 VSARGUES / ISTOCK / GETTY IMAGES PLUS
VSARGUES / ISTOCK / GETTY IMAGES PLUS
Recognizing Orofacial Myofunctional Disorders and Obstructive Sleep Apnea
The following strategies can help clinicians recognize and manage sleep apnea in the dental setting.
This course was published in the April 2022 issue and expires April 2025. The author has no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Discuss the link between orofacial myofunctional disorder and obstructive sleep apnea (OSA).
- Identify oral health professionals’ role in detecting sleep-disordered breathing and OSA.
- List treatment options for OSA.
Oral health professionals are well positioned to recognize the signs and symptoms of obstructive sleep apnea (OSA) and make the appropriate referrals for medical consultation. The frequency of appointments, and prophylaxis appointments in particular, makes the dental office an ideal setting to conduct OSA screenings. Oral healthcare providers, however, may lack sufficient knowledge about OSA to deliver such screening with confidence.1
Sleep-disordered breathing (SDB) is a dysfunction of the upper airway that occurs during sleep due to increased upper airway resistance and weakened oropharyngeal musculature.2 As a sleeping disorder, OSA is characterized by repetitive episodes of upper airway occlusion during sleep,3 which decrease blood oxygen levels and cause fragmented sleep.4 This condition affects both adults and children, and occurs in 15% to 25% of middle-aged and older adults.5 Approximately 70% to 90% of cases remain undiagnosed.5
During sleep, repetitive episodes of total or partial upper airway collapse lead to decreased oxygen levels or hypoxia, which negatively impacts mental and systemic health.6 The effects of hypoxia — which include inflammation, oxidative stress, and increased sympathetic nervous system activity — may be associated with serious comorbidities, such as heart disease, hypertension, type 2 diabetes, Alzheimer’s disease and dementia.7 The physiological changes resulting from impaired breathing may also cause low mood; increased automobile, home and work-related accidents; abnormal dentition development; and even premature death.8–10
The root cause of OSA often stems from improper jaw growth and facial development during childhood.11 Adenotonsillar hypertrophy is a common cause of OSA, particularly in children; however, other anatomical factors may be involved.2 Nocturnal breathing disorders, such as OSA and snoring, impact the developing dental arches. Individuals with SDB and OSA often present with a narrow maxilla, deep palatal height, short mandibular arch, and high prevalence of lateral crossbites.12 Emerging research shows orofacial structures as the predisposing factor involved in the development of SDB; therefore, the relationship between normal breathing and normal orofacial growth is now receiving recognition in sleep medicine.13
OBSTRUCTIVE SLEEP APNEA AND OROFACIAL MYOFUNCTIONAL DISORDER
Clinically, SDB is an umbrella term that encompasses several sleep-related breathing disorders, including OSA. As noted, SDB is caused by an increase in the resistance of the upper airway and pharyngeal collapse, and is characterized by snoring and greater respiratory effort.2 More specifically, OSA occurs when breathing is interrupted during sleep because the upper airway becomes obstructed repeatedly by the soft palate, base of the tongue, and/or musculature collapse, reducing or completely stopping airflow.4 During sleep, individuals may experience apnea, which is when breathing stops entirely, or hypopnea, which is when breathing is abnormally slow or shallow. Risk factors for developing OSA include a large neck circumference,7 large tonsils, large tongue or uvula, recessed chin, overbite, obesity, improper oral rest posture, and smoking or alcohol use.5
Sleep apnea is diagnosed using a polysomnogram (PSG), a formal sleep study that detects abnormal levels of activity in the muscles that control breathing. In a PSG assessment, clinicians also monitor blood oxygen levels and brain and heart activity.10 The severity of OSA is measured using the apnea-hypopnea index (AHI), indicating the number of times that breathing stops, or slows, during sleep. This value is expressed as the number of events per hour (Table 1).5 
Orofacial myofunctional disorders (OMD) are commonly seen in patients with OSA. A dysfunction of the stomatognathic system, OMD results in alterations to the appearance, posture and mobility of the lips, tongue, mandible and cheeks.14,15
Oral dysfunction may begin at birth, and often becomes apparent as speech develops and solid foods are introduced. This dysfunction may affect facial skeletal growth and development, mastication, speech, occlusion, stability of orthodontic treatment, facial esthetics, and upper airway function.16 In addition, OMD may lead to abnormal facial development and malocclusion if timely intervention does not occur.14
Most OMDs originate with insufficient nasal breathing as the muscles and orofacial functions adapt to a disordered breathing pattern.14 Those with collapsed airways during sleep often change their body position or head posture, or the position of their tongue and/or mandible in order to breathe. Long-term postural changes impact the developing dental arches because of the positional changes of the head, mandible, and tongue caused by an insufficient airway.12 Those with SDB commonly breathe through their mouth and have a forward head posture in an effort to open the airway. Mouth breathing alters oral rest posture, increasing nasal resistance, causing tongue malposition, exacerbating impaired development, and hampering the growth of the maxilla and mandible.2 Mouth breathing during sleep causes changes in the muscular balance between the tongue and cheeks, leading to a narrow maxillary arch. Increased lip pressure occurs from the low tongue position and forward head posture, leading to a shorter length of the mandible and mandibular crowding.12
IMPORTANCE OF PROPER ORAL REST POSTURE
Oral rest posture is where the teeth, lips and tongue lie when a patient is in a neutral resting position. In proper oral rest posture, the distance between the maxillary and mandibular teeth at rest should be 2 to 3 mm in the posterior and 2 to 5 mm in the anterior, the lips should gently touch, and the apex of the tongue should rest just lingual to the maxillary anterior teeth, on the incisive papilla.17 This posture encourages nasal breathing and allows the lips to act as an outward muscular force on the teeth. Upon swallowing, the tongue has a modeling role on the oral cavity, stimulating intermaxillary synchondrosis, and leading to normal orofacial growth and development. Although the maxilla fuses in utero, it continues to grow during childhood, with only about 80% of skull and jaw growth completed by age 6,17 therefore, a low or forward tongue posture, especially in early childhood, can cause a high-vaulted palate, open bite, or posterior crossbites. A low tongue posture can also cause the outward facial appearance to change, resulting in a narrower, longer forehead and nose, closer-set eyes, flattened cheekbones, and a recessed chin.17
Poor oral habits, such as prolonged thumb sucking, pacifier use, or tongue thrust, can also cause the tongue to rest in a low position. In children older than 3, nonnutritive sucking habits may cause permanent damage.18 Sucking habits cause a high, narrow palatal vault because the buccal muscular forces are normal, but the tongue is not reciprocating these forces. When the tongue is thrust forward against the teeth when swallowing, a low tongue posture is usually present as well.
ROLE OF DENTAL PROFESSIONALS
As noted at the outset, the dental office is an ideal setting to conduct OSA screenings.1 During extraoral and intraoral examinations, oral cancer screening is common, and evaluating the oral cavity for anatomical variations indicative of OSA is a practical addition to this routine assessment. Thus, standardized OSA screenings can be easily incorporated into clinical practice.1
Oral health professionals can screen for OSA by identifying associated comorbid conditions, recognizing craniofacial and oral risk factors, using OSA-related health questionnaires, and referring patients to their primary care physicians, as indicated. Additionally, dental teams can provide support and education to patients with OSA by discussing behavior modifications, such as tobacco cessation and nutritional counseling.5 Only a physician can legally diagnose OSA, but oral healthcare providers can promote awareness, recognize and screen for signs and symptoms, and make referrals to appropriate medical professionals for a definitive diagnosis.4 Research shows that how well an individual breathes (during the day and at night) significantly impacts craniofacial, neurological and airway development.12
Dental professionals should conduct an airway screening on every patient, which should include:
Patient History — A medical history review may provide evidence of OSA symptoms, such as medication use, attention deficit hyperactivity disorder, frequent ear infections, throat infection, hypertension, cardiac incidents, or depression.4 Patients who are overweight are more likely to have OSA, especially if there is excess fat stored around the neck and waist.19 Questions, such as “Do you snore at night?” or “Do you stop breathing for short periods during sleep?” should be asked. Additional questions include whether the patient is restless during sleep, difficult to rouse in the morning, or, for pediatric patients, if behavior issues are present.4
Questionnaires — The Epworth sleepiness scale, which assesses daytime sleepiness, can be integrated into the screening process.20 Additional questionnaire tools used to identify OSA risk include:
- STOP (snoring history, tired during the day, observed stopped breathing during sleep, high blood pressure)
- STOP-BANG (snoring history, tired during the day, observed stopped breathing during sleep, high blood pressure, body mass index more than 35 kg/m2, age older than 50, neck circumference more than 40 cm, and male gender)
- BEARS (bedtime, excessive daytime sleepiness, awakening during the night, regularity, and duration of sleep and snoring), which is for children21–23
Extraoral and Intraoral Examination — A forward neck posture is common among patients with SDB because it helps open the airway. Facial changes related to airway issues may include a long face and nose, close-set eyes, flattened cheekbones, and venous pooling under the eyes. Additionally, dental team members can look for signs of sleep deprivation or mouth breathing.19 Areas of the mouth that can provide evidence of SDB include the tissues, teeth, tongue, throat and tonsils. The teeth may show signs of occlusal wear from bruxism, caries on the anterior teeth due to mouth breathing, and a high, narrow maxillary arch, class II malocclusion, or deep overbite. A crowded dentition may also be evident, with patients presenting with linguoverted teeth and/or crossbites.24 The tissues may show evidence of mouth breathing by appearing inflamed and erythematous.
Periodontal conditions are also common in patients with SDB. While the underlying mechanism explaining this association is not clear, neither is the direction of the association. Periodontitis may reflect an inflammatory burden that results in a robust host response to bacterial plaques among individuals with OSA. As with other cardiovascular and metabolic diseases, both OSA and periodontitis pathophysiology involve oxidative stress, chronic inflammation, and elevated C-reactive protein levels.25
The size, placement and movement of the tongue should be evaluated. Macroglossia and a scalloped tongue are indicators for OSA, mostly in adults. Ankyloglossia is also a common finding, as it prevents the tongue from pressing into and shaping the maxilla, resulting in a high vaulted palate.17 The tonsils may be enlarged, and frequent throat infections may be reported. Tonsils vary in size and are classified accordingly (Table 2).26 The pharynx should be evaluated for signs of irritation. The tonsillar pillars may appear red and edematous from snoring or recurrent infections. 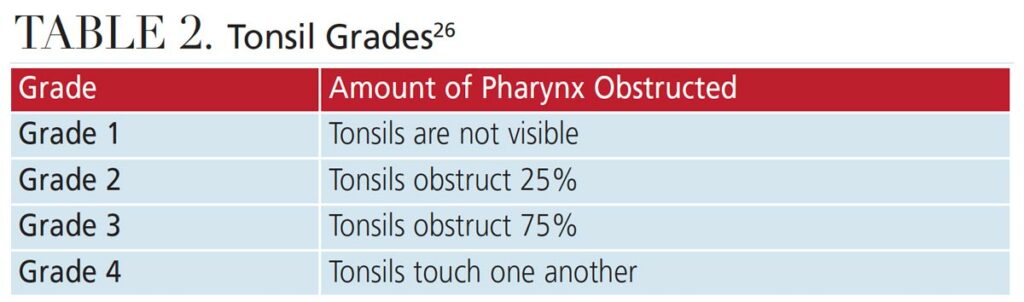
The Mallampati classification can be used to evaluate the size and anatomy of the airway, using visual inspection to evaluate the distance from the base of the tongue to the soft palate, and to classify the opening of the airway based on what oral structures are visible (Table 3).27 Mallampati class is assessed with the head in full extension, the mouth wide open and the tongue extruded, with the patient in an upright position with and without phonation.28 Patients with a class III or IV Mallampati may be more prone to airway obstruction.27 When evaluating the pharynx, patients with a restricted airway may display common airway protective reflexes such as making a “gnn” sound (as opposed to an “ahh” sound) when depressing the tongue or pushing the tongue against the dental mirror.27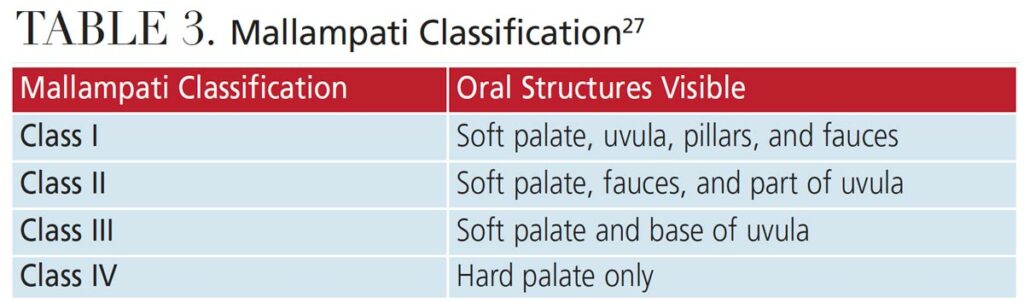
TREATMENT OF OBSTRUCTIVE SLEEP APNEA
Integrating sleep medicine and dental sleep medicine improves patient care, reduces treatment costs, and improves physician education. An integrated, multidisciplinary approach to OSA management is most effective.29 Several treatment options are available to manage OSA. The most common and effective is a continuous positive airway pressure (CPAP) device that uses positive airflow to keep the airway open during sleep, allowing for sufficient oxygen intake. Patient compliance with CPAP machines may be low because the device is cumbersome, and many patients find its use uncomfortable during sleep.4
Dentist-fabricated oral appliances, such as mandibular advancement and tongue-retaining devices, are designed to open the upper airway. They prevent occlusion of the airway by holding the mandible and tongue in a forward position. Oral appliances are most effective for patients in the AHI range of 5 to 30 (mild-moderate).4 Sleep testing should be completed prior to fabrication. Not every patient is a good candidate for an oral appliance; therefore, a thorough examination of the temporomandibular joint, bony structures, gingiva, and teeth is required.4
Myofunctional therapy is a noninvasive treatment option that uses an interdisciplinary approach. This treatment works with the orofacial muscles and their related functions, such as breathing, sucking, chewing, swallowing and speech, and can improve facial esthetics, as well as the airway.30 Myofunctional therapists work collaboratively with other health professionals to treat both OMD and OSA. When initiated early, myofunctional therapy can prevent the evolution of improper development, restore correct stomatognathic function and nasal breathing, and reestablish tongue position. It can be used with other treatments to avoid residual OSA. Dental team members can learn to perform these treatments.30
Additional therapeutic approaches include lifestyle changes and surgery. For example, increasing physical activity, weight loss, smoking cessation, and eliminating or reducing alcohol consumption will also decrease OSA risk. Surgical procedures that remove the soft tissues in the posterior pharynx can expand the size of the airway.4
CONCLUSION
Implementing an interprofessional approach will help optimize treatment for SDB and OSA. The most common referrals for these disorders are to a physician, ear, nose and throat specialist, sleep specialist, orthodontist and myofunctional therapist.2 Oral healthcare providers can be trained to identify physical, behavioral, and birth history clues that can lead to diagnosis and intervention.4 Unhealthy sleep patterns negatively impact performance in all aspects of development and education; therefore, dental teams should routinely screen for airway disorders, thus serving as the first line of defense against sleep disruption. As such, incorporating a screening protocol that uses a comprehensive, whole-body approach is recommended.
REFERENCES
- Reibel Y, Pusalavidyasagar S, Flynn P. Obstructive sleep apnea knowledge: attitudes and screening practices of Minnesota dental hygienists. J Dent Hyg. 2019;93:29–36.
- Villa MP, Evangelisti M, Martella S, Barreto M, Del Pozzo M. Can myofunctional therapy increase tongue tone and reduce symptoms in children with sleep-disordered breathing? Sleep Breath. 2017;21:1025–1032.
- Guimarães KC, Drager LF, Genta PR, Marcondes BF, Lorenzi-Filho G. Effects of oropharyngeal exercises on patients with moderate obstructive sleep apnea syndrome. Am J Respir Crit Care Med. 2009;179:962–966.
- Agarwal T, Shahidi H. Treating obstructive sleep apnea in the dental setting: clinical and practice management considerations. Dental Academy of Continuing Education. 2018;1–10.
- An SL, Ranson C. Obstructive sleep apnea for the dental hygienist: overview and parameters for interprofessional practice. Can J Dent Hyg. 2011;45:238–252.
- Rohra AK, Demko CA, Hans MG, Rosen C, Palomo JM. Sleep disordered breathing in children seeking orthodontic care. Am J Orthod Dentofacial Orthop. 2018;154:65–71.
- Hakes H. Sleep apnea: a growing health concern hygienists need to know about. Available at: todaysrdh.com/sleep-apnea-a-growing-health-concern-hygienists-need-to-know-about/. Accessed March 9, 2022.
- American College of Cardiology. Sleep apnea and high blood pressure a dangerous pair. Available at: cardiosmart.org/news/떏/䁳/sleep-apnea-and-high-blood-pressure-a-dangerous-pair. Accessed March 9, 2022.
- Peppard PE, Young T, Barnet JH, Palta M, Hagen EW, Hla KM. Increased prevalence of sleep-disordered breathing in adults. Am J Epidemiol. 2013;177:1006–1014.
- Maspero C, Giannini L, Galbiatti G, Rosso G, Farronato G. Obstructive sleep apnea syndrome: a literature review. Minerva Stomatol. 2015;64:97–109.
- Hodge M. Obstructive sleep apnea: how lifetime care can change everything. Available at: dentaleconomics.com/science-tech/article/줤/obstructive-sleep-apnea-how-lifetime-care-can-change-everything. Accessed March 9, 2022.
- Pirila-Parkkinen K, Pirttiniemi P, Nieminen P, Tolonen U, Pelttari U, Lopponen H. Dental arch morphology in children with sleep-disordered breathing. Eur J Orthod. 2009;31:160–167.
- Guilleminault C, Sullivan SS. Towards restoration of continuous nasal breathing as the ultimate treatment goal in pediatric obstructive sleep apnea. Available at: semanticscholar.org/paper/Towards-Restoration-of-Continuous-Nasal-Breathing-Dm-Guilleminault/ⲍbc102b4f26bd 93124f7f7dd27706e80b8e24f. Accessed March 9, 2022.
- Folha GA, Valera FCP, Felício CMD. Validity and reliability of a protocol of orofacial myofunctional evaluation for patients with obstructive sleep apnea. Eur J Oral Sci. 2015;123:165–172.
- Andrade RAD, Cunha MDD, Da Costa Dos Santos Reis A. Morphofunctional analysis of the stomatognathic system in conventional complete dentures users from the Integrated Health Center. Revista CEFAC. 2017;19:712–725.
- D’onofrio L. Oral dysfunction as a cause of malocclusion. Orthod Craniofac Res. 2019;22(S1):43–48.
- D’Urso N. Muscle memory: a review of tongue muscles and its functions and dysfunctions. Dental Academy of Continuing Education. 2018;1–10.
- Wilkins EM. Clinical Practice of the Dental Hygienist. 13th ed. Philadelphia: Lippincott Williams & Wilkins; 2021.
- Bewley B. The role of the dental hygienist in screening for sleep apnea. J Dent Hyg. 2014;88:5.
- Fabbie P. Myofunctional analysis and its role in dental assessments and oral health. Available at: dentalacademyofce.com/courses/뤝/PDF/뎔cei_Fabbie_WEB.pdf. Accessed March 9, 2022.
- Kornegay E, Brame J. Obstructive sleep apnea and the role of dental hygienists. J Dent Hyg. 2015;89:286–292.
- Pavarangkul T, Jungtrakul T, Chaobangprom P, et al. The STOP-BANG questionnaire as a screening tool for obstructive sleep apnea induced hypertension in Asian population. Neurol Int. 2016;8:6104.
- Ebrahim A, Babak G, Alimohammad A, Shabnam J, Alireza A, Forough F. High prevalence of sleep problems in school- and preschool-aged children in Tehran: a population based study. Iran J Pediatr. 2013;23:45–52.
- Schroeder K, Gurenlian JR. Recognizing poor sleep quality factors during oral health evaluations. Clin Med Res. 2019;17:20–28.
- Billings ME. Putting some teeth into it: connecting periodontitis with sleep apnea. Sleep. 2015;38:1153–1154.
- Cahali MB, Soares CF, Dantas DA, Formigoni GG. Tonsil volume, tonsil grade and obstructive sleep apnea: Is there any meaningful correlation? Clinics. 2011;66:1347–1352.
- Kandray D, Juruaz D, Yacovone M, Chang A. Inter-rater reliability of the Mallampati classification for patients in a dental hygiene clinic. J Dent Hyg. 2013;87:134–139.
- Khan ZH, Eskandari S, Yekaninejad MS. A comparison of the Mallampati test in supine and upright positions with and without phonation in predicting difficult laryngoscopy and intubation: a prospective study. J Anaesthesiol Clin Pharmacol. 2015;31:207.
- Nakai T, Matsuo A, Takata Y, Usui Y, Kitamura K. Chikazu D. Role of dental sleep medicine in management of patients with obstructive sleep apnea disorders using a team approach. Acta Odontologica Scandinavica. 2018;76:605–611.
- Academy of Orofacial Myofunctional Therapy. What is Myofunctional Therapy? Available at: aomtinfo.org/myofunctional-therapy/. Accessed March 9, 2022.
From Decisions in Dentistry. April 2022;8(4)34-36,39.




