissue thickness is also important in maintaining the peri-implant mucosa. Thinner soft tissue (e.g., < 2 mm) has been significantly correlated with increased bone loss after molar implant placement, likely due to remodeling of supracrestal attached tissues.24 Adding a connective graft helps increase and maintain the dimensions of keratinized mucosa around implants for better wound healing and esthetic outcomes.25 This is especially true in patients with thin periodontal phenotypes, which are more often associated with recession.3 The process of thickening a phenotype or “phenotype modification” via soft tissue grafting has led to long-term tissue stability around teeth.26 This technique was more recently adapted to implants. A connective tissue graft at time of immediate implant placement converted thin phenotypes to thick, with better esthetic criteria after two years.27 Taken together, these studies support a shift toward peri-implant health with soft tissue grafting.
CASE REPORT
The following case outlines the steps taken to correct implant soft tissue deficiencies by means of a free soft tissue autograft. A 75-year old male presented to the graduate periodontics clinic at the University of Texas Health Science Center, San Antonio School of Dentistry interested in implant treatment to replace nonrestorable teeth in a partially edentulous mandible (Figure 1 through Figure 12). The patient had a medical history significant for joint replacement, peripheral neuropathy, benign prostatic hyperplasia, and ulcerative colitis. He was taking loperamide, mesalamine, tamsulosin, zolpidem, finasteride and tolterodine. After a thorough evaluation and review of treatment options, a plan was developed for placing implants. Procedures were performed using IV conscious sedation. The treatment plan included:
- Extraction of nonrestorable teeth, with site preservation to minimize any future hard tissue deficiencies.
- Placement of two implants, with alveoloplasty for an implant-assisted overdenture.
- Free soft tissue autograft at time of implant uncovery to increase the amount and thickness of keratinized mucosa around both implants for better implant health and self-care maintenance.
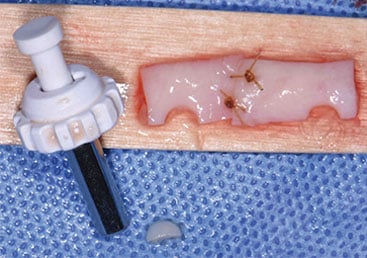
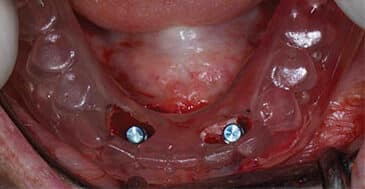
![Recipient site with apically sutured flap margins]()
FIGURE 6. Recipient site with apically sutured flap margins after attachment of implant healing abutments.
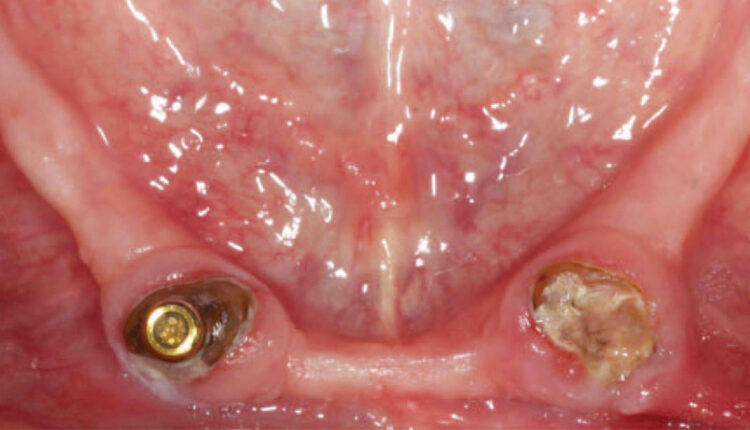
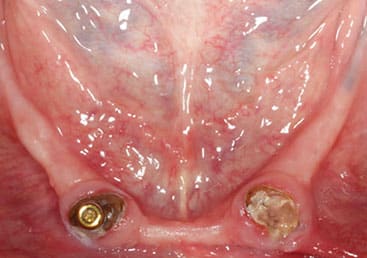
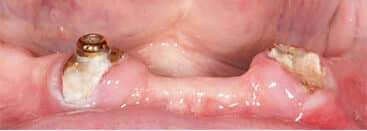
Upon initial evaluation, teeth #22 and 27 served as root-supported locator attachments for a mandibular overdenture. They were deemed nonrestorable due to recurrent caries. While the maxilla met anatomical expectations to support a complete denture, the mandible had fair to poor support, stability and retention because of moderate ridge atrophy. The posterior ridge was thin, with significant loss of buccal vestibular depth, while the anterior ridge was compromised by a shallow labial vestibule with a high lingual frenum attachment. Multiple options were presented for the mandible, including an implant fixed complete denture, implant-assisted bar overdenture, implant-assisted locator attachment overdenture, and conventional complete denture.
Due to gingival recession and a complete lack of attached keratinized tissue, bony dehiscences were anticipated after extractions. The need for hard and soft tissue augmentation was explained, and the patient elected to proceed with treatment, choosing a maxillary complete denture opposing a mandibular, implant-assisted locator attachment overdenture.
On the first day of surgery, care was taken to ensure teeth #22 and 27 were extracted with a minimally traumatic technique. Full-thickness flaps were elevated to allow for complete visualization of the buccal and lingual plates. After socket degranulation, 7 mm and 4 mm buccal dehiscences were noted at each site. Sharp bony peaks were smoothed, then extraction sockets were grafted with bovine-derived xenograft blocks with 10% collagen. While primary closure was not achieved, it was anticipated the lack of keratinized tissue would prove challenging for implant placement.
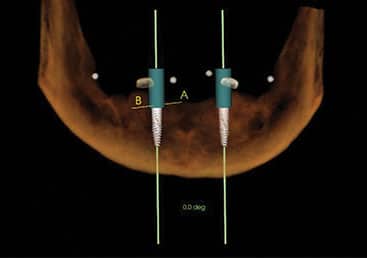
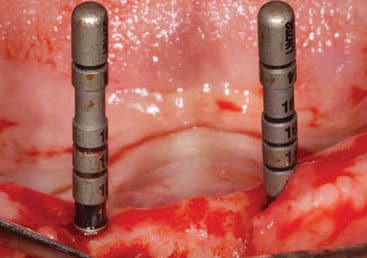
After four months of healing, cone beam imaging was used to evaluate ridge dimensions. Due to the displacement of the anterior loop of the mental nerve toward the midline, implants were planned at the #23 and 26 sites to avoid nerve impingement. At time of implant surgery, full-thickness flaps were elevated with a slight lingual bias to preserve remaining keratinized tissue and transpose it buccally around the implant. Two bone-level, tapered, 3.3-mm-wide and 10-mm-long implants were placed. A bone tapping protocol was also used due to dense cortical bone. No further bone grafting was indicated.
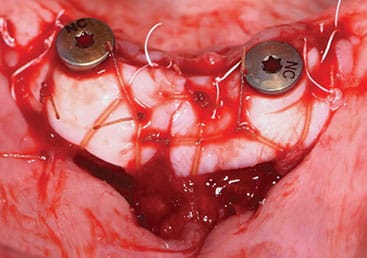
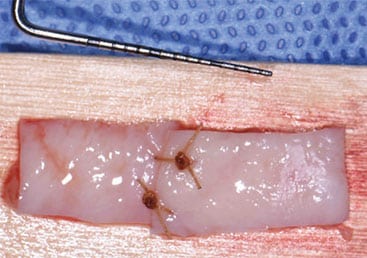
Three months following implant placement, a free soft tissue autograft was performed at the time of uncovering to increase the amount of keratinized mucosa around the implants and deepen the labial vestibule. A linear incision was placed coronal to the mucogingival junction from sites #23 to 26, with oblique vertical releases at the distal aspects of sites #23 and 26. A partial thickness flap was reflected at least 12 mm apical to the incision. After 4-mm-tall healing abutments were attached, the apical mucosal border of the flap margin was sutured. Next, free soft tissue autografts were harvested and approximated to achieve adequate width for the recipient bed. Before the grafts were stabilized to the recipient bed, a tissue punch was used to ensure intimate contact against the implant surfaces around the buccal and interproximal aspects.
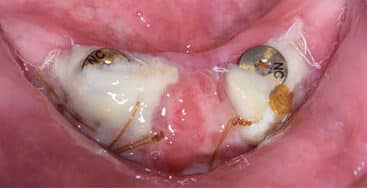
The patient’s existing complete denture served as a palatal stent to minimize bleeding and discomfort postoperatively. He was instructed to avoid manipulating the surgical sites, as it is important to remind patients that what may appear abnormal is, in fact, normal healing. For example, at the one- and two-week follow-ups, the graft may seem infected or necrosed, when it is simply incorporating into the recipient site. A thicker graft, as used here, undergoes greater primary contraction, but has long-term resistance to functional stresses from the removable appliance. A thick, healthy band of keratinized mucosa and increased vestibular depth were evident in this case at four weeks postsurgery. The patient returned to his restorative dentist for fabrication of his final mandibular, implant-assisted overdenture, and will be seen for yearly recalls.
SUMMARY AND CONCLUSIONS
Despite the controversy that surrounds the impact of keratinized mucosa on implant health, most evidence supports its benefits in superior plaque control and maintenance.16 Patients should be asked about their comfort during oral hygiene practices to ensure regular home care around their implant sites. While techniques to augment this band of attached tissue have evolved with the introduction of various allograft and xenograft materials, autografts have demonstrated more predictable clinical outcomes over time.2,23 Free soft tissue autografts can appreciably increase the width and thickness of keratinized mucosa,18 while connective tissue grafts serve to thicken periodontal phenotypes and improve esthetics, especially for immediate implant protocols.26
When identified early and presented as part of the treatment plan, implant soft tissue deficiencies can be managed successfully.
REFERENCES
- Araujo MG, Lindhe J. Peri‐implant health. J Periodontol. 2018;89:S249–S256.
- Wu Q, Qu Y, Gong P, Wang T, Gong T, Man Y. Evaluation of the efficacy of keratinized mucosa augmentation techniques around dental implants: A systematic review. J Prosthet Dent. 2015;113:383–390.
- Chen S, Buser D. Esthetic outcomes following immediate and early implant placement in the anterior maxilla: a systematic review. Int J Oral Maxillofac Implants. 2014;29(Supp):186–215.
- Berglundh T, Gislason Ö, Lekholm U, Sennerby L, Lindhe J. Histopathological observations of human periimplantitis lesions. J Clin Periodontol. 2004;31:341–347.
- Salvi GE, Bosshardt DD, Lang NP, et al. Temporal sequence of hard and soft tissue healing around titanium dental implants. Periodontol 2000. 2015;68:135–152.
- Chang M, Wennström JL, Odman P, Andersson B. Implant supported single-tooth replacements compared to contralateral natural teeth. Crown and soft tissue dimensions. Clin Oral Impl Res. 1999;10:185–194.
- Gerber JA, Tan WC, Balmer TE, Salvi GE, Lang NP. Bleeding on probing and pocket probing depth in relation to probing pressure and mucosal health around oral implants. Clin Oral Impl Res. 2009;20:75–78.
- Hämmerle C, Tarnow D. The etiology of hard- and soft-tissue deficiencies at dental implants: A narrative review. J Periodontol. 2018;89(Suppl):S291–S303.
- Perussolo J, Souza AB, Matarazzo F, Oliveira RP, Araújo MG. Influence of the keratinized mucosa on the stability of peri‐implant tissues and brushing discomfort: a 4‐year follow‐up study. Clin Oral Impl Res. 2018;29:1177–1185.
- Lin GH, Chan HL, Wang HL. The significance of keratinized mucosa on implant health: a systematic review. J Periodontol. 2013;84:1755–1767.
- Boynuegri D, Nemli SK, Kasko YA. Significance of keratinized mucosa around dental implants: a prospective comparative study. Clin Oral Impl Res. 2013;24:928–933.
- Bonino F, Steffensen B, Natto Z, Hur Y, Holtzman LP, Weber HP. Prospective study of the impact of peri‐implant soft tissue properties on patient–reported and clinically assessed outcomes. J Periodontol. 2018;89:1025–1032.
- Grischke J, Karch A, Wenzlaff A, Foitzik MM, Stiesch M, Eberhard J. Keratinized mucosa width is associated with severity of peri‐implant mucositis. A cross‐sectional study. Clin Oral Impl Res. 2019;30:457–465.
- Lim HC, Wiedemeier DB, Hämmerle CH, Thoma DS. The amount of keratinized mucosa may not influence peri‐implant health in compliant patients: A retrospective 5‐year analysis. J Clin Periodontol. 2019;46:354–362.
- Chung DM, Oh TJ, Shotwell JL, Misch CE, Wang HL. Significance of keratinized mucosa in maintenance of dental implants with different surfaces. J Periodontol. 2006;77:1410–1420.
- Roccuzzo M, Grasso G, Dalmasso P. Keratinized mucosa around implants in partially edentulous posterior mandible: 10–year results of a prospective comparative study. Clin Oral Impl Res. 2016;27:491–496.
- Schwarz F, Derks J, Monje A, Wang HL. Peri‐implantitis. J Clin Periodontol. 2018;45(Suppl):S246–S266.
- Thoma D, Lim H, Paeng K, et al. Augmentation of keratinized tissue at tooth and implant sites by using autogenous grafts and collagen-based soft-tissue substitutes. J Clin Periodontol. 2020;47:64–71.
- Halperin‐Sternfeld M, Zigdon‐Giladi H, Machtei EE. The association between shallow vestibular depth and peri‐implant parameters: a retrospective 6 years longitudinal study. J Clin Periodontol. 2016;43:305–310.
- Poskevicius L, Sidlauskas A, Galindo‐Moreno P, Juodzbalys G. Dimensional soft tissue changes following soft tissue grafting in conjunction with implant placement or around present dental implants: a systematic review. Clin Oral Impl Res. 2017;28:1–8.
- Park JB. Increasing the width of keratinized mucosa around endosseous implant using acellular dermal matrix allograft. Implant Dent. 2006;15:275–281.
- Lorenzo R, García V, Orsini M, Martin C, Sanz M. Clinical efficacy of a xenogeneic collagen matrix in augmenting keratinized mucosa around implants: a randomized controlled prospective clinical trial. Clin Oral Impl Res. 2012;23:316–324.
- Cairo F, Barbato L, Tonelli P, Batalocco G, Pagavino G, Nieri M. Xenogeneic collagen matrix versus connective tissue graft for buccal soft tissue augmentation at implant site. A randomized, controlled clinical trial. J Clin Periodontol. 2017;44:769–776.
- Linkevicius T, Puisys A, Steigmann M, Vindasiute E, Linkeviciene L. Influence of vertical soft tissue thickness on crestal bone changes around implants with platform switching: a comparative clinical study. Clin Implant Dent Relat Res. 2015;17:1228–1236.
- Bianchi AE, Sanfilippo F. Single-tooth replacement by immediate implant and connective tissue graft: a 1–9-year clinical evaluation. Clin Oral Impl Res. 2004;15:269–277.
- Chambrone L, Pini Prato G. Clinical insights about the evolution of root coverage procedures: the flap, the graft, and the surgery. J Periodontol. 2019;90:9–15.
- Kan J, Rungcharassaeng K, Morimoto T, Lozada J. Facial gingival tissue stability after connective tissue graft with single immediate tooth replacement in the esthetic zone: consecutive case report. J Oral Maxillofac Surg. 2009;67(Suppl 11):40–48.
From Decisions in Dentistry. September 2020;6(8):40-44.