
The Rheumatoid Arthritis/Periodontal Disease Connection
Rheumatoid arthritis and periodontal disease share similar inflammatory pathways and environmental mechanisms. This potential association has generated new thinking about possible links between these two common conditions.
The author has no commercial conflicts of interest to disclose.
This 2 credit hour self-study activity is electronically mediated.
Objectives
After reading this course, the participant should be able to:
- Discuss the natural history of rheumatoid arthritis and periodontitis.
- Recognize the similarities in inflammatory mechanisms between rheumatoid arthritis and periodontal disease.
- Describe the environmental factors that influence both rheumatoid arthritis and periodontal disease.
Not only do periodontitis and rheumatoid arthritis have similar patterns of natural history, but their disease manifestation is also controlled by similar inflammatory pathways.2 In addition, these chronic inflammatory conditions are associated with many other systemic and inflammatory disorders.3,4 Not surprisingly, the management of both periodontitis and rheumatoid arthritis includes treatment of the symptoms of the disease, modulation of the inflammatory response, and surgery.
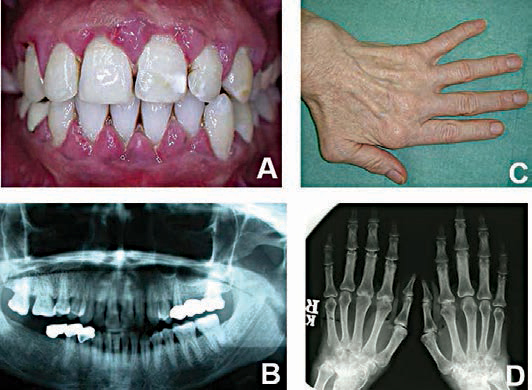
Reproduced with permission from: Bartold PM, Mariotti A. Association between periodontitis and rheumatoid arthritis. In: Periodontal Disease and Overall Health: A Clinician’s Guide. Genco RJ, Williams RC, eds. Yardley, Pennsylvania: Professional Audience Communications Inc.; 2010:180.
Manifesting as a wide spectrum of clinical entities, rheumatoid arthritis results from the combination of individual genetic susceptibility and environmental triggers. It occurs worldwide, affecting approximately 1% to 2% of the population in a female/male ratio of 3:1. Its peak onset is in the fourth and fifth decades of life.5 Men may be shielded by their hormonal composition and need a stronger genetic influence in order for the disease to develop.6 Infectious agents have been proposed to act as triggers for the development of rheumatoid arthritis, but, to date, no single organism has been identified to support this concept.7
Rheumatoid arthritis has a considerable impact on individuals’ quality of life, as well as their ability to work.8 Long duration with periods of disease remission are significant features of the natural history of this disease. Functional disability and radiologic evidence of damage to the joints are the most studied outcomes in rheumatoid arthritis. The majority of patients show radiographic evidence of joint damage within the first few years, with subsequent radiographs showing its progression over the next five to 10 years.6 Studies show that some individuals experience disease progression despite receiving the best available treatment.4
Periodontal diseases are a group of heterogeneous conditions, ranging from relatively benign gingivitis to the more destructive forms, such as chronic and aggressive periodontitis. Up to 95% of the population may experience gingivitis; as many as 50% have some form of chronic periodontitis; and between 5% to 15% develop advanced and aggressive periodontitis.9,10 Studies now indicate that the prevalence of severe periodontitis is confined to a small portion of the general population.9,10 If left untreated, gingivitis may progress to periodontitis and, ultimately, tooth loss.11 On the other hand, the disease may become self-contained and not progress — or may even enter into long periods of remission.12
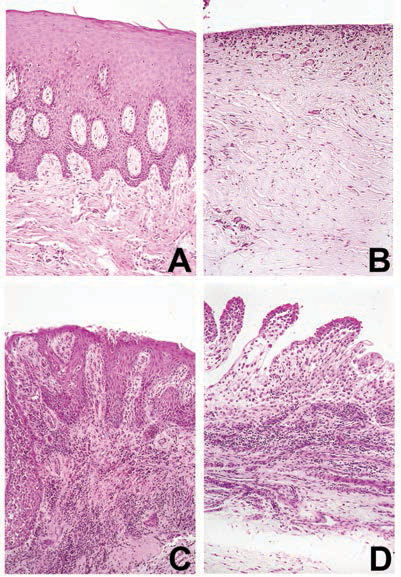
Just as with rheumatoid arthritis, some individuals continue to experience disease progression in the form of refractory periodontitis, despite the best available treatment.12 Whether the individuals with refractory forms of periodontitis are the same as those who experience uncontrollable rheumatoid arthritis remains to be established.
MECHANISMS OF TISSUE DESTRUCTION
There is no doubt about the importance of inflammation and the body’s immune cells in the pathogenesis of rheumatoid arthritis and periodontitis (Figure 2). The task now is to identify the specific molecular mediators of inflammation (cytokines) produced by the immune cells, and determine their effects on target cells and tissues.13,14 While the role of cytokines on normal cellular processes is important, it is their role in disease and tissue damage that has attracted the most attention. The unwanted effects of cytokines result from excessive production, poor control or inadequate inhibition of their biological activity. One of the most important actions of these inflammatory cytokines is to activate the molecular and cellular events associated with bone resorption.13,14
Patients with periodontitis have similar cytokine profiles to patients with rheumatoid arthritis.1 In both conditions, tissue destruction and disease progression involve the persistent presence of high levels of cytokines that induce inflammation (e.g., interleukin 1β and tumor necrosis factor-α), and low levels of cytokines that would normally suppress inflammation (e.g., interleukin–10 and transforming growth factor β).15 Furthermore, high levels of enzymes capable of degrading the tissues and low levels of inhibitors of these enzymes define the active stages of both rheumatoid arthritis and periodontitis (Table 1).
Because the primary mode of tissue destruction in these diseases is the exuberant host inflammatory response, host-modifying agents and genetically engineered chemicals that can target specific molecules involved in the inflammatory process are being developed. It is hoped these may play a role in restoring balance in the inflammatory mediators.16
ENVIRONMENTAL INFLUENCES
The outcome of both rheumatoid arthritis and periodontitis is influenced not only by genetic factors, but also by environmental factors.15,17 In the broadest context, environmental factors are those that modify or alter the host response. Thus, demographics, socioeconomic status, lifestyle, diet, hormonal and psychological variables may be considered environmental factors — each of which has the potential to influence the outcome of these two diseases (Table 2).
Age is an important variable for rheumatoid arthritis and periodontitis, with both diseases manifesting juvenile and adult forms. Interestingly, age seems to play more of a key role in rheumatoid arthritis than periodontitis.18,19 For example, patients who experience an onset of rheumatoid arthritis at age 30 or younger tend to have a higher incidence of entering spontaneous remission. On the other hand, patients with an onset at age 50 or older tend to have more active disease and become more disabled.18 The opposite seems to hold true for periodontitis — whereby the early-onset forms of the disease (younger than 30) tend to be more aggressive than the later adult-onset forms (older than 35).19 But, as with early onset rheumatoid arthritis, spontaneous remission has been noted in early onset forms of periodontitis.19
Women are more likely to develop rheumatoid arthritis than men.18 Although it is reported that periodontitis is more prevalent among men, to date, the evidence is insufficient to confirm that gender is a significant environmental variable for periodontitis.20
Socioeconomic status is a significant factor, influencing incidence and severity of both rheumatoid arthritis and periodontitis.21,22 While it is still not clear why this link with lower socioeconomic class exists, it might be that such individuals are less attentive in regard to their general health care. Alternatively, it is possible that low socioeconomic status leads to increased exposure to psychosocial stress which, in turn, exerts a physiologic toll that contributes to disparities in some health conditions.23
Smoking is another important environmental modifying factor for many conditions. Through its effects on the cardiovascular system, immune cell function and general tissue physiology, smoking is considered a risk factor for developing both rheumatoid arthritis and periodontitis.24 Furthermore, smoking is associated with the modification of protein structures in the lungs through a process known as protein citrullination, which may be a key factor in activating and exacerbating autoimmune responses and tissue destruction in rheumatoid arthritis.25
Stress and other psychological factors — such as depression and coping mechanisms — have been well documented as influencing inflammatory responses.26 These interrelationships are defined within the context of an emerging field known as psychoneuroimmunology — a multilevel theory that encompasses neural, physiological, molecular and genomic mechanisms to link experiences of social-environmental stress with internal biological processes that drive chronic diseases.27 Both rheumatoid arthritis and periodontitis are influenced by stress and psychological factors, therefore, psychoneuroimmunology has been proposed to play a role in both conditions.28,29
Hormonal factors have the potential to influence a wide range of physiological and pathological responses. Hormonal changes associated with diabetes and pregnancy are well documented and can influence a number of systemic conditions, including rheumatoid arthritis and periodontitis. Diabetes is a well-recognized modifying factor in both rheumatoid arthritis and periodontitis.30,31 Pregnancy can exacerbate periodontal inflammation,32 while the converse holds true for rheumatoid arthritis, as remission of rheumatoid arthritis has been noted among pregnant women.33
Diet can have both a protective and destructive effect on rheumatoid arthritis and periodontitis. Malnutrition, as well as vitamin and mineral deficiencies, can significantly affect normal and pathological tissue physiology.34 Obesity and metabolic syndrome may also be significant risk factors for the development of rheumatoid arthritis and periodontitis.35–38 A protective role for diet has also been proposed. Both theoretical and clinical trial evidence have shown that omega-3 fatty acids (most commonly found in fish oil) are natural anti-inflammatory agents, which can significantly influence the outcome and progression of inflammatory disorders.39 There is evidence to indicate that dietary supplementation with omega-3 fatty acids can have a beneficial effect on the outcome of rheumatoid arthritis.40 Initial studies are also showing promising results for the adjunctive use of omega-3 fatty acids for the management of periodontitis.41
CONCLUSION
It has long been recognized that periodontitis and rheumatoid arthritis share many common pathological features. Recent studies indicate that a strong relationship exists between disease severity and extent for individuals suffering from both conditions. While causality between the two diseases is unlikely, a number of possibilities exist and should be investigated.
Editor’s Note: Part two of this series, which will appear in a future issue, will expand on theories about the possible connection between rheumatoid arthritis and periodontitis.
REFERENCES
- Bartold PM, Marshall RI, Haynes DR. Periodontitis and rheumatoid arthritis: a review. J Periodontol. 2005;76(Suppl 11):2066–2074.
- de Pablo P, Chapple IL, Buckley CD, Dietrich T. Periodontitis in systemic rheumatic diseases. Nat Rev Rheumatol. 2009;5:218–224.
- Moreland LW, Curtis JR. Systemic nonarticular manifestations of rheumatoid arthritis: focus on inflammatory mechanisms. Semin Arthritis Rheum. 2009;39:132–143.
- Linden GJ, Lyons A, Scannapieco FA. Periodontal systemic associations: review of the evidence. J Periodontol. 2013;(Suppl 4):S8–S19.
- Aletaha D, Neogi T, Silman AJ, et al. 2010 Rheumatoid arthritis classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Ann Rheum Dis. 2010;69:1580–1588.
- Ollier WE, Harrison B, Symmons D. What is the natural history of rheumatoid arthritis? Best Pract Res Clin Rheumatol. 2001;15:27–48.
- Li s, Yu Y, Yue Y, Zhang Z, Su K. Microbial infection and rheumatoid arthritis. J Clin Cell Immunol. 2013;4:4–6.
- Looper KJ, Mustafa SS, Zelkowitz P, Purden M, Baron M. Work instability and financial loss in early inflammatory arthritis. Int J Rheum Dis. 2012;15:546–553.
- Albander JM, Rams TE. Global epidemiology of periodontal disease: an overview. Periodontol 2000. 2002;29:7–9.
- Eke PI, Dye BA, Wei L, Thornton-Evans GO, Genco RJ. Prevalence of periodontitis in adults in the United States: 2009 and 2010. J Dent Res. 2012;91:914–920.
- Kinane DF, Bartold PM. Clinical relevance of the host responses of periodontitis. Periodontol 2000. 2007;43:278–293.
- Armitage GC. Development of a classification system for periodontal diseases and conditions. Ann Periodontol. 1999;4:1–6.
- Souza PP, Lerner UH. The role of cytokines in inflammatory bone loss. Immunol Invest. 2013;42:555–622.
- Bartold PM, Cantley MD, Haynes DR. Mechanisms and control of pathologic bone loss in periodontitis. Periodontol 2000. 2010;53:55–69.
- Page RC, Offenbacher S, Schroeder HE, Seymour GJ, Kornman KS. Advances in the pathogenesis of periodontitis: summary of developments, clinical implications and future directions. Periodontol 2000. 1997;14:216–248.
- Gokhale SR, Padhye AM. Future prospects of systemic host modulatory agents in periodontal therapy. Br Dent J. 2013;214:467–471.
- Karlson EW, Deane K. Environmental and gene-environment interactions and risk of rheumatoid arthritis. Rheum Dis Clin North Am. 2012;38:405–426.
- Katchamart W, Johnson S, Lin HJ, Phumethum V, Salliot C, Bombardier C. Predictors for remission in rheumatoid arthritis patients: A systematic review. Arthritis Care Res. 2010;62:1128–1143.
- Page RC, Altman LC, Ebersole JL, et al. Rapidly progressive periodontitis. A distinct clinical condition. J Periodontol. 1983;54:197–209.
- Haytac MC, Ozcelik O, Mariotti A. Periodontal disease in men. Periodontol 2000. 2013;61:252–265.
- McBurney CA, Vina ER. Racial and ethnic disparities in rheumatoid arthritis. Curr Rheumatol Rep. 2012;14:463–471.
- Thornton-Evans G, Eke P, Wei L, et al. Periodontitis among adults aged <30 years — United States, 2009–2010. MMWR Surveill Summ. 2013;22;62(Suppl 3):129–135.
- Juster RP, McEwen BS, Lupien SJ. Allostatic load biomarkers of chronic stress and impact on health and cognition. Neurosci Biobehav Rev. 2010;35:2–16.
- Johannsen A, Susin C, Gustafsson A. Smoking and inflammation: evidence for a synergistic role in chronic disease. Periodontol 2000. 2014;64:111–126.
- Klareskog L, Malmström V, Lundberg K, Padyukov L, Alfredsson L. Smoking, citrullination and genetic variability in the immunopathogenesis of rheumatoid arthritis. Semin Immunol. 2011;23:92–98.
- Slavich GM, Irwin MR. From stress to inflammation and major depressive disorder: a social signal transduction theory of depression. Psychol Bull. 2014;140:774–815.
- Jaremka LM, Lindgren ME, Kiecolt-Glaser JK. Synergistic relationships among stress, depression, and troubled relationships: insights from psychoneuroimmunology. Depress Anxiety. 2013;30:288–296.
- McFarlane AC, Brooks PM. Psycho-immunology and rheumatoid arthritis: concepts and methodologies. Int J Psychiatry Med. 1990;20:307–322.
- Doyle CJ, Bartold PM. How does stress influence periodontitis? J Int Acad Periodontol. 2012;14:42–49.
- Wasko MC, Kay J, Hsia EC, Rahman MU. Diabetes mellitus and insulin resistance in patients with rheumatoid arthritis: risk reduction in a chronic inflammatory disease. Arthritis Care Res. 2011;63:512–521.
- Chee B, Park B, Bartold PM. Periodontitis and type II diabetes: a two-way relationship. Int J Evid Based Health. 2013;11:317–329.
- Figuero E, Carrillo-de-Albornoz A, Martin C, Tobias A, Herrera D. Effect of pregnancy on gingival inflammation in systemically healthy women: a systematic review. J Clin Periodontol. 2013;40:457–473.
- Barrett JH, Brennan P, Fiddler M, Silman A. Breast-feeding and postpartum relapse in women with rheumatoid and inflammatory arthritis. Arthritis Rheum. 2000;43:1010–1015.
- Ritchie CS, Kinane DF. Nutrition, inflammation, and periodontal disease. Nutrition. 2003;19:475–476.
- Abella V, Scotece M, Conde J, et al. Adipokines, metabolic syndrome and rheumatic diseases. J Immunol Res. 2014;2014:343746.
- Watanabe K, Cho YD. Periodontal disease and metabolic syndrome: a qualitative critical review of their association. Arch Oral Biol. 2014;59:855–870.
- Chaffee BW, Weston SJ. Association between chronic periodontal disease and obesity: a systematic review and meta-analysis. J Periodontol. 2010;81:1708–1724.
- Scrivo R, Vasile M, Müller-Ladner U, Neumann E, Valesini G. Rheumatic diseases and obesity: adipocytokines as potential comorbidity biomarkers for cardiovascular diseases. Mediators Inflamm. 2013;2013:808125.
- Calder PC. n-3 fatty acids, inflammation and immunity: new mechanisms to explain old actions. Proc Nutr Soc. 2013;72:326–336.
- Norling LV, Perretti M. The role of omega-3 derived resolvins in arthritis. Curr Opin Pharmacol. 2013;13:476–481.
- Elkhouli AM. The efficacy of host response modulation therapy (omega-3 plus low-dose aspirin) as an adjunctive treatment of chronic periodontitis (clinical and biochemical study). J Periodontal Res. 2011;46:261–268.





[…] more about the possible link between periodontal disease and RA at https://decisionsindentistry.com/article/the-rheumatoid-arthritis-periodontal-disease-connection/ and […]