 TOP PHOTO CORPORATION/TOP PHOTO GROUP/GETTY IMAGES PLUS
TOP PHOTO CORPORATION/TOP PHOTO GROUP/GETTY IMAGES PLUS
New Approach in Endodontics: Cryotherapy for Vital Pulp Treatment
Vital pulp cryotherapy represents a new way to reduce inflammation and postoperative pain in endodontic care.
Cryotherapy is derived from the Greek word kr´yos, meaning “cold.”1 The goal of cryotherapy is to remove heat by reducing the temperature of the tissue.2 The application of cold to human tissue decreases nerve conduction velocity and reduces hemorrhaging, edema and inflammation.3 In addition, cryotherapy has been shown to reduce the number of leukocytes adhering to the endothelial wall of capillaries; thus, fewer of these types of cells migrate to the affected tissue. In turn, this reduces endothelial dysfunction, resulting in a further reduction in inflammation.4 Another benefit is that cryotherapy can diminish the clinical need for pharmacologic intervention via narcotics or local anesthesia.5 In endodontic treatment, the integration of cryotherapy on pulpal tissue exposed (or indirectly exposed) due to a caries lesion, in conjunction with ethylenediaminetetraacetic acid (EDTA), bioceramics and restorative materials, has also been shown to reduce postoperative pain.
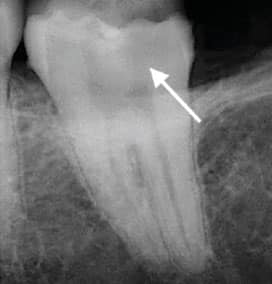
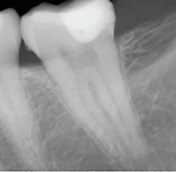
periapical radiograph of tooth #19
with arrow demarcating
radiolucent appearance of caries.
Prior to initiating vital pulp cryotherapy, a pretreatment pulpal and periradicular diagnosis is needed. The pretreatment pulpal and periradicular diagnosis begins by first reviewing the patient’s medical and dental history. This also should include taking a patient’s blood pressure, pulse and temperature (if indicated). Next, current preoperative radiographs — periapical, bitewing and cone beam computed tomography (as indicated) — should be taken (Figure 1).
After obtaining current radiographs, objective pulpal and periradicular clinical sensibility testing should be performed. Endodontic patients who present with a pretreatment pulpal diagnosis of a normal pulp, reversible pulpitis or symptomatic irreversible pulpitis — along with the possibility of a direct or indirect pulp exposure resulting from caries excavation — are good candidates for pulp capping or a partial pulpectomy.6,7 Pulps that have a pretreatment diagnosis of necrosis, or upon pulp chamber access in a multi-rooted tooth are determined to have partial pulpal necrosis, are contraindicated for vital pulp cryotherapy.
In regard to the pretreatment periradicular diagnosis, normal or symptomatic apical periodontitis — percussion and/or palpation sensitive, without radiographic evidence of a periradicular radiolucency (lesion) — are not contraindicated for vital pulp cryotherapy. Asymptomatic apical periodontitis (periapical radiolucency, without percussion/palpation sensitivity) or chronic (sinus tract present)/acute apical abscesses are contraindicated, however, because these are radiographic and clinical signs the pulpal tissue could be either necrotic or partially necrotic.
PRIOR TO CRYOTHERAPY
Lastly, prior to performing vital pulp cryotherapy, the treatment tooth must be restorable with a permanent restoration using composite or amalgam. Restoring the tooth posttreatment with a fabricated crown or provisional direct restoration and ultimately placing a permanent restoration at a subsequent appointment is contraindicated. The reason a permanent composite or amalgam restoration is placed is twofold. First, it is important to maintain a proper seal in order to prevent leakage from causing reoccurrence of pulpal inflammation.8 Second, if the tooth remains asymptomatic posttreatment, the provider does not risk causing an iatrogenic inflammatory response of the pulpal tissue by performing a crown preparation or replacing the provisional restoration with a permanent restoration at a subsequent appointment.9
After obtaining the proper pretreatment pulpal and periradicular diagnosis for vital pulp cryotherapy and confirming the tooth’s restorability (meeting the above criteria), the clinician can proceed with local anesthesia. After local anesthesia is administered and objective testing is performed on the treatment tooth to confirm pulpal anesthesia (no response to cold and/or electric pulp testing),10 a rubber dam is placed and the caries lesion is removed with a high-speed bur under water spray coolant. If the pulp is exposed or indirectly exposed as a result of removing all the caries, vital pulp cryotherapy is indicated.

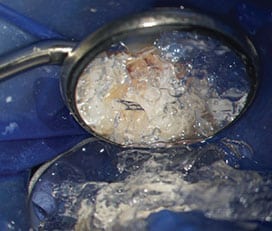
ice (0° C) prepared with an ice shaving device.
Shaved sterile-water ice (0° Celsius) is prepared and placed over the direct or indirect exposure of the pulpal tissue, along with the entire tooth (Figures 2A and 2B). After approximately 60 seconds, the ice will melt and should be removed with high-speed suction.
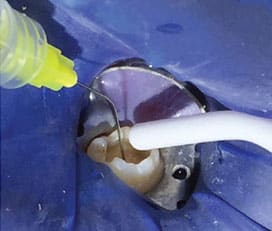
Following removal of the melted sterile ice, the exposed or indirect exposed pulp should be irrigated with 17% EDTA solution for 60 seconds (Figure 3).11,12 Although it has been stated in the literature to use sodium hypochlorite to control pulpal hemorrhage,13 it is important to note sodium hypochlorite should not be applied to a direct or indirect pulp exposure when performing a vital pulp cryotherapy procedure. The reasons are because cryotherapy is being used for hemostasis, and sodium hypochlorite has been shown to destroy dental pulpal stem cells and reduce the release of growth factor from the dentin.14 Clinicians should use 17% EDTA solution in place of sodium hypochlorite because it has been shown to release bioactive growth factors from the dentin.12 The conditioning of the dentin with EDTA will also promote the migration, adhesion and differentiation of dental pulp stem cells.12,14 In a report by Finnegan and Percival,15 EDTA was shown to have antimicrobial effects on gram-negative and gram-positive bacteria, yeasts and fungi. This agent also has been shown to induce antioxidants and anti-inflammatory activities.16
It is important to note that EDTA is an anticoagulant, and because it is a chelating agent, it can remove the calcium that plays an important role in blood clotting.15 In regard to its endodontic use in vital pulp cryotherapy, the authors have found that if employed after the use of cryotherapy, there appears to be no anticoagulant effect. It is speculated the hemostasis effect of cryotherapy blocks the effect of the chelating of the calcium by the EDTA solution. If a clinician observes reversal of pulpal hemostasis after EDTA application, he or she can reapply ice to the exposed pulp in order to obtain a new hemostatic effect.
A 2014 study by Ricucci et al17 found that a clinical pulpal diagnosis of a normal pulp or reversible pulpitis had a 96.6% histological match to the actual pulp tissue in a tooth, whereas a pulp diagnosis of symptomatic irreversible pulpitis had only an 84.4% histological correlation to the actual pulp tissue. This study noted that bacterial invasion of the pulp tissue and increased signs of inflammation were common in teeth diagnosed with symptomatic irreversible pulpitis, but absent in teeth diagnosed with normal pulps or reversible pulpitis. This research also demonstrated that when a caries lesion encroaches a pulp that is diagnosed as normal or having reversible pulpitis, only the coronal portion of the pulpal tissue (immediately adjacent to the caries) showed signs of inflammation — and not the entire pulp tissue as previously thought. Therefore, the clinician can treat the carious exposed or indirect exposed pulp tissue with a pulp cap procedure and will not need to perform a full or partial pulpectomy.
PULPAL BLEEDING DUE TO INFLAMMATION
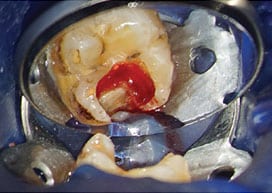
A study by Matsuo et al18 demonstrated that pulpal bleeding is a clinical marker for the extent of pulpal inflammation. This paper also showed the diameter of a pulpal exposure had no bearing on the success rate of the pulp capping or pulpotomy procedure. In pulps that have a pretreatment diagnosis of normal or reversible pulpitis, the hemorrhaging tends to be stagnated and lacks flow after caries removal (Figure 4); this is easily controlled with the application of ice.

In clinical cases that have a pretreatment pulpal diagnosis of symptomatic irreversible pulpitis and, clinically, the caries has entered the pulp chamber on a treatment tooth, the hemorrhaging tends to be greater due to an increase in pulpal inflammation (Figure 5).
Therefore, if the bleeding is profuse the clinician needs to perform a partial pulpectomy to remove the inflamed pulp tissue immediately under the carious dentinal exposure. This can be accomplished with a spoon excavator in order to remove the pulpal tissue while minimizing tissue trauma that can occur with a high-speed bur. After the partial pulpectomy, the shaved sterile ice should be placed until bleeding is controlled. A pulp that continues to bleed means one of two things. First, the clinician needs to remove more coronal pulpal tissue because the inflammation of the pulp has spread more apically. Second, the shaved sterile ice needs to be placed for a longer period for hemostasis to occur due to the advancement of the inflammation of the pulpal tissue.
If the bleeding is stagnated upon access to a tooth having a pretreatment pulpal diagnosis of symptomatic irreversible pulpitis, the pulpal diagnosis is considered clinically reversible pulpitis. This correlates with the Ricucci et al17 study that reported a 15.6% chance a tooth with a pretreatment pulpal diagnosis of symptomatic irreversible pulpitis is clinically reversible pulpitis.
BIOCERAMIC MATERIALS, GLASS IONOMER AND RESTORATIVE TREATMENT
After the exposed or indirect exposed pulp has been treated with shaved sterile ice and EDTA, it is covered with a bioceramic material (Figure 6).19 In cases of vital pulp cryotherapy on pulps with symptomatic irreversible pulpitits, it can be optional for a clinician to place a resorbable collagen matrix over the partial pulpectomy prior to the placement of the bioceramic material. After placing the bioceramic on a pulp cap or partial pulpectomy, a light-cured glass ionomer or non-light-cured glass ionomer material should be placed directly over the bioceramic pulp cap or partial pulpectomy.20 Following this step, a permanent restoration (composite or amalgam) is placed. After the rubber dam is removed and the patient’s occlusion checked, final periapical and bitewing radiographs should be taken (Figure 7).
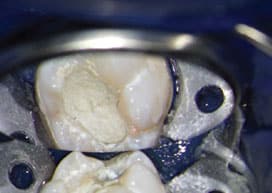
material over a carious pulp exposure and
entire pulpal floor on tooth #19 following the
application of shaved sterile-water ice and
ethylenediaminetetraacetic acid.
TWO-WEEK RECALL
The patient should be placed on a two-week recall. A study by Linu et al21 reported that all vital pulp therapy failures occurred in the first two weeks after treatment. The paper also noted that teeth remaining asymptomatic at the two-week failure point remained asymptomatic, vital and functional over the follow-up period of 12 to 18 months.
If the patient presents within the first two weeks (or any time after the first two-week period) following vital pulp cryotherapy (phase I treatment) with a symptomatic irreversible pulpitis that has either not resolved, arose as a result of treatment, or with a necrotic pulp, asymptomatic apical periodontitis, and/or a chronic/acute apical abscess, clinicians are advised to reevaluate the tooth for conventional endodontic therapy (phase II treatment).
placement of a permanent composite restoration.
SUMMARY
Using this new vital pulp cryotherapy technique on exposed or indirect exposed pulp tissue (by incorporating cryotherapy, EDTA, bioceramics, and a composite or amalgam permanent restoration) has been shown to reduce endodontic inflammation and postoperative pain. Further clinical studies are needed to observe the long-term prognosis of a pulp after undergoing vital pulp cryotherapy.
Acknowledgement: The authors wish to thank Doris Basali, DDS, Vikash Hullyar, DDS, and Gail Tischke, DDS, endodontic residents at the University of Illinois-Chicago College of Dentistry, for the clinical treatment and documentation of vital pulp cryotherapy procedures.
KEY TAKEAWAYS
- Cryotherapy, the application of cold to human tissue, decreases nerve conduction velocity and reduces hemorrhaging, edema, inflammation3 and postoperative pain.
- In endodontic treatment, cryotherapy can diminish the clinical need for pharmacologic intervention via narcotics or local anesthesia.5
- Prior to initiating vital pulp cryotherapy, a pretreatment pulpal and periradicular diagnosis is needed.
- In addition, vital pulp cryotherapy is only indicated for a treatment tooth that is restorable with a permanent restoration using composite or amalgam.
- After the exposed or indirect exposed pulp has been treated with shaved sterile-water ice and ethylenediaminetetraacetic acid, it is covered with a bioceramic material.19
- A light-cured glass ionomer or non-light-cured glass ionomer should be placed directly over the bioceramic pulp cap or partial pulpectomy.20
- Next, a permanent restoration (composite or amalgam) is placed and final periapical and bitewing radiographs are taken.
REFERENCES
- Vera J, Ochoa-Rivera J, Vazquez-Carcaño M, Romero M, Arias A, Sleiman P. Effect of intracanal cryotherapy on reducing root surface temperature. J Endod. 2015;41:1884–1887.
- Hubbard TJ, Denegar CR. Does cryotherapy improve outcomes with soft tissue injury? J Athl Train. 2004;39:278–279.
- Modabber A, Rana M, Ghassemi A, et al. Three-dimensional evaluation of postoperative swelling in treatment of zygomatic bone fractures using two different cooling therapy methods: a randomized, observer-blind, prospective study. Trials. 2013;14:1–10.
- Laureano Filho JR, de Oliverira e Silva ED, Batista CI, et al. The influence of cryotherapy on reduction of swelling, pain and trismus after third-molar extraction: a preliminary study. J Am Dent Assoc. 2005;136:774–778.
- Watkins AA, Johnson TV, Shrewsberry AB, et al. Ice packs reduce postoperative midline incision pain and narcotic use: a randomized controlled trial. J Am Coll Surg. 2014;219:511–517.
- Taha NA, Ahmad MB, Ghanim A. Assessment of mineral trioxide aggregate pulpotomy in mature permanent teeth with carious exposures. Int Endod J. 2017;50:117–125.
- Taha NA, Khazali M. Partial pulpotomy in mature permanent teeth with clinical signs indicative of irreversible pulpitis: a randomized clinical trial. J Endod. 2017;43:1417–1421.
- Awawdeh L, Al-Qudah A, Hamouri H, Chakra RJ. Outcomes of vital pulp therapy using mineral trioxide aggregate or biodentine: a prospective randomized clinical trial. J Endod. 2018;44:1603–1609.
- Kim S. Neurovascular interactions in the dental pulp in health and inflammation. J Endod. 1990; 16:48–55.
- Bahcall J, Xie Q. Clinically enhancing local anesthesia techniques for endodontic treatment. Compend Contin Educ Dent. 2017;38:80–84.
- Casagrande L, Demarco FF, Zhang Z, Araujo FB, Shi S, Nör JE. Dentin-derived BMP-2 and Odontoblast Differentiation. J Dent Res. 2010;89:603–608.
- Gallar KM, Buchalla W, Hiller KA, et al. Influence of root canal disinfectants on growth factor release from dentin. J Endod. 2015;41:363–368.
- Witherspoon DE. Vital pulp therapy with new materials: new directions and treatment perspectives–permanent teeth. J Endod. 2008;34(Suppl):S25–S28.
- Cao Y, Song M, Kim E, et al. Pulp-dentin regeneration: current state and future prospects. J Dent Res. 2015;94:1544–1551.
- Finnegan S, Percival S. EDTA: an antimicrobial and antibiofilm agent for use in wound care. Adv Wound Care. 2015;4:415–421.
- Gonzalez-Cuevas J, Navarro-Partida J, Marquez-Aquirre AL, et al. Ethylenediaminetetraacetic acid induces antioxidant and anti-inflammatory activities in experimental liver fibrosis. Redox Report. 2011;16:2,62–70.
- Ricucci D, Loghin S, Siqueira J. Correlation between clinical and histological pulp diagnosis. J Endod. 2014;40:1932–1939.
- Malsuo T, Nakanishi T, Shimizu H, Ebisu S. A clinical study of direct pulp capping applied to carious-exposed pulps. J Endod. 1996;22:551–556.
- Miller AA, Takimoto K, Wealleans J, Diogenes A. Effect of 3 bioceramic materials on stem cells of the apical papilla proliferation and differentiation using a dentin disk model. J Endod. 2018;4:599–603.
- Wang Z. Bioceramic materials in endodontics. Endod Topics. 2015;32:3–30.
- Linu S, Lekshmi MS, Varunkumar VS, Sam Joseph V. Treatment outcome following direct pulp capping using bioceramic materials in mature permanent teeth with carious exposure: a pilot retrospective study. J Endod. 2017;43:1635–1639.
The authors have no commercial conflicts of interest to disclose.
From Decisions in Dentistry. October 2019;5(9):14–18.


