
Esthetic Anodization of Orthodontic Archwires
Helpful for identification and patient-customization purposes, this technique can alter the color of titanium molybdenum alloy through a simple electrochemical process.

Orthodontic archwires made from various alloys — each with different mechanical properties — allow alternative sequences of archwire usage during all phases of orthodontic treatment. Archwires made of beta titanium molybdenum alloy (TMA) were introduced for orthodontic use in 1979 by Goldberg and Burstone,1 and have been utilized due to their favorable characteristics. These include excellent formability, low stiffness, and efficient working range for tooth movement.2 Titanium and titanium alloys are naturally a grey metallic color. Due to the reactive nature of these metals, a clear titanium dioxide (TiO2) covering layer develops spontaneously when the material is exposed to oxygen.3,4 The TiO2 layer is only a few nanometers thick, but, under certain conditions, has the capability of increasing in dimension. When this TiO2 layer approaches wavelengths in the order of fractions of the wavelength of colored light, a phenomenon known as light interference effects will cause a change in color.5
This colorization, which can be clinically useful for identifying archwire types and sizes, as well as esthetic-customization purposes, results from interference between light reflecting off the surface of the clear oxide layer, and light passing through the oxide layer and reflecting off the underlying metal surface. Changing the thickness of the TiO2 in the nanometer range will change the color, but will not alter the alloy’s surface chemistry, biocompatibility or mechanical properties.6
In clinical practice, colored beta TMA archwires could be useful for esthetic purposes in a manner similar to the application of colored elastic ligature ties that are commonly used with fixed appliances. An electrochemical anodization process can be used to increase the TiO2 layer thickness and alter the color.6–9 For certain titanium alloys, such as beta TMA, this process is simple and can be performed with easily obtained materials. In this process, a direct-current electrical potential is passed through an electrolytic solution, with the archwire serving as the anode (or positive electrode) while it is submerged in the electrolyte. The electrolyte is usually an acidic solution, but can be composed of any solution that will allow electrons to flow through it.10,11 The color that results depends on the voltage potential between the anode and cathode,7,10 thus, a wide range of colors can be obtained. The anodization process is self-limiting and will cease once the TiO2 layer has reached the thickness that corresponds to the selected voltage.5,6
TECHNIQUE

- Prepare an electrolytic solution for anodization by filling a 500-ml glass vessel with 400 ml of distilled water and dissolving 1 gram of disodium trioxosilicate (e.g., TSP-90 heavy duty cleaner) into the water.
- Prepare a direct current (DC) power supply by selecting the voltage necessary to obtain the desired anodization color. As depicted by the sampling shown in Figure 1, various voltages will produce specific colors. Additional hues can be produced with smaller voltage changes.
- Place a 3×6-cm strip of aluminum cooking foil in the solution as the negative electrode (cathode), and connect to the power supply’s negative terminal.
- Attach the end of a beta TMA archwire (anode) to the power supply’s positive terminal and submerge the archwire into the electrolyte, being careful not to submerge the positive lead (Figure 1).
- Anodization will occur via current flowing through the circuit, with surface oxide film formation occurring within approximately 15 seconds. The process is complete when the current shown on the DC power supply drops close to a zero-ampere reading.
- Remove the archwire from the electrolyte and check for uniform color under white light conditions. Next, disconnect the connection, wash the specimen with deionized water and confirm the desired color (Figure 2, page 33).
- Caution: When using electricity, even relatively small voltages can induce electrical shock. Do not touch the terminal leads (positive and negative) together or allow either lead to come in contact with the electrolyte solution when the power supply is turned on. Wear insulating rubber gloves or equivalent protection. Insulation clamps are also advisable.

DISCUSSION
Colorization by electrochemical anodization of titanium alloy has been reported in the dental implant literature, and is commonly used as a means of identifying componentry.11,12 Similarly, archwire colorization can serve as means of identification, as a range of colors can be achieved through anodization. Figure 3 (page 33) shows a beta TMA archwire that
was anodized in segments, with only one segment at a time submerged in the electrolyte. Each segment was anodized at a different voltage to illustrate a sampling of possible colors. The thickness of the oxide film growth has been shown to be linearly proportional to the voltage applied, approximating to 1.5 nm/V.13 However, this thickness, and the resultant color, may vary slightly, depending on the composition of the electrolyte and titanium alloy used. Anodization has no effect on the elasticity or ductility of the archwire.14
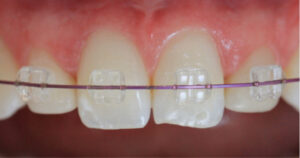
As this study demonstrates, anodization of beta TMA archwires could have applicability in orthodontic practice. Anodization coloring can be used to identify archwire types and sizes, as well as to allow patients to further customize their fixed appliances. The literature has reported a preference among adolescent patients for brightly colored elastic ligature ties, and it is logical to suggest this predilection would apply to archwires — although further investigation is needed to confirm this.15 Figure 4 shows a patient with a beta TMA archwire that has been anodized to alter its color to pink. The availability of colored archwires, in addition to colored elastic ligature ties, may facilitate active patient involvement in the treatment process, which, in turn, may enhance patient compliance.
SUMMARY
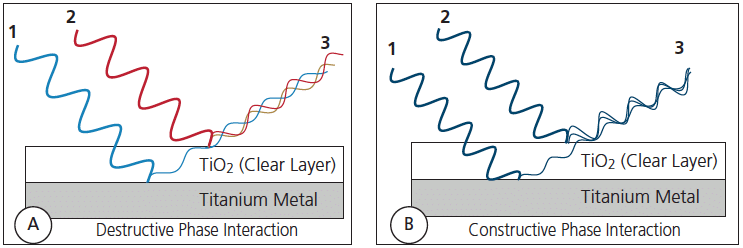
This anodization process is applicable to beta TMA archwires and may affect other alloys used in orthodontics, such as nickel-titanium or stainless steel. Thin film interference patterns are readily achieved and have a place in dental treatment. The ability of clear films to reflect interference patterns can result in destructive phase patterns, in which no one particular colored light interference pattern is produced, or enhance a particular color, depending on the specific film thickness of the clear TiO2 layer (Figures 5A and 5B).
Ultimately, this technique offers a simple, inexpensive method to alter the color of beta TMA archwires for esthetic and identification purposes. The ability to offer brightly colored archwires can potentially enhance patient involvement and cooperation during the treatment process.
KEY TAKEAWAYS
- Featuring excellent formability, low stiffness, and efficient working range for tooth movement,2 beta titanium molybdenum alloy (TMA) archwires are well suited for orthodontic treatment.
- Due to the nature of titanium and titanium alloys, a clear titanium dioxide (TiO2) covering layer develops when the material is exposed to oxygen.3,4 The TiO2 layer is only a few nanometers thick, but, under certain conditions, has the capability of increasing in dimension.
- An electrochemical anodization process can be used to increase the TiO2 layer thickness and alter the color.6–9
- Although changing the thickness of the TiO2 in the nanometer range will alter the color, it will not affect the alloy’s surface chemistry, biocompatibility or mechanical properties.6
- Colored beta TMA archwires could be useful for esthetic purposes in a manner similar to the application of colored elastic ligature ties that are commonly used with fixed appliances.
- Anodization coloring can be used to identify archwire types and sizes, as well as to allow patients to further customize their fixed appliances.
REFERENCES
- Goldberg J, Burstone CJ. An evaluation of beta titanium alloys for use in orthodontic appliances. J Dent Res. 1979;58:593–600.
- Kusy RP, Whitley JQ. Thermal and mechanical characteristics of stainless steel, titanium-molybdenum, and nickel-titanium archwires. Am J Orthod Dentofacial Orthop. 2007;131:229–237.
- Wang RR, Fenton A. Titanium for prosthodontic applications: a review of the literature. Quintessence Int. 1996;27:401–408.
- Nakajima H, Okabe T. Titanium in dentistry: development and research in the USA. Dent Mater J. 1996;15:77–90.
- Stavenga DG. Thin film and multilayer optics cause structural colors of many insects and birds. Mater Today Proceedings. 2014;1(Suppl):109–121.
- Jaeggi C, Kern P, Michler J, Zehnder T, Siegenthaler H. Anodic thin films of titanium used to mask for surface micro patterning of biomedical devices. Surf Coat Technol. 2005;200:1931–1919.
- Karim MA. Electro-Optical Displays. New York: Marcel Dekker Inc; 1992.
- Kern P, Schwalier P, Michler J. Electrolytic deposition of titania films as interference coatings on biomedical implants: microstructure, chemistry and nano-mechanical properties. Thin Solid Films. 2006:494;279–286.
- Paschalis EI, Chodosh J, Spurr-Michaud S, et al. In vitro and in vivo assessment of titanium surface modification for coloring the backplate of the Boston keratoprosthesis. Invest Ophthalmol Vis Sci. 2013;54:3863–3873.
- Bressan E, Paniz G, Lops D, Corazza B, Romeo E, Favero G. Influence of abutment material on the gingival color of implant-supported all-ceramic restorations: a prospective multicenter study. Clin Oral Implants Res. 2011;22:631–637.
- Wadhwani CP, O’Brien R, Kattadiyil MT, Chung KH. A laboratory technique for coloring titanium abutments to improve esthetics. J Prosthet Dent. 2016;115:409–411.
- Lin WS, Harris BT, Zandinejad A, Morton D. Use of digital data acquisition and CAD/CAM technology for the fabrication of a fixed complete dental prosthesis on dental implants. J Prosthet Dent 2014;111:1–5.
- Brunette DM, Tengvall P Textor M, Thomsen P. Titanium in Medicine. Berlin: Springer-Verlag Berlin Hedelberg; 2001:249.
- Yang CL, Chen FL, Chen SW. Anodization of the dental arch wires. Mats Chem Phys. 2006;100:268–274.
- Elekdag-Turk S, Ozkalayci N, Isci D, Turk T. Color preferences of patients receiving elastic ligatures. Eur J Dent. 2010;4:171–174.
The authors have no commercial conflicts of interest to disclose.
From Decisions in Dentistry. January 2018;4(1):32-34.


