 ALEX-MIT/ISTOCK/GETTY IMAGES PLUS
ALEX-MIT/ISTOCK/GETTY IMAGES PLUS
Evolving Management of Peri-Implant Disease
New thinking in one of the driving etiologies for peri-implantitis — metallosis — is bringing about a profound shift in management strategies for peri-implant disease.
There has been a recent and profound shift in the management of peri-implant disease. Many of the materials and techniques previously used to debride and regenerate intrabony defects have proven harmful to the implant surface.1 In large part, this is due to new thinking in one of the driving etiologies for peri-implant disease — metallosis. This phenomenon was adopted from histopathological findings in prosthetic joint complications and describes a pathogenic process that is initiated by the destructive sequela of the oral biofilm. A secondary inflammatory response results from the titanium particle and ion release of the dioxide layer around implants.
Implant insertion, corrosion, fretting, and a combination of these factors called tribocorrosion contribute to metallosis.1 Certain chemical agents can also damage wound-healing cells and stifle their regenerative potential.2 Therefore, learning to readily identify and treat peri-implant disease can improve long-term treatment success and health.
It is important to first understand the unique progression and presentation of peri-implant disease in the context of today’s literature. Peri-implant mucositis is an inflammatory mucosal lesion without supporting bone loss. Bleeding on probing is the hallmark sign, while suppuration, swelling and erythema may also appear as early as four months post-loading.3 If peri-implant mucositis is left untreated, it can progress to peri-implantitis given local, systemic and/or environmental risk factors. Peri-implantitis has clinical and radiographic signs of bone loss that include bleeding on probing, probing depths more than 6 mm, and/or suppuration. These histologic lesions are twice the size of those seen in periodontitis. The pattern of bone loss is often rapid and nonlinear, with significantly increased proportions of pro-inflammatory cytokines, neutrophils, neuropeptides and macrophages.4
Furthermore, the peri-implant microbiome is unique. Titanium readily forms a thin dioxide layer when inserted into the alveolar bone, which contributes to its biocompatibility and passivation. The electrostatic forces and ionic bonding differ between tooth- and implant-bound biofilms because this titanium dioxide alters bacterial adhesion and colonization during biofilm maturation. The alteration is such that the resulting biofilm has proven resistant to beta-lactam antibiotics normally effective against periodontal niches as a monotherapy.5 Additionally, 16S rRNA gene sequencing of healthy and diseased periodontal and peri-implant sites found that a vast majority of individuals shared less than 8% of species between teeth and implants.6
Titanium dissolution can destabilize the peri-implant biofilm, making way for more virulent, opportunistic bacteria and dysbiosis.5 Implants cannot fully reform their dioxide layer once it is lost, as was previously thought, and because no metal or alloy is ever completely inert, particles are readily released into the surrounding tissues from function and inflammatory attacks. More specifically, particle release may result due to friction from implant installation, micro-movements from abutments, or wear from implant debridement.7 Therefore, treatment modalities must meet the new model of peri-implant infection to safeguard the integrity of the implant body and dioxide layer.
EVOLVING TREATMENT TRENDS
The 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions echoed many of these concerns and, in fact, recognized peri-implant disease as its own entity. It is neither the same as periodontitis, nor should it be treated as such. Only in recent years has the approach to managing peri-implant disease changed.4
Many antimicrobial and mechanical treatments that successfully treated periodontitis were first employed against peri-implantitis. For example, osseous resective surgery was adapted from teeth to implants. In a randomized clinical trial, patients received resective surgery with or without recontouring of the implant surface (i.e., implantoplasty). Survival rates ranged 87.5% to 100% after three years, with significantly greater reduction in probing depths and attachment levels in the implantoplasty group from baseline.8 With current thinking, implantoplasty must be used judiciously to prevent metallosis and overheating. In a recent retrospective study, implantoplasty was performed as an adjunct to resective and reconstructive surgeries after nonsurgical debridement, with an implant survival rate of 97.8% reported after 24 months. However, only 66% of total cases had complete disease resolution with no residual pocketing or progressive bone loss.9
Several regenerative approaches later grew in popularity as new materials came to market. A prospective study grafted intrabony defects around ailing implants with deproteinized bovine bone mineral containing 10% collagen and found that probing depths were stable (less than 5 mm) with supportive maintenance at the 10-year follow-up. However, 15 of the original 26 patients required further antibiotic or surgical intervention or implant removal.10 A more aggressive protocol, respectfully dubbed the “kitchen sink,” included flap access; debridement of the implant surface with titanium scalers; a timed, six-step decontamination protocol with sodium bicarbonate air abrasives, saline, tetracycline, and 0.12% chlorhexidine gluconate; bovine or solvent-dehydrated bone grafting with enamel- or platelet-derived growth factors; connective tissue grafting or resorbable collagen membrane placement; and coronal flap advancement. After mean follow-up at four years, probing depth reduction of 5.1 mm and bone gain of 1.8 mm were noted. Nevertheless, 15% of implants required further surgery.11
While the above studies had limited, widespread therapeutic success, it is now understood these materials and techniques damage the implant surface and wound-healing cells.1 Osseous resective surgery and implantoplasty strip the implant’s protective dioxide layer, leading to dysbiosis and chronic inflammation.5 Ultrasonic scalers formerly used during debridement also release titanium particles into the mucosa and damage implant surfaces.12 Chemical agents (such as hydrogen peroxide, citric acid, antibiotics and sodium fluoride) applied to implants during regenerative procedures cause severe discoloration, pitting and surface corrosion from mechanical friction.13 In fact, no chemotherapeutic has been found superior to normal saline.2 Additionally, 0.12% chlorhexidine exhibited cytotoxic effects on osteoblasts and fibroblasts when used for implant decontamination. Nylon brushes and waterjets significantly reduced the bacterial load and best maintained corrosion resistance.14 It is clear that in the treatment of peri-implantitis today, less is actually more.
A targeted, anti-infective surgical therapy should therefore be considered. In a prospective study, patients received nonsurgical and surgical debridement with titanium scalers and saline. An amoxicillin/metronidazole regimen was prescribed during the perioperative period. Though 100% of implants had probing depths less than 5 mm without bleeding and progressive bone loss after the first year, 63% of implants met these criteria at five years.15 Regardless of the approach, surgical flap access exposes the implant to the oral cavity and potentially initiates corrosion. Beta-lactam antibiotics are also less effective against implant-bound biofilms.5 Laser irradiation and videoscope-assisted surgery can minimize trauma and surface damage, but requires further investigation to establish superiority.16,17
CURRENT GUIDELINES FOR IMPLANT MAINTENANCE
Implant maintenance should be tailored to each patient given his or her dental and medical histories. For example, patients with a history of periodontitis should maintain a three-month recall regimen, as they are more susceptible to peri-implantitis. Otherwise healthy patients should be seen every six months at the very least.18 At these appointments, clinicians are encouraged to evaluate the implant and prosthesis. Sites should be visualized, probed and palpated for signs of infection, such as pocketing, bleeding or suppuration. The presence of any calculus, cement or occlusal interferences should be assessed and removed. Radiographs should be taken yearly or if tissue conditions deteriorate.19 Patients should be forewarned of any failing implants that may require removal so they can make informed decisions about future replacement.
Self-care regimens have also evolved with new evidence. As floss remnants were increasingly found in peri-implant sulci, a preclinical study was developed; it showed that misfit implant-abutment connections and exposed implant threads had significantly greater floss residue at the implant collar.20 Waterjets removed supra- and subgingival plaque and improved tissue indices better than floss in short-term findings.21 Patients with full-arch, implant-borne restorations should be advised how to clean their prosthesis twice daily with different hygiene aids. Fixed appliances should be removed, cleaned extraorally, and reinserted with new prosthetic screws at the actual maintenance visit (when deemed necessary to do so).
Clinicians should use hygiene instruments compatible with the specific implants, abutments and prostheses.18 After proper isolation, use of low-abrasion subgingival air polishers with glycine powder can remove the superficial biofilm. Titanium scalers with Rockwell C hardness of 28 to 31 or titanium piezo tips are then used for further debridement and to prevent scratching of the implant itself. Restorations are polished with prophy paste before applying an antimicrobial varnish in the mucosal seal.22 Use of a time-release varnish containing 1% chlorhexidine diacetate and 1% thymol has been shown to slow bacterial leakage from the implant-abutment interface in vitro.23 This protocol can be adapted to treat peri-implant mucositis. In fact, a randomized controlled trial demonstrated that such varnish significantly reduced pocketing and bleeding around peri-implant mucositis lesions when used as part of a daily self-care regimen.24
CASE REPORT
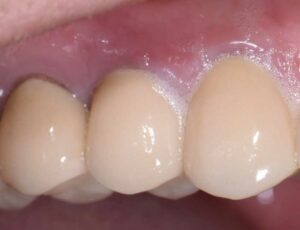
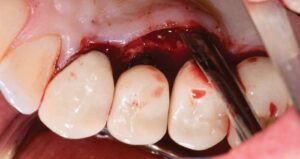
The following case outlines the steps taken to treat peri-implantitis after five-years post-loading. A 40-year old female presented to the graduate periodontics clinic at the University of Texas Health Science Center, San Antonio, School of Dentistry reporting increasing discomfort, bleeding and tenderness around implant #5 (Figure 1 through Figure 8). The patient had a medical history significant for depression and took sertraline. She reported smoking less than 10 cigarettes daily.
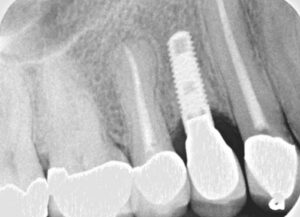
The patient’s periodontal tissues were generally pink and healthy, with mild plaque, calculus and debris throughout. The peri-implant tissues around implant #5 were inflamed, with localized vertical intrabony defects. Probing depths ranged from 6 to 8 mm, with moderate bleeding on probing. She reported frequent food impaction between #5 and 6, where an open contact was present. According to the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions, she had peri-implantitis.4
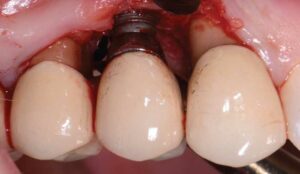
After a thorough patient evaluation and review of treatment options, a surgical plan was developed. The procedure would be performed using IV conscious sedation. The treatment plan included implant flap debridement, with osseous recontouring or bone grafting. This decision would be made after defect visualization at the time of surgery. The patient was informed that she would need a new crown/abutment at site #5 and/or 6 to close the contact and address any esthetic deficiencies after treatment. Tobacco cessation was also advised.
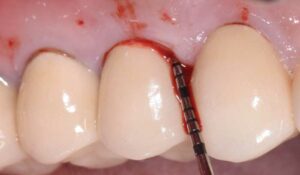
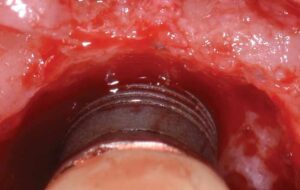
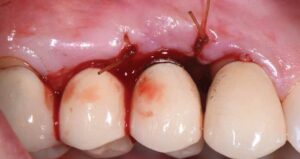
During surgery, full-thickness flaps were elevated to allow for visualization of the intrabony defects. After degranulation with titanium scalers and saline, the defect was not favorable to grafting because it was too broad and shallow. Osseous resective surgery was instead performed to achieve flat, bony architecture around implant #5. Flaps were apically repositioned with resorbable sutures.
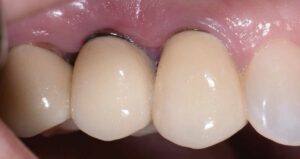
After several months of healing, the site was clinically stable, and the patient returned to her restorative dentist for fabrication of a new implant abutment and crown at site #5 and placed on six-month recalls. She was encouraged to use a waterjet around her implant restoration, as there was now a hygienic cleansable space.
SUMMARY AND CONCLUSIONS
Changes in scientific understanding of the pathogenesis of peri-implant disease now dictate how ailing implants should be managed and maintained. It is increasingly important to separate the diagnosis and treatment of periodontal and peri-implant conditions. Current nonsurgical and surgical interventions aim to preserve the titanium dioxide layer using instruments and materials compatible with the implant body, abutment and prosthesis. Maintenance visits and self-care regimens should be customized to meet site-specific and individual needs for long-term health and patient satisfaction.
Key Takeaways
- There has been a profound shift in the management of peri-implant disease, as evidence now suggests that many of the materials and techniques previously used to debride and regenerate intrabony defects are harmful to the implant surface.1
- To a large degree, this paradigm shift stems from new thinking in one of the driving etiologies for peri-implant disease, metallosis, which is a pathogenic process initiated by the destructive sequela of the oral biofilm associated with dental implant maintenance.
- In addition, a secondary inflammatory response results from the titanium particle and ion release of the dioxide layer around implants.
- Electrostatic forces and ionic bonding differ between tooth- and implant-bound biofilms because this titanium dioxide alters bacterial adhesion and colonization during biofilm maturation.
- Titanium dissolution can destabilize the peri-implant biofilm, making way for more virulent, opportunistic bacteria and dysbiosis.5
- In addition, titanium particles are readily released into the surrounding tissues due to friction from implant installation, micro-movements from abutments, and wear from implant debridement.7
- Ultimately, today’s treatment modalities must meet the new model of peri-implant infection to safeguard the integrity of the implant body and dioxide layer.
References
- Wilson TG Jr. Bone loss around implants — is it metallosis? J Periodontol. 2021;92:181–185.
- Berglundh T, Jepsen S, Stadlinger B, Terheyden H. Peri-implantitis and its prevention. Clin Oral Implants Res. 2019;30:150–155.
- Heitz-Mayfield LJ, Salvi GE. Peri-implant mucositis. J Clin Periodontol. 2018;45:S237–S245.
- Schwarz F, Derks J, Monje A, Wang H-L. Peri-implantitis. J Clin Periodontol. 2018;45(Suppl 20):S246–S266.
- Kotsakis GA, Olmedo DG. Peri-implantitis is not periodontitis: Scientific discoveries shed light on microbiome-biomaterial interactions that may determine disease phenotype. Periodontol 2000. 2021;86:231–240.
- Dabdoub SM, Tsigarida AA, Kumar PS. Patient-specific analysis of periodontal and peri-implant microbiomes. J Dent Res. 2013;92(Suppl 12):168S–175S.
- Berbel LO, Banczek ED, Karoussis IK, Kotsakis GA, Costa I. Correction: Determinants of corrosion resistance of Ti-6Al-4V alloy dental implants in an in vitro model of peri-implant inflammation. PLoS One. 2019;14:e0217671.
- Romeo E, Ghisolfi M, Murgolo N, Chiapasco M, Lops D, Vogel G. Therapy of peri-implantitis with resective surgery. Clin Oral Implants Res. 2005;16:9–18.
- Monje A, Pons R, Amerio E, Wang H-L, Nart J. Resolution of peri-implantitis by means of implantoplasty as adjunct to surgical therapy: A retrospective study. J Periodontol. April 26, 2021. Epub ahead of print.
- Roccuzzo M, Fierravanti L, Pittoni D, Dalmasso P, Roccuzzo A. Implant survival after surgical treatment of peri-implantitis lesions by means of deproteinized bovine bone mineral with 10% collagen: 10-year results from a prospective study. Clin Oral Implants Res. 2020;31:768–776.
- Froum SJ, Froum SH, Rosen PS. A regenerative approach to the successful treatment of peri-implantitis: A consecutive series of 170 implants in 100 patients with 2- to 10-year follow-up. Int J Periodontics Restorative Dent. 2015;35:857–863.
- Harrel SK, Wilson TG Jr, Pandya M, Diekwisch TG. Titanium particles generated during ultrasonic scaling of implants. J Periodontol. 2019;90:241– 246.
- Wheelis SE, Gindri IM, Valderrama P, Wilson TG Jr, Huang J, Rodrigues DC. Effects of decontamination solutions on the surface of titanium: investigation of surface morphology, composition, and roughness. Clin Oral Implants Res. 2016;27:329–340.
- Kotsakis GA, Black R, Kum J, et al. Effect of implant cleaning on titanium particle dissolution and cytocompatibility. J Periodontol. 2021;92:580–591.
- Heitz-Mayfield LJ, Salvi GE, Mombelli A, et al. Supportive peri-implant therapy following anti-infective surgical peri-implantitis treatment: 5-year survival and success. Clin Oral Implants Res. 2018;29:1–6.
- Natto ZS, Aladmawy M, Levi PA Jr, Wang HL. Comparison of the efficacy of different types of lasers for the treatment of peri-implantitis: a systematic review. Int J Oral Maxillofac Implants. 2015;30:338–345.
- Harrel SK. Videoscope-assisted minimally invasive surgery (VMIS) for bone regeneration around teeth and implants: a literature review and technique update. Dent J (Basel). 2018;6:30.
- Bidra AS, Daubert DM, Garcia LT, et al. Clinical practice guidelines for recall and maintenance of patients with tooth-borne and implant-borne dental restorations. J Prosthodont. 2016;25(Suppl 1):S32–S40.
- Mombelli A. Maintenance therapy for teeth and implants. Periodontol 2000. 2019;79:190–199.
- Montevecchi M, Valeriani L, Franchi L, Sforza NM, Piana G. Evaluation of floss remnants after implant flossing in three different implant conditions: a preclinical study. Int J Oral Maxillofac Implants. 2021;36:569–573.
- Magnuson B, Harsono M, Stark PC, Lyle D, Kugel G, Perry R. Comparison of the effect of two interdental cleaning devices around implants on the reduction of bleeding: a 30-day randomized clinical trial. Compend Contin Educ Dent. 2013;34:2–7.
- Wingrove SS. Safe, effective, in-office implant maintenance. In: Peri-Implant Therapy for the Dental Hygienist. Hoboken, NJ: Wiley-Blackwell; 2013.
- Besimo CE, Guindy JS, Lewetag D, Meyer J. Prevention of bacterial leakage into and from prefabricated screw-retained crowns on implants in vitro. Int J Oral Maxillofac Implants. 1999;14:654–660.
- Hallström H, Lindgren S, Twetman S. Effect of a chlorhexidine-containing brush-on gel on peri-implant mucositis. Int J Dent Hyg. 2017;15:149–153.
From Decisions in Dentistry. December 2021;7(11):16-18,21.


