 WAVEBREAKMEDIA / ISTOCK / GETTY IMAGES PLUS
WAVEBREAKMEDIA / ISTOCK / GETTY IMAGES PLUS
Rethinking the Use of Opioids in Moderate Sedation
The risks associated with opioids can be greater than the benefits, so dentists should consider alternative strategies during minimal or moderate sedation.
This course was published in the April 2022 issue and expires April 2025. The author has no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- In the context of dental care, explain the use and risks of opioids in minimal and moderate sedation.
- Describe the effects and potential effects of opioid sedatives on the respiratory system.
- List accepted alternatives to opioids in minimal and moderate dental sedation.
Morbidity and mortality during sedation of dental patients has occurred at an unacceptable level for many years. Due to the absence of a national database to report adverse events related to dental sedation — and because every state has different laws regarding the reporting of adverse events — exact numbers correlating to injury or death are difficult to obtain. However, several sources have reported that sedation accidents are extremely prevalent.1,2 Some estimates place the occurrence of morbidity/mortality events associated with dental sedation at approximately once a month in the United States.1
Examining closed-claim database reports reveals that for more than four decades the primary contributing factor to sedation deaths has been “adverse respiratory events” secondary to the “unanticipated effects of anesthetic medications”.1,3,4 With this information in mind, it is important for every dental provider who administers sedation to be aware of which medications have the potential to induce respiratory depression. Clinicians should use those medications judiciously — or consider not using them at all. Because opioid analgesics can produce acute respiratory depression and other severe morbidities, this article seeks to encourage dental providers to carefully reconsider the use of opioids in moderate sedation.
SEDATION CONTINUUM
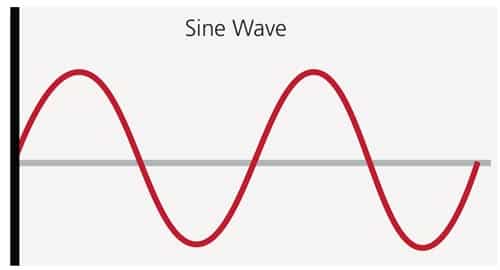
According to the American Society of Anesthesiologists and American Dental Association, there are four easily identifiable levels of sedation:5,6 minimal sedation, moderate sedation, deep sedation, and general anesthesia. Sedation is like a vacillating sinusoidal wave; sometimes the oscillation of the wave is near its top, while, at other times, the oscillation is near its bottom. Still, at other times, the wave is somewhere in the middle. The only thing constant about a sinusoidal wave is that it is always changing (Figure 1). Similarly, levels of sedation are not static; they are continuously changing. This means that a patient (due to many contributing factors) may move from a less sedated state to a more sedated state, or vice versa.
The definition of minimal sedation is “a drug-induced state during which patients respond normally to verbal commands.”5,6 Previously, minimal sedation has been referred to as anxiolysis, which implies a breaking down or dissolution of anxiety. When a patient is in minimal sedation, the individual is awake, and because of this, the patient is safe. Patients in minimal sedation may have a decrease in cognition and/or coordination secondary to the effects of the drugs that have been administered. However, these patients can independently maintain their own airway reflexes, and their respiratory/cardiovascular functions are unaffected.5,6
Moderate sedation, also commonly referred to as conscious sedation, is at a slightly deeper level than minimal sedation. In moderate sedation, the patient may be very relaxed, with closed eyes; however, the patient is still awake and will respond purposefully to verbal or light tactile stimulus.5 This means that if a sedation provider gently taps the patient on the shoulder or calls the individual by name, the patient will immediately respond. Because the patient is awake, moderate sedation (like minimal sedation) is safe, as patients in moderate sedation are also able to independently maintain airway reflexes and respiratory/cardiovascular functions.
Deep sedation is defined as a drug-induced state in which the patient will only purposely respond to a repeated or painful stimulus. General anesthesia is the deepest form of sedation in which the patient cannot be aroused, even when subjected to an acutely painful stimulus. The reason that deep sedation and general anesthesia are dangerous is that patients in these states often may not be able to maintain a patent airway by themselves. Often, their respiratory and cardiovascular functions are compromised, and these patients may need assistance maintaining a patent airway, and may require ventilatory assistance.
Purposefully inducing deep sedation or general anesthesia on a patient without a license to do so is dangerous and illegal. Most states require a dentist to have completed an accredited dental anesthesiology residency or an accredited oral maxillofacial surgery residency to purposefully induce deep sedation or general anesthesia. It is worth noting that the definition of general anesthesia does not mention the presence of an endotracheal tube. In other words, a pa-tient can very easily be in general anesthesia without being intubated. The state of general anesthesia is solely defined by a patient’s failure to respond to a painful stimulus. Because of this misunderstanding, many dental providers allow their patients to drift into deep sedation or general anesthesia without realizing it.
As noted, sedation is a continuum; therefore, it is common for a patient to move from one level of sedation to another. Dentists who choose to administer sedation must be able to distinguish one level from another. If a patient becomes more sedated than intended, or progresses to a level of sedation or anesthesia that the provider does not have a license to perform, the provider must be capable of recognizing this deeper level and able to “rescue” and return the patient to a safe, acceptable level.7
RECOGNITION OF PATIENT INDIVIDUALITY IN RESPIRATORY CONTROL
It is crucial to remember that every patient is different. The age, weight, medications, allergies, organ function, and comorbidities are nuanced and vary with each individual. It is incumbent upon every sedation provider to approach each person as an individual and provide treatment according to the patient’s specific needs. Sedation providers must consider the effect the administration of an opioid will have on a particular individual. Differences in patients’ ages, comorbidities and medications will result in different levels of respiratory depression secondary to opioid administration, especially when combined with other sedative drugs.8,9
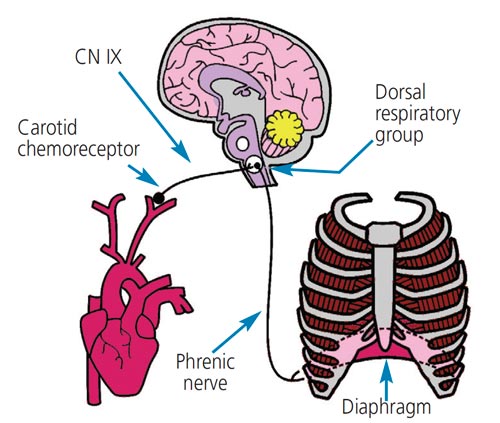
Located at the bifurcation of the common carotid arteries are a group of specialized cells called chemoreceptors that constantly monitor arterial blood for changes in the amount of oxygen (PaO2), carbon dioxide (PaCO2), and acid (H+). When a person’s respiratory rate decreases or stops, the PaO2 in arterial blood decreases and the amounts of PaCO2 and H+ increase. These changes in blood chemistry stimulate the chemoreceptors in the carotid bodies to send a signal to the brain by way of the glossopharyngeal nerve.10 When the signal reaches the dorsal respiratory group of the medulla, the brain sends a signal by way of the phrenic nerve to stimulate the diaphragm to contract. This results in the patient taking faster, deeper breaths to increase the amounts of PaO2 in the bloodstream and decrease the amounts of PaCO2 and H+ (Figure 2).10 When a patient is exposed to an opioid, this reaction is inhibited and respiratory drive decreases or ceases, depending on the amount of the opioid and the individual’s sensitivity to the opioid.9 In other words, some patients’ respiratory drive is affected differently than others.
OPIOIDS AND OPIOID RECEPTORS
Three natural opiates (morphine, codeine and thebaine) can be isolated from the milky extract of the poppy plant. These substances have been used for thousands of years to treat pain and other maladies, such as dysentery.11 Opioids are any chemical (natural, synthetic or endogenous) that bind to the different kinds of opioid receptors found throughout the body. The mu opioid receptor can be subdivided into mu1 and mu2 receptors. Mu1 receptors are responsible for analgesic responses to painful stimuli. Mu2 receptors are responsible for the less desirable effects of opioid use, including respiratory depression. The three opioids most used in sedation are morphine, meperidine and fentanyl. These drugs all bind to the mu2 receptor and cause a dose-related respiratory depression.
Morphine is the prototypical opioid that all other opioids are compared to. While morphine is extremely effective at achieving sedation and analgesia, there are many undesirable side effects and complications associated with its administration. For example, the peak effect of action after morphine is given via the intravenous route is approximately 20 minutes due to its low lipid solubility, and its duration of action can be more than six hours due to its active metabolite, morphine-6-glucuronide.12 It is not uncommon for a provider to administer a second dose of morphine before the effects of the initial dose are appreciated. The result may be an additive effect of both doses, resulting in oversedation and respiratory depression. It is also common for patients who receive morphine in a short outpatient setting (such as the dental office) to return home and experience lingering sedation and respiratory depression. Morphine causes histamine release, which can exacerbate conditions in patients with obstructive lung diseases (chronic obstructive pulmonary disease and asthma, for example). The histamine release related to morphine administration can also cause hemodynamic instability in susceptible patients.
In elixir form, meperidine has often been used for the sedation of pediatric patients and individual with special needs. It is also used as an intravenous drug for adult sedation. Meperidine has a quicker onset of action and shorter duration than morphine. However, the safety profile of meperidine is extremely narrow, and its use should be limited, if used at all. This drug has many dangerous side effects. Like morphine, it causes profound respiratory depression and histamine release, which can result in severe hypotension and bronchospasm. The drug, while technically a synthetic opioid, has marked anticholinergic effects, which can result in severe tachycardia, biliary spasm and urinary retention.13 It interacts with numerous common drugs, including erythromycin, acyclovir and cimetidine.14 It can cause a fatal interaction if administered within 14 days of a monoamine oxidase inhibitor, and can cause a life-threatening condition called serotonin syndrome if given to patients taking selective serotonin reuptake inhibitors or tricyclic antidepressants.
Meperidine is metabolized in the liver to form a toxic metabolite called normeperidine, which has the propensity to cause severe central nervous system excitation, including seizures, severe hallucinations, anxiety, mood disorders, myoclonus and hyperreflexia.13 Its use is directly contraindicated in patients with renal dysfunction.
Fentanyl is a synthetic opioid and is 100 times as potent as morphine.12 Providers who dose fentanyl in the same manner as morphine will massively overdose the patient. Because fentanyl is lipid soluble and has a quick onset of action (approximately two minutes), it has become a popular drug for inducing sedation. Fentanyl’s duration of action is short (approximately 40 minutes), making it safer than morphine for short outpatient procedures. However, fentanyl has several serious side effects that must be considered. Rapid doses of fentanyl can cause life-threatening chest wall rigidity. Fentanyl can cause severe, potentially life-threatening bradycardia, especially in beta-blocked patients and older patients. Most importantly, fentanyl is a potent respiratory depressant; its ability to decrease or eliminate a patient’s ability to breathe must not be underappreciated. In addition, life-threatening respiratory depression can be magnified if combined with other drugs (such as benzodiazepines) or other opioids.
METABOLIZATION
A thorough understanding of how and where drugs are metabolized is important for clinicians who administer any drug enterally or parenterally. Most drugs are metabolized in the liver through phase I or phase II metabolic pathways. Many drugs, including several opioids, form active or toxic metabolites that must be considered during and after sedation. Failure to properly understand the pharmacokinetics of opioids can have dire consequences.
Fentanyl and meperidine are both metabolized in the liver through phase I reactions by the cytochrome P450 enzyme CYP3A4. This is significant because any drug that inhibits the CYP3A4 enzyme can cause a buildup of fentanyl or meperidine in the blood plasma and lead to exacerbated side effects. Especially concerning is the potential for acute, and potentially fatal, respiratory depression.12,14,15 Common drugs that inhibit CYP3A4 enzymes include:
- Selective serotonin reuptake inhibitors (e.g., fluoxetine and sertraline)
- Statins (e.g., simvastatin)
- Antibiotics (e.g., erythromycin and ciprofloxacin)
- Antifungal agents (e.g., fluconazole and clotrimazole)
- Histamine blockers (e.g., cimetidine)
OPIOID REVERSAL
Many sedation practitioners justify the use of opioids by reasoning that opioid receptor blockade can be fully reversed with the antagonist naloxone. While it is true the effects of opioid overdose can be reversed with naloxone, this should never be used as the deciding factor in choosing an opioid for sedation. There are many issues that need to be considered before any opioid is reversed with naloxone.
Naloxone is an opioid antagonist that binds to mu receptors and reverses the effects of opioid administration, including analgesia, sedation, bradycardia and respiratory depression. Unfortunately, the serum half-life of naloxone is only 60 minutes, while the serum half-life of most opioids is greater than 60 minutes.16 This means most patients will experience some opioid effects after the naloxone has been metabolized. Some patients will become re-sedated and may have significant respiratory depression when the naloxone wears off. All patients who are given a reversal of naloxone must be observed for at least one hour to see if re-sedation and/or respiratory depression occurs.
There are many other undesirable side effects associated with the use of naloxone. Because it reverses the effects of analgesia, its administration results in the release of endogenous catecholamines. Patients who receive large doses of naloxone are at risk for developing tachyarrhythmias, hypertension, acute algesia and pulmonary edema.16 The cardiac and pulmonary effects of naloxone can be fatal and must be considered before administration.
OTHER CONSIDERATIONS
Fentanyl and meperidine are both basic lipophilic amines that bind to lung tissue when injected into the bloodstream. Normally, up to 80% of fentanyl is bound to lung tissues, meaning that approximately 20% of the fentanyl administered intravenously is immediately available to bind to opioid receptors. When another basic lipophilic amine is bound to lung tissues, fentanyl is unable to bind to the same tissues at its normal rate. For example, when a patient is taking propranolol, a non-selective beta blocker used for angina and hypertension, intravenous fentanyl is only bound to lung tissue at a rate of 20% instead of 80%.17 This can result in a massive amount of fentanyl circulating freely in the bloodstream and lead to a fatal overdose. A recent trend in treating patients with intellectual disabilities is to prescribe propranolol for behavior management.18 Patients with special needs, or anyone who receives propranolol, should not be sedated with fentanyl or meperidine.
ALTERNATIVE AGENTS
Opioids possess desirable sedative and analgesic properties, but can also produce potentially life-threatening side effects and complications. Most general dentists and specialists (outside of oral maxillofacial surgeons and dentist anesthesiologists) who offer sedation only have a license to practice minimal or moderate sedation. Opioids, especially when combined with other drugs, such as antihistamines, benzodiazepines and nitrous oxide, have a propensity to push patients into a deeper plane of anesthesia than intended. These agents can also interact with multiple drugs and cause undesirable side effects. Most importantly, all opioids cause respiratory depression. The question must then be posed: If opioids have such a high risk factor, why use them for sedation? If the benefit of using a drug is outweighed by its risks, the drug should not be used.
There are many alternatives to opioids (e.g, nitrous oxide and/or benzodiazepines) for achieving minimal and moderate sedation. Remember, the level of sedation a patient achieves is not defined by the drug that is administered. Rather, it is defined by the patient’s response to stimulus. If minimal or moderate sedation can be safely and easily induced without the use of an opioid, that option should be considered first.
CONCLUSION
Morbidity and mortality during sedation in the dental setting occurs too often in the United States. These adverse events most commonly result from a patient being oversedated and losing an airway. Respiratory depression secondary to opioid administration is one of many undesirable side effects of using opioids as part of a sedation regimen. Because the risks associated with opioid use can be greater than the benefits when performing minimal or moderate sedation, dental practitioners should consider alternatives to opioids when sedating patients.
REFERENCES
- Bennett JD, Kramer KJ, Bosack RC. How safe is deep sedation or general anesthesia while providing dental care? J Am Dent Assoc. 2015;146:705–708.
- Lee HH, Milgrom P, Starks H, Burke W. Trends in death associated with pediatric dental sedation and general anesthesia. Paediatr Anaesth. 2013;23:741–746.
- Metzner J, Posner KL, Domino KB. The risk and safety of anesthesia at remote locations: the U.S. closed claims analysis. Curr Opin Anaesthesiol. 2009;22:502–508.
- Bhananker SM, Posner KL, Cheney FW, Caplan RA, Lee LA, Domina KB. Injury and liability associated with monitored anesthesia care. Anesthesiology. 2006;104:228–234.
- American Society of Anesthesiologists. Continuum of Depth of Sedation: Definition of General Anesthesia and Levels of Sedation/Analgesia. Available at: https://www.asahq.org/standards-and-guidelines/continuum-of-depth-of-sedation-definition-of-general-anesthesia-and-levels-of-sedationanalgesia. Accessed March 11, 2022.
- American Dental Association. Guidelines for the Use of Sedation and General Anesthesia by Dentists. Available at: https://www.ada.org/resources/research/science-and-research-institute/oral-health-topics/anesthesia-and-sedation. Accessed March 11, 2022.
- American Dental Association. Guidelines for Teaching Pain Control and Sedation to Dentists and Dental Students. Available at: https://www.mouthhealthy.org/~/media/ADA/Education% 20and%20Careers/Files/ADA_Sedation_Teaching_Guidelines.pdf?la=en. Accessed March 11, 2022.
- Dasgupta N, Jonsson Funk M, Proescholdbell S, Hirsch A, Ribisl KM, Marshall S. Cohort study of the impact of high-dose opioid analgesics on overdose mortality. Pain Med. 2016;17:85–98.
- Pattinson KTS. Opioids and the control of respiration. Br J Anaesth. 2008;100:747–758.
- Johnson DC. Kazemi H. Control of ventilation. Available at: https://somepomed.org/articulos/contents/mobipreview.htm?10/葬/?source=HISTORY. Accessed March 11, 2022.
- Pasternak GW, Pan Y-X. Mu opioids and their receptors: evolution of a concept. Pharmacol Rev. 2013;65:1257–1317.
- Alexander JC, Casserly E. Perioperative uses of intravenous opioids in adults: General considerations. Available at: https://www.uptodate.com/contents/perioperative-uses-of-intravenous-opioids-in-adults-general-considerations. Accessed March 11, 2022.
- Latta KS, Ginsberg B, Barkin RL. Meperidine: a critical review. Am J Ther. 2002;9:53–68.
- Highlights of Prescribing Information: Demerol. Available at: https://www.accessdata.fda. gov/drugsatfda_docs/label/떓/⭳s032lbl.pdf. Accessed March 11, 2022.
- Smith HS. Opioid metabolism. Mayo Clin Proc. 2009;84:613–624.
- Lynn RR, Galinkin JL. Naloxone dosage for opioid reversal: current evidence and clinical implications. Ther Adv Drug Saf. 2018;9:63–88.
- Roerig DL, Kotrly KJ, Ahlf SB, Dawson CA, Kampine JP. Effect of propranolol on the first pass uptake of fentanyl in the human and rat lung. Anesthesiology. 1989;71:62–68.
- Hiremath A, Gangavati S, Bhaumik S, Kiani R, Devapriam J. A study on the use of propranolol in managing behavioural problems in people with intellectual disability. Br J Dev Disabilities. 2010;56:147–154.
From Decisions in Dentistry. April 2022;8(4)30-33




