AJA KOSKA/E+/GETTY IMAGES PLUS
AJA KOSKA/E+/GETTY IMAGES PLUS
Shared Decision-Making in Evidence-Based Dentistry
The use of shared decision-making during treatment planning can help build consensus between providers and patients
This course was published in the November 2020 issue and expires November 2023. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Describe traditional evidence-based logic models for treatment planning.
- Explain the concept and clinical implementation of the shared decision-making model, which is based on consensus between providers and patients.
- Discuss the role that patient decision aids play in shared decision-making.
Healthcare providers are constantly helping patients choose the best treatment option, based on the available evidence and clinician’s professional judgment. The traditional logic model used for evidence-based decision-making has four elements: (1) input of available evidence; (2) activities, including analyzing the best evidence available; (3) output, leading to the best clinical decision; and (4) impact of the treatment provided.1 Recently, this concept has evolved, as more patients become involved in the decision-making process. In medicine, some difficult decisions include chemo versus radiation therapies for cancer, or when to choose a hip replacement. In dentistry, complex situations also arise, such as deciding if a compromised tooth should be extracted — and, if so, when to opt for a dental implant.
Since there may be limited evidence available to fit each patient’s condition, evidence-based decision-making must conceptually evolve to shared decision-making (SDM), which involves the patient. While patient preference is an essential part of evidence-based dentistry, it can be challenging for authoritative clinicians to communicate open-ended information. With the goal of reaching informed consensus, SDM has gained traction in medicine,2 but it is still lacking adequate representation in dentistry.3
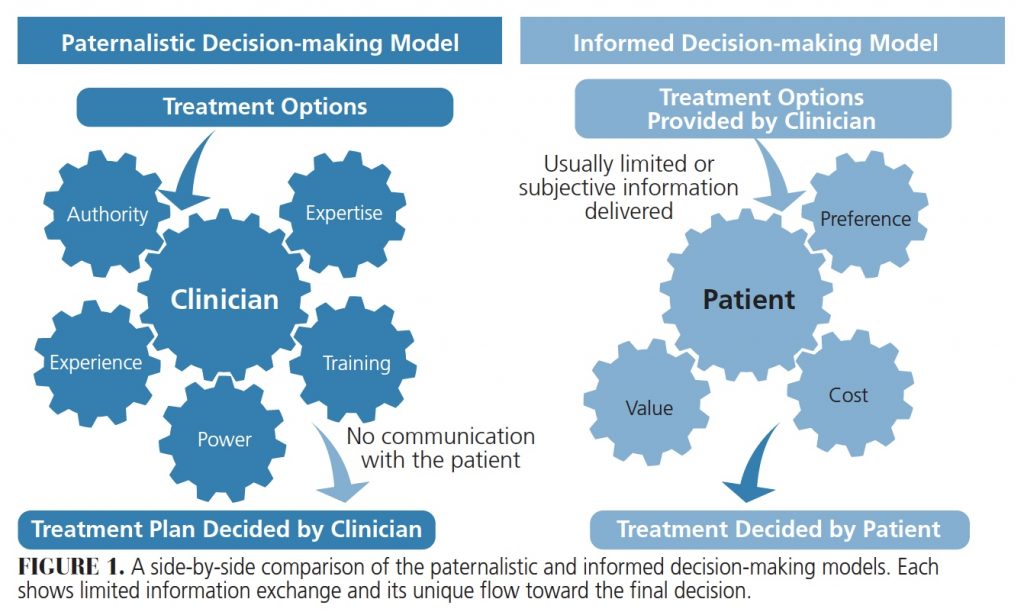
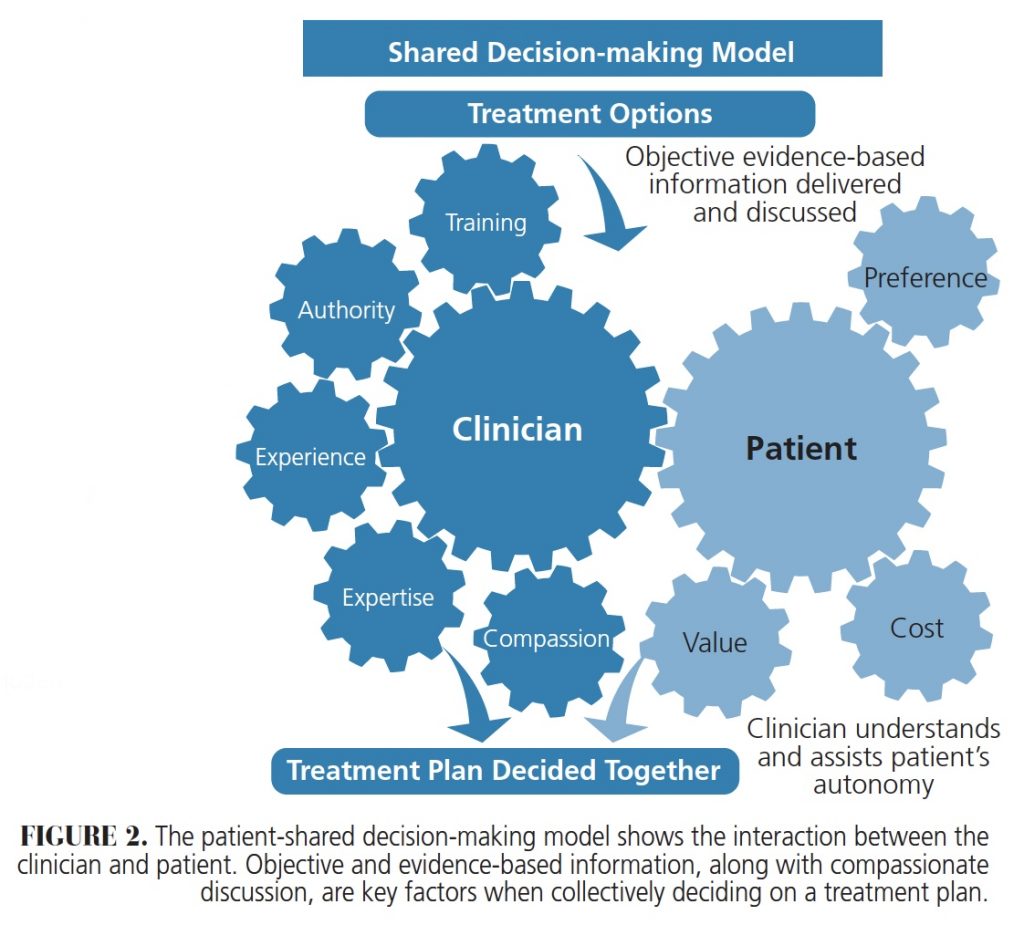
There are ethical considerations for SDM. As noted by Coulter and Collins,4 “It is any healthcare provider’s duty to enrich their patients with evidence-based information.” This patient-centered approach involves discussions between providers and patients to facilitate mutual agreement on treatment. The concept of SDM lies between two other decision-making models: paternalistic, where the clinician makes the decision alone; and informed, where the patient makes the treatment decision based on information provided by the clinician (Figure 1).5,6 The SDM process creates a hybrid of these two models by allowing the patient and provider to both have input (Figure 2).
BENEFITS AND CHALLENGES
This hybrid concept has many benefits. It allows patients to make decisions based on their situation, social circumstances and attitude toward risk, as well as their values and preferences. Similarly, SDM takes into account the clinician’s diagnosis, prognosis and treatment options.4 Among patients, this approach encourages a sense of control and overall satisfaction before, during and after treatment.7 It has also been found to reduce patient anxiety.8
Research indicates patients and clinicians agree that SDM improves the quality of healthcare.9 Saleh et al10 note that three-quarters of the physicians in their study practiced SDM by discussing the pros and cons of each treatment option, and encouraging patients to be involved in decisions. The same study reports that a large majority of patients are in favor of this decision-making model.10
In general, dentistry tends to make patients nervous and uncomfortable. In this respect, SDM could increase patient comfort, making for smoother procedures and a better overall experience. Despite its advantages, adopting SDM to clinical situations is not necessarily easy. This may be due to operators’ lack of knowledge and skills with the SDM model, time demands for implementation, and patients not always being willing to accept decisional responsibility.11
Since its introduction, SDM has focused on creating evidence-based patient decision aids (PDAs) that help translate the science behind each treatment option to a format that is easily understandable.12 Tables, flowcharts, videos and photos are examples of PDAs that help patients formulate an opinion about each option, and work with their practitioner to select the best overall treatment in terms of outcome, price, recovery, appearance, and longevity of the result. Additionally, since each patient is different, SDM still requires clinical judgment as to any risk factor or prognostic indicator pertinent to the patient and case.
IMPLEMENTATION IN MEDICINE
The development of PDAs for a specific procedure can be accomplished by analyzing data from patients who have undergone the treatment. For example, Swedish researchers analyzed the background and treatment of patients who received hip replacements. This was done on the premise that such a decision should be based on the patient’s values and preferences, as well as the surgeon’s experience and judgment.13 The same principle applies to dentistry: A patient will feel more positive about treatment if he or she is treated as an autonomous person and not a subject of the healthcare system. Although developing PDAs can be time-consuming, the benefit of these tools would immediately be seen in practice. With further acceptance of SDM and PDAs, their use will become increasingly important to dental providers and patients.
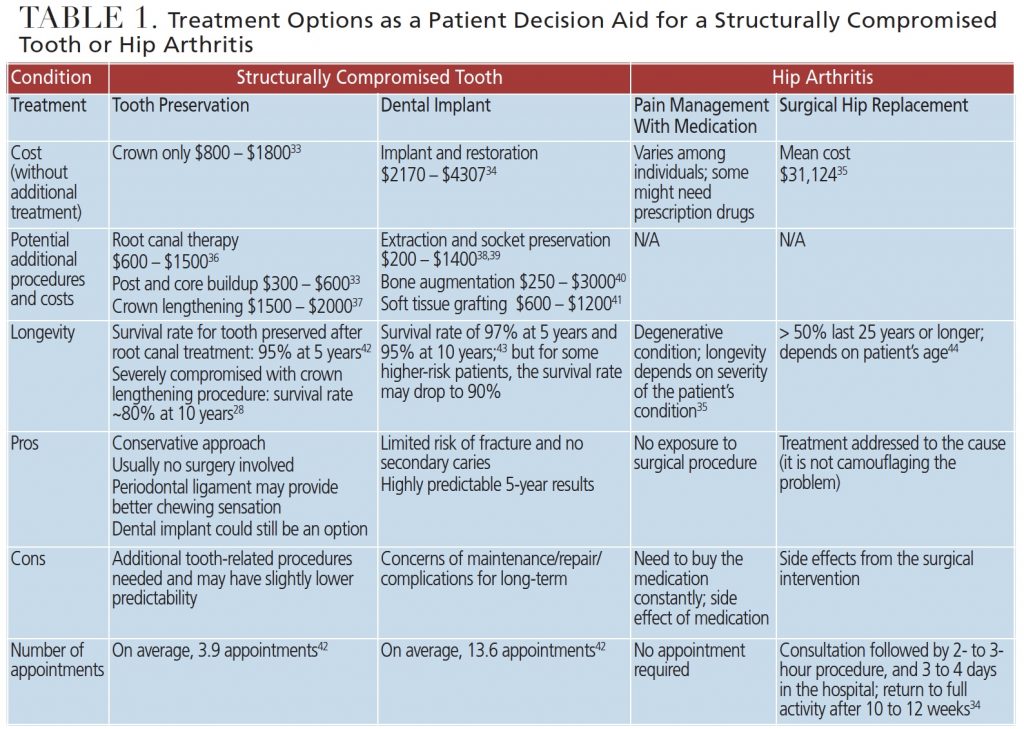
In medicine, a comparable example to the dental scenario of keeping or removing a tooth would be the decision-making process for hip replacements. Both procedures involve the choice of either preserving a skeletal structure or surgically replacing it with an artificial version. For each condition, noninvasive treatment and invasive surgical options exist — with each having a unique set of costs and benefits (Table 1). A study by Trenaman et al14 reports that using PDAs in joint replacement was associated with lower treatment costs and greater cost-effectiveness. Additionally, Slover et al15 found that in similar procedures, PDAs improved patient participation, knowledge and satisfaction.
IMPLEMENTATION IN DENTISTRY
While the SDM model is widely used in medicine — especially with patients diagnosed with cancer,16 arthritis,17 multiple sclerosis,18 atopic dermatitis,19 psoriasis20 and asthma21 — as noted, it has yet to gain significant traction in dentistry.7 Fortunately, the literature contains many examples that can be utilized to advocate its importance, including (but not limited to) its use in orthodontics,22 implants,23 endodontics,24 and management of asymptomatic third molars.25
A review by Alzahrani and Gibson9 notes that SDM is not well addressed with regard to implant dentistry, as they found a significant lack of patient-provider interaction aimed at understanding patients’ values and preferences. Risk communication was almost never conducted to educate patients about potential complications and the need for ongoing maintenance.
Under the SDM model, all treatment options should be presented to patients. Using the earlier example, the decision of whether to extract or preserve a tooth is complex and involves considerations from a restorative, endodontic and periodontal perspective. Often, the treatments required to develop an ideal implant site are overly simplified, and some intrinsic risks of implant complications are never communicated. Clinicians often tell patients that implants are more “predictable,” without explaining what that means.
Sharing objective information is a cornerstone of SDM. For instance, an endodontically treated tooth26 has a survival rate comparable to a dental implant,27 as both have survival rates of nearly 95% at the 5-year follow-up. A retrospective study showed a structurally compromised tooth that received a crown lengthening procedure had an average 5-year survival rate of 88%.28 Although the survival of these teeth dropped closer to 80% after 10 years (which may be lower than the success of the implant restoration), it is still higher than most clinicians and patients perceived. Additionally, if clinicians can control the risk of fracture by managing occlusal forces and providing a night guard — as well as fluoride to reduce secondary caries — success rates will go up simply by addressing these top two reasons for failure.28 The takeaway is that providing patients with treatment survival/success rates and discussing how to manage the risk for failure should be elemental parts of the SDM process.
For a tooth that may need to be extracted, the evidence-based model should not only communicate the tooth’s prognosis, but also include the average survival expectancy of the preserved tooth or alternative dental implant, the need for additional treatment, and cost. Additionally, the discussion should include how each procedure might affect the surrounding area so the patient knows exactly what he or she is getting beyond just a new tooth.
Dental teams can use PDAs to organize this information in a clear way. This objective information can help the patient reach a decision with the provider. For instance, a patient with a poor prognosis for a tooth can typically either replace an extracted tooth with a dental implant, or preserve the tooth.29 In this example, a patient could be given a PDA that explains both options (Table 1).
EXAMPLES IN USE
Although patient-shared and evidence-based decision-making is underrepresented in dentistry, research shows promising results with SDM implementation. In a randomized controlled trial, Johnson et al30 tested the efficacy of a PDA called an Endodontic Decision Board (EndoDB) in deciding whether to extract or preserve a tooth. The EndoDB presents five treatment options, all of which include an image representing the final product after treatment. In addition, each option includes a bulleted list communicating duration, cost, efficacy (against infection), and longevity of treatment. The trial involved randomly splitting 70 patients into two groups: one received the EndoDB before treatment, and the other did not. According to the authors, the EndoDB produced a “small, but statistically significant improvement in the knowledge of treatment options.” However, they also found that use of the EndoDB yielded more realistic patient expectations, less decisional conflict, and a higher rate of patient involvement in decision-making.
Park et al31 developed a decision system for choosing the most suitable restorations. This PDA was based on tooth anatomy, classification of disease, and treatment options. Another was created by Parker et al22 as a part of a clinical trial that sought to gauge patient satisfaction with a specific orthodontic appliance. Likewise, a PDA has been developed to help choose between conscious sedation and general anesthesia. This tool incorporates a booklet that is given to patients and parents/guardians.32 As opposed to a test group that received counseling only, the authors found knowledge was significantly higher in the group that received the PDA and counseling.
There is no doubt that PDA-assisted SDM needs additional study. In the meantime, clinicians and patients would be well served by using evidence-based PDAs to explain the pros and cons of various dental scenarios.
IN CONCLUSION
The success of SDM in medicine reflects its potential to enhance dental care. As noted, research shows patient knowledge and satisfaction can benefit from using PDAs in the decision process.13 In conclusion, SDM is a tremendous tool for treatment planning. Although not all patients are a good fit for SDM, educated patients appreciate the autonomy of consulting with their provider and reaching a well-informed decision based on the available evidence.
Acknowledgment: The authors thank Janet Zalucha, DDS, a resident in the graduate periodontics program at the University of Michigan School of Dentistry, and Omar Karadsheh, BDS, DChDent, an assistant professor at the University of Jordan School of Dentistry, for their valuable input in the development of this paper.
References
- Chiappelli F, Cajulis O. The logic model for evidence-based clinical decision making in dental practice. J Evid Based Dent Pract. 2009;9:206–210.
- Hoffmann TC, Montori VM, Del Mar C. The connection between evidence-based medicine and shared decision making. J Am Dent Assoc. 2014;312:1295–1296.
- Bauer J, Spackman S, Chiappelli F, Prolo P. Model of evidence-based dental decision making. J Evid Based Dent Pract. 2005;5:189–197.
- Coulter A, Collins A. Making shared decision making a reality: no decision about me, without me. Available at: https://tinyurl.com/ybwynh78. Accessed October 8, 2020.
- Charles C, Gafni A, Whelan T. Decision making in the physician-patient encounter: revisiting the shared treatment decision making model. Soc Sci Med. 1999;49:651–661.
- Charles C, Gafni A, Whelan T. Shared decision-making in the medical encounter: What does it mean? (or it takes at least two to tango). Soc Sci Med. 1997;44:681–692.
- Asa’ad, F. Shared decision‐making (SDM) in dentistry: A concise narrative review. J Eval Clin Pract. 2019;25:1088–1093.
- Thornton H, Edwards A, Elwyn G. Evolving the multiple roles of ‘patients’ in health-care research: reflections after involvement in a trial of shared decision making. Health Expect. 2003;6:189–197.
- Alzahrani AA, Gibson BJ. Scoping review of the role of shared decision making in dental implant consultations. JDR Clin Trans Res. 2018;3:130–140.
- Saleh HA, Ramadan R, Ghazzawi G, et al. Patient shared decision making: Physicians’ and patients’ perspective. Int J Pure Applied Sci Tech. 2014;22:10–17.
- Elwyn G, Scholl I, Tietbohl C, et al. “Many miles to go …”: a systematic review of the implementation of patient decision support interventions into routine clinical practice. BMC Med Inform Decis Mak. 2013;13(Suppl 2):S14.
- Giguere A, Legare F, Grad R, et al. Decision boxes for clinicians to support evidence-based practice and shared decision making: the user experience. Implement Sci. 2012;7:72.
- Cnudde P, Rolfson O, Nemes S, et al. Linking Swedish health data registers to establish a research database and a shared decision-making tool in hip replacement. BMC Musculoskelet Disord. 2016;17:414.
- Trenaman L, Stacey D, Bryan S, et al. Decision aids for patients considering total joint replacement: a cost-effectiveness analysis alongside a randomised controlled trial. Osteoarthritis Cartilage. 2017;25:1615–1622.
- Slover J, Shue J, Koenig K. Shared decision-making in orthopaedic surgery. Clin Orthop Relat Res. 2012;470:1046–1053.
- Clarke A, Paraskeva N, White P, Tollow P, Hansen E, Harcourt D. PEGASUS: the design of an intervention to facilitate shared decision-making in breast reconstruction. J Cancer Educ. 2020;doi:10.1007/s13187-019-01656-6.
- Binder-Finnema P, Dzurilla K, Hsiao B, Fraenkel L. Qualitative exploration of triangulated, shared decision-making in rheumatoid arthritis. Arthritis Care Res (Hoboken). 2019;71:1576–1582.
- Colligan E, Metzler A, Tiryaki E. Shared decision-making in multiple sclerosis. Mult Scler. 2017;23:185–190.
- LeBovidge J, Borok J, Udkoff J, Yosipovitch G, Eichenfield LF. Atopic dermatitis: therapeutic care delivery: therapeutic education, shared decision-making, and access to care. Semin Cutan Med Surg. 2017;36:131–136.
- Tan J, Linos E, Sendelweck MA, et al. Shared decision making and patient decision aids in dermatology. Br J Dermatol. 2016;175:1045–1048.
- Kew KM, Malik P, Aniruddhan K, Normansell R. Shared decision-making for people with asthma. Cochrane Database Syst Rev. 2017;10:CD012330.
- Parker K, Cunningham SJ, Petrie A, Ryan FS. Randomized controlled trial of a patient decision-making aid for orthodontics. Am J Orthod Dentofacial Orthop. 2017;152:154–160.
- Alzahrani AA, Gibson BJ. Scoping review of the role of shared decision making in dental implant consultations. JDR Clin Trans Res. 2018;3:130–140.
- Azarpazhooh A, Dao T, Ungar WJ, et al. Clinical decision making for a tooth with apical periodontitis: the patients’ preferred level of participation. J Endod. 2014;40:784–789.
- Edwards MJ, Brickley MR, Goodey RD, Shepherd JP. The cost, effectiveness and cost effectiveness of removal and retention of asymptomatic, disease free third molars. Br Dent J. 1999;187:380–384.
- Salehrabi R, Rotstein I. Endodontic treatment outcomes in a large patient population in the USA: an epidemiological study. J Endod. 2004;30:846–850.
- American Dental Association Council on Scientific Affairs. Dental endosseous implants: an update. J Am Dent Assoc. 2004;135:92–97.
- Ashnagar S, Barootchi S, Ravidá A, Tattan M, Wang HL, Wang CW. Long‐term survival of structurally compromised tooth preserved with crown lengthening procedure and restorative treatment: A pilot retrospective analysis. J Clin Periodontol. 2019;46:751–757.
- Levin L, Halperin-Sternfeld M. Tooth preservation or implant placement. J Am Dent Assoc. 2013;144:1119–1133.
- Johnson B, Schwartz A, Goldberg J, Koerber A. A chairside aid for shared decision making in dentistry: a randomized controlled trial. J Dent Educ. 2006;70:133–141.
- Park SG, Lee S, Kim MK, Kim HG. Shared decision support system on dental restoration. Expert Syst Appl. 2012;39:11775‐11781.
- Hulin J, Baker SR, Marshman Z, Albadri S, Rodd HD. Development of a decision aid for children faced with the decision to undergo dental treatment with sedation or general anaesthesia. Int J Paediatr Dent. 2017;27:344–355.
- Estimate Cost of Dental Care. Available at: https://www.guardiananytime.com/dceapp/DentalCostEstimatorWeb/dentalCostEstimator.process. Accessed October 8, 2020.
- Bouchard P, Renouard F, Bourgeois D, Fromentin O, Jeanneret MH, Beresniak A. Cost-effectiveness modeling of dental implant vs. bridge. Clin Oral Implants Res. 2009; 20:583–587.
- A Study of Cost Variations for Knee and Hip Replacement Surgeries in the U.S. Available at: https://www.bcbs.com/the-health-of-america/reports/study-of-cost-variations-knee-and-hip-replacement-surgeries-the-us. Accessed October 8, 2020.
- Resendiz J. Average Cost of a Root Canal in the United States: What You Can Expect. Available at: https://www.valuepenguin.com/average-cost-root-canal. Accessed October 8, 2020.
- Crown Lengthening. Available at: https://www.dental-treatment-guide.com/dental-crowns/crown-lengthening. Accessed October 8, 2020.
- How much does tooth extraction cost near you? Available at: https://www.authoritydental.org/tooth-extraction-cost. Accessed October 8, 2020.
- How Much Does a Dental Bone Graft Cost? Available at: https://health.costhelper.com/dental-bone-grafts.html. Accessed October 8, 2020.
- Alison A. Dental Bone Graft: Materials, Cost and Procedure. Available at: https://www.dentalimplantcostguide.com/dental-bone-graft/#cost. Accessed October 8, 2020.
- How Much Does a Gum Tissue Graft Cost? Available at: https://health.costhelper.com/gum-tissue-grafts.html. Accessed October 8, 2020.
- Vahdati SA, Torabinejad M, Handysides R, Lozada R. A retrospective comparison of outcome in patients who received both nonsurgical root canal treatment and single-tooth implants. J Endod. 2019;45:99–103.
- Jung R, Zembic A, Pjetursson B, Zwahlen M, Thoma D. Systematic review of the survival rate and the incidence of biological, technical, and aesthetic complications of single crowns on implants reported in longitudinal studies with a mean follow-up of 5 years. Clin Oral Implants Res. 2012;23(Suppl 6):2–21.
- Evans JT, Evans JP, Walker R, Blom A, Whitehouse M, Sayers A. How long does a hip replacement last? A systematic review and meta-analysis of case series and national registry reports with more than 15 years of follow-up. Lancet. 2019;393:16–22.
From Decisions in Dentistry. November 2020;6(10): 36-39.