EMMUZCAN/ISTOCK/GETTY IMAGES PLUS
EMMUZCAN/ISTOCK/GETTY IMAGES PLUS
Preventing and Managing Infection Control Breaches
The following strategies will help oral health professionals prevent infection control breaches — such as needlestick injuries — and manage postexposure events.
As highlighted by the COVID-19 pandemic, safety in the workplace is critical for dental healthcare workers (DHCW) and patients in the provision of care. Experts at the U.S. Centers for Disease Control and Prevention (CDC) and Occupational Safety and Health Administration (OSHA) provide evidence-based rules, recommendations, and guidelines designed to ensure workplace safety. This article will focus on the safety aspects related to preventing and managing infection control exposure incidents.
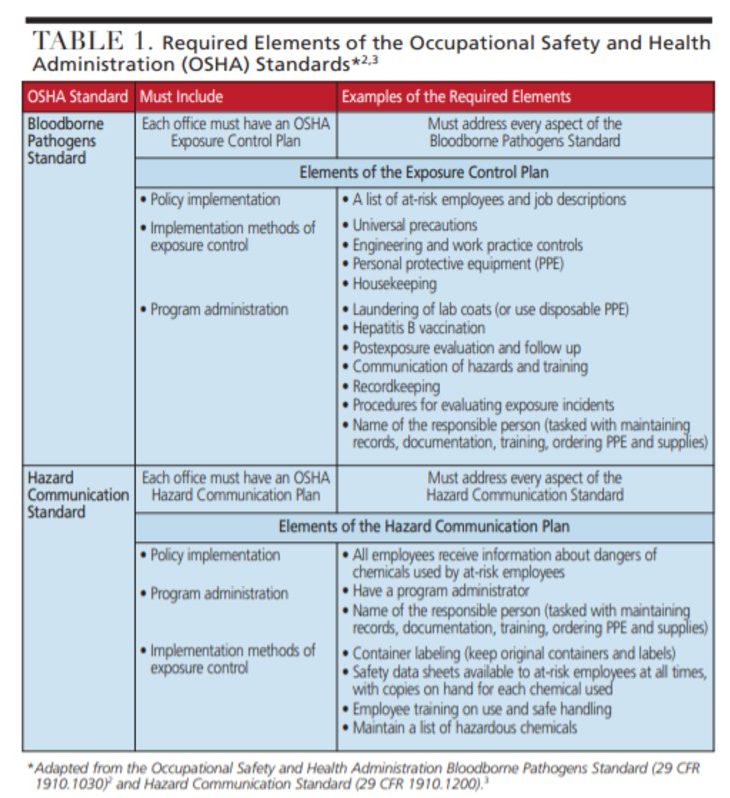
As a key regulatory agency for the workplace, OSHA was founded in 1971 in response to a rise in worker injuries and deaths. In 1970, nearly 14,000 workers were killed on the job.1 Under OSHA regulations, this number had fallen to approximately 4300 by 2009.1 Tasked with protecting workers, OSHA ensures workplace safety and has enforcement capability that allows it to investigate violations and impose fines. Two OSHA standards that dental offices must comply with include the OSHA Bloodborne Pathogens (BBP) Standard (29 CFR 1910.1030)2 and Hazard Communication Standard (29 CFR 1910.1200).3
Due to the risk of transmission in clinical settings, OSHA mandates protection for all DHCW exposed to bloodborne pathogens and other potentially infectious materials (OPIM), such as human immunodeficiency virus (HIV), hepatitis B virus (HBV) and hepatitis C virus (HCV).2 The BBP standard requires employers to provide employees with personal protective equipment (PPE), such as gloves, masks, protective eyewear, protective garments (e.g., lab coats — including their laundering), HBV vaccination, safe work practices (including safety devices, such as sharps containers and needle recapping devices), and annual training.2
Federal rules also mandate protection from chemical hazards in the workplace. Employers must provide training and PPE for employees who handle chemicals.3 Many types of chemicals are used in dentistry, including surface disinfectants, cleaning and housekeeping agents, and dental materials. Safety data sheets must accompany all chemicals and contain information about safe handling, including methods for storage, disposal, and first aid instructions in the event of accidental exposure. Current safety data sheet information must be kept for every chemical used and be accessible at all times.3
The CDC’s Guidelines for Infection Control in Dental Health-Care Settings — 2003 contains guidance on a wide range of personal health elements, including prevention of disease transmission, hand hygiene, PPE to prevent splash and spatter to the skin and mucous membranes (e.g., eyes, nose and mouth), sterilization, and several other special considerations.4 The CDC published companion documents in 2016 outlining its recommendations and basic expectations for safe care.5 Using the CDC guidelines, along with OSHA standards, will help DHCWs make sense of the expectations for safe care.
EXPOSURE CONTROL PLAN
Employers are responsible for having written policies and procedures that demonstrate compliance with OSHA standards, and these policies must be accessible to employees at all times.2 Known as an exposure control plan (ECP), the written policy should outline how various procedures will be performed safely in the office.2 Infection control policies may be added to this document to make it more convenient for staff. An annual review of OSHA and infection control policies and procedures is a key step in providing a safe environment for DHCWs and patients. Contents of the ECP include:2
- A listing of each job classification that has occupational exposure risk (to blood or OPIM); this document must also list the tasks performed by the employee
- A listing of the steps that are followed if an exposure to blood or OPIM incident occurs (i.e., a postexposure protocol)
- Policies and procedures for how the OSHA BBP standard is implemented in the office; this includes methods for complying with the HBV vaccination, record keeping, universal precautions, PPE, hand hygiene, housekeeping, handling of waste, hazard communication, engineering controls (i.e., safety devices), and work practice controls (i.e., safe work practices)
- Documentation by workers with occupational exposure risk of the annual identification, evaluation, and selection of engineering controls and work practice controls
- A record or documentation of information and training provided to employees
- The ECP must be reviewed at least annually with all at-risk workers, when new employees are hired, and when an employee takes on new tasks that involve risk for occupational exposure
- The plan must be updated whenever new policies, products, or procedures are implemented, and these updates must be communicated to all employees
TRAINING REQUIREMENTS
Employers must comply with specific OSHA training requirements.2 Training on the BBP standard must be made available to employees during their regular working hours at no charge. It also must be provided upon hire before any tasks involving occupational risk occur. Additionally, training must take place on an annual basis (minimally), and when any new tasks or procedures are implemented. The training material must be provided in a language and at a level the employee understands, and an opportunity to ask questions must be provided. The person conducting the training must possess knowledge of epidemiology, symptomology, modes of transmission of BBPs, vaccinations and postexposure prophylaxis. The trainer must also review the ECP and provide site-specific information on how the OSHA and infection control procedures are implemented in the facility. Components of an OSHA training session include:2
- A copy of the BBP standard, with an explanation related to that setting
- An explanation of the epidemiology, symptomology, and routes of transmission of BBPs
- A discussion about the ECP, its contents, and how employee can access the plan
- Information about the tasks that pose a risk for exposure to blood or OPIM
- Methods or procedures to reduce the risk of exposure to blood or OPIM
- Instruction regarding the engineering controls, work practice controls, PPE, and waste handling in the office
- Information on HBV vaccination (employee immunizations must be provided free of charge) and postexposure procedures if an exposure occurs
- Explanation of signs and labels used in the office
- The session must be interactive so employees can ask questions
REQUIRED RECORDKEEPING
Employers must keep records of OSHA compliance and training.2 The ECP must be kept up to date and reviewed at least annually. The employer must also keep a medical record on file for each employee with occupational risk. At minimum, the medical record must include:2
- The employee’s name and social security number
- A copy of the employee’s HBV vaccination status, including the dates of the vaccine
- A signed vaccine declination form if the employee decides not to receive the vaccine
- If an employee has an occupational exposure, copies of all results of medical testing, follow up, and the healthcare professional’s written opinion
- The medical record is confidential and should be kept in a locked cabinet and not disclosed to anyone without the employee’s permission
- The medical record must be kept for the length of the employment term, plus an additional 30 years
PREVENTING OCCUPATIONAL EXPOSURES
Occupational exposure incidents with blood or OPIM include percutaneous (sharps) injuries and splashes to mucous membranes or non-intact skin.4 Thankfully, exposure incidents in dentistry have declined over the past 30 years due to advances in infection prevention education, safer work behaviors, and safer devices.4 A 2012 study by Cleveland et al6 reported 6% of dentists experienced a percutaneous injury within the past year, as compared to 14.4% of staff members. The most common percutaneous injuries occur outside the mouth when handling sharp instruments (such as burs, needles, and blades).4 Needlesticks are the most common type of exposure incident, with the fingers being the most frequent area of exposure.7,8 A 2016 study by Alvear Fa and Cuny7 reported that 37% of all exposure incidents over a 10-year period in one dental school were due to needlesticks.
The best way to prevent exposure incidents is to avoid contact with blood and OPIM through the use of safety devices (i.e., engineering controls) and safe working behaviors (i.e., work practice controls).4 Engineering controls include needle recapping devices and sharps containers. Work practice controls include using a one-handed-scoop method for needle recapping, not passing a needle during four-handed dentistry, and the use of retraction devices versus finger retraction. Although further research is needed, Alvear Fa and Cuny7 reported a reduction in needlestick exposures with the use of dental mirrors as a retraction device.
POSTEXPOSURE PROTOCOLS
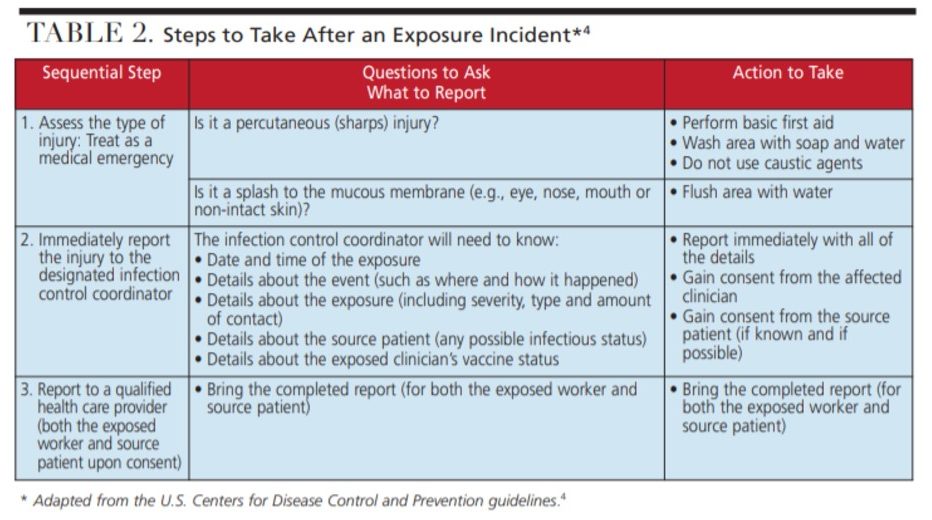
Exposure incidents should be treated as a medical emergency and evaluated by a qualified healthcare provider immediately after — or as soon as possible after — the exposure. The first step in handling an exposure incident is to perform basic first aid, such as flushing the area with water for mucous membranes and/or using soap and water to wash percutaneous injuries.4 It is the employee’s responsibility to immediately report the incident to the office’s designated infection control coordinator, safety officer or senior employee so postexposure management can be initiated quickly. This individual should draft a report about the incident, including the date and time of exposure, details about the incident and how it occurred, information about the severity of the exposure, whether the source material was known, and the exposed individual’s vaccination record. This report should accompany an exposed worker to an evaluation appointment with a healthcare provider who can assess the risk of exposure and recommend any further treatment or follow up.4 Despite safe work behaviors, exposure incidents occasionally occur, and DHCWs should not hesitate to report such incidents.
In the event of an exposure incident, the potential for transmission should be evaluated to include the type and amount of bodily fluid (blood or OPIM), the kind of exposure that occurred (percutaneous or mucous membrane splash), the infectious status of the source individual (patient), and the immune status or disease susceptibility of the exposed DHCW.4 The qualified healthcare professional will assess this information and make recommendations for further treatment or follow up, if necessary.4 Postexposure prophylaxis may be indicated if the medical provider deems necessary.
PROBLEMS WITH UNDERREPORTING
Several studies show underreporting of exposure incidents is common and could be problematic for postexposure management protocols and preventive measures.7,9,10 A 2019 investigation by Kan et al9 showed that underreporting of percutaneous injuries in a dental school setting was a problem, with only 21 of the 54 (38.9%) injuries studied being properly reported because students thought “it was only minor.” An investigation by Myers et al10 also noted failures in reporting exposures, as well as a lack of knowledge among dental students about postexposure protocols. The Myers study found that basic first aid was often skipped and reporting delayed, which likely impeded medical attention. Even among practicing clinicians, studies show that only about half (48.4%) of DHCWs with percutaneous injuries sought medical attention/referral and documented their injuries.6
CALL TO ACTION
Improvements in educating DHCWs about postexposure protocols and injury reporting are clearly needed. Delays in reporting and seeking treatment could be problematic and lead to disease transmission. The 2012 study by Cleveland et al6 offers helpful strategies to increase awareness and compliance in this area. The team’s suggestions include improving instruction in educational settings, requiring mandatory continuing education credits in infection control for licensure, the use of various modes of technology in teaching infection control (e.g., podcasts and webinars), and focus groups.
Dental professionals are routinely exposed to BBPs and OPIM in the provision of care. Disease transmission from BBPs carries potentially serious consequences. Being aware of OSHA requirements and CDC guidelines for the handling of exposure incidents is a necessary component of safe practice. Employing safety devices and following recommended protocols helps reduce the risk of exposure. Although exposure incidents are infrequent, it is imperative to know how to properly handle these situations when they arise.
KEY TAKEAWAYS
- Clinicians should receive regular training on ways to prevent and manage infection control exposure incidents.
- Dental offices must comply with the federal Occupational Safety and Health Administration (OSHA) Bloodborne Pathogens (BBP) Standard (29 CFR 1910.1030)2 and Hazard Communication Standard (29 CFR 1910.1200),3 which are designed to ensure safe practice by oral health professionals.
- Employers are responsible for having written policies and procedures that demonstrate compliance with OSHA standards.2
- Known as an exposure control plan, the written policy should outline how various procedures will be performed safely in the office.2 Infection control policies may be added to this document to make it more convenient for staff.
- The best way to prevent exposure incidents is to avoid contact with blood and other potentially infectious materials through the use of safety devices (i.e., engineering controls) and safe working behaviors (i.e., work practice controls).4
- In the event an exposure occurs, the incident should be treated as a medical emergency and evaluated by a qualified healthcare provider immediately after — or as soon as possible after — the exposure.
REFERENCES
- Occupational Safety and Health Administration. Timeline of OSHA’s 40 Year History. Available at: https://www.osha.gov/osha40/timeline.html. Accessed March 30, 2020.
- Occupational Safety and Health Administration. Bloodborne Pathogens Standard 29 CFR. 1910.1030. Available at: https://www.osha.gov/pls/oshaweb/owadisp.show_document?p_table=standards&p_id=10051. Accessed March 30, 2020.
- Occupational Safety and Health Administration. Hazard Communication Standard 29 CFR 1910.1200. Available at: https://www.osha.gov/laws-regs/regulations/standardnumber/1910/1910.1200. Accessed March 30, 2020.
- Kohn WG, Collins AS, Cleveland JL, et al. Guidelines for infection control in dental health-care settings — 2003. MMWR Recomm Rep. 2003;52(RR-17):1–61.
- U.S. Centers for Disease Control and Prevention. Summary of Infection Prevention Practices in Dental Settings: Basic Expectations for Safe Care. Available at: https://www.cdc.gov/oralhealth/infectioncontrol/summary-infection-prevention-practices/index.html. Accessed March 30, 2020.
- Cleveland JL, Bonito AJ, Corley TJ, et al. Advancing infection control in dental care settings: factors associated with dentists’ implementation of guidelines from the Centers for Disease Control and Prevention. J Am Dent Assoc. 2012;143:1127–1138.
- Alvear Fa B, Cuny E. Preliminary evidence supports modification of retraction technique to prevent needlestick injuries. Anesth Prog. 2016:63:192–196.
- Pasha L, Farid H, Faisal MR. Dental professionals experience regarding sharps injuries during practice. Pak Oral Dent J. 2016;36:455–459.
- Kan S, Ho V, Siddiqi A, Zafar S. The prevalence of percutaneous exposure incidents among staff and students treating pediatric patients. J Dent Children. 2019:86:81–87.
- Myers JE, Myers R, Wheat ME, Yin MT. Dental students and bloodborne pathogens: occupational exposures, knowledge, and attitudes. J Dent Educ. 2012:76:479–486.
The author has no commercial conflicts of interest to disclose.
From Decisions in Dentistry. June 2020;6(6):22–24.