 KATERYNA KON/SHUTTERSTOCK.COM
KATERYNA KON/SHUTTERSTOCK.COM
Presentation of Monkeypox in the Dental Setting
Understanding the origins of this viral disease and how it spreads will help oral health professionals advise patients and prevent cross-transmission during dental care.
This course was published in the December 2022 issue and expires December 2025. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
AGD Subject Code: 148
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Describe the etiology and presentation of monkeypox.
- List the vaccines available to prevent monkeypox.
- Explain appropriate infection prevention protocols to mitigate
possible cross-transmission during dental care.
A viral transmissible disease, monkeypox occurs chiefly in Central and Western Africa,1 where it is endemic to animals in these regions. The monkeypox virus belongs to the Orthopoxvirus genus in the Poxviridae family, whose other members include the variola virus (which causes smallpox), vaccina virus (which is used in the smallpox vaccine), and cowpox virus.2 It was first discovered in 1958 with two pox-like disease outbreaks in colonies of monkeys used for research.3 While called monkeypox, the exact animal of origin is unknown. African rodents and nonhuman primates, however, may carry the virus and infect people.3
There are two types of monkeypox virus: West African and Central African. The 2022 outbreak is from the West African type and is rarely fatal, as more than 99% of people who contract the West African type will survive.4 However, people with weakened immune systems, children younger than 8, individuals with a history of eczema, and pregnant and nursing women may be more likely to become seriously ill or die.4
The first human case of monkeypox was reported in August 1970 in a 9-month-old child living in a remote village in the Democratic Republic of Congo. Initially, smallpox was suspected,2 but further testing revealed the child had contracted the monkeypox virus. The baby’s parents reported occasionally eating monkeys as a delicacy, but could not recall if monkey was eaten prior to the outbreak or if the infant had encountered monkeys. Notably, the child was the only one in the family who had not received the smallpox vaccine.
The first known monkeypox virus infection in the United States occurred in the Midwest in 2003 in prairie dogs infected by rodents imported from Ghana in Western Africa. The rodents consisted of two giant pouched rats, nine dormice and three rope squirrels. Forty-seven cases were reported in six states: Illinois, Indiana, Kansas, Missouri, Ohio and Wisconsin. All those infected with the virus came in direct contact with the prairie dogs.5 In 2021, two cases were reported, one in July and one in November. Both involved Americans who traveled from Nigeria to the United States.5
Prior to 2022, all diagnosed cases of monkeypox outside of Africa were related to travel. As of presstime, the U.S. Centers for Disease Control and Prevention (CDC) reported 28,657 confirmed cases in the United States.6 The states with the highest number of reported cases were New York, California and Florida. It is not clear how these individuals were infected, but early data suggest that men who have sex with men account for a high number of cases.6 There is no clear link between the cases reported and travel from endemic countries and no link with infected animals.7
TRANSMISSION
The monkeypox virus is transmitted to humans during the handling of infected animals or by direct contact with the infected animal’s bodily fluids or lesions.1 The virus is spread person-to-person through direct contact with the infectious rash, scab or bodily fluids. Monkeypox can spread through respiratory droplets during prolonged face-to-face contact, such as those passed during kissing, cuddling or sex. It is also spread by touching items, such as clothes and bed linens, that previously touched the infectious rash or bodily fluids of an infected individual. Monkeypox can also be spread from a pregnant woman to her fetus. An individual can contract the disease by being scratched or bitten by an infected animal, as well as by preparing, eating, or using products from an infected animal.8
SIGNS AND SYMPTOMS
Infection with monkeypox virus causes lymphadenopathy, which typically occurs with fever onset, one or two days before rash onset, or, rarely, with rash onset. Swelling of the lymph nodes may be in the neck (submandibular and cervical), armpits (axillary) or groin (inguinal), and occur on both sides of the body or just one.9
After infection with the monkeypox virus, the incubation period typically lasts seven to 14 days — but incubation can also range from five to 21 days. Infected individuals do not have symptoms and are not contagious during this period. The development of an early set of symptoms (prodrome) includes fever, malaise, headache, sometimes sore throat and cough, lymphadenopathy and weakness. Infected individuals may sometimes be contagious during this period.
Shortly after the onset of prodrome, a rash appears. Lesions will first develop in the mouth and tongue (enanthem stage), and then on the body. They typically develop simultaneously and evolve together on any given part of the body. The evolution of lesions progresses through four stages — macular, papular, vesicular to pustular — before scabbing over and resolving. This process happens over a period of two to three weeks. The infected individual is contagious from the onset of the enanthem through the scabbing stage. Pitted scars and/or areas of lighter or darker skin may remain after scabs have resolved, after which an individual is no longer contagious.9
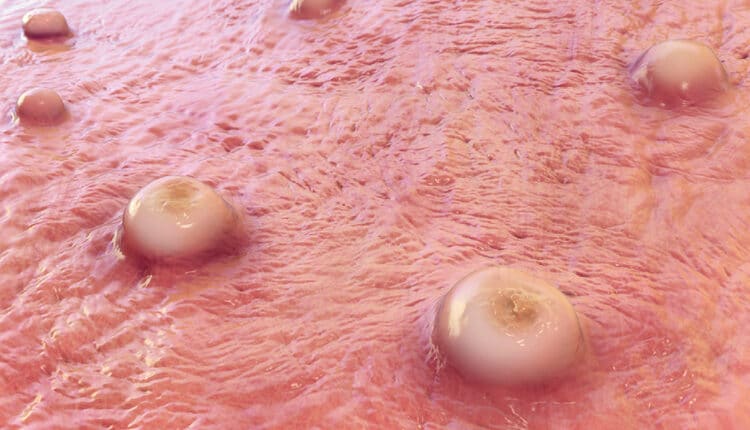
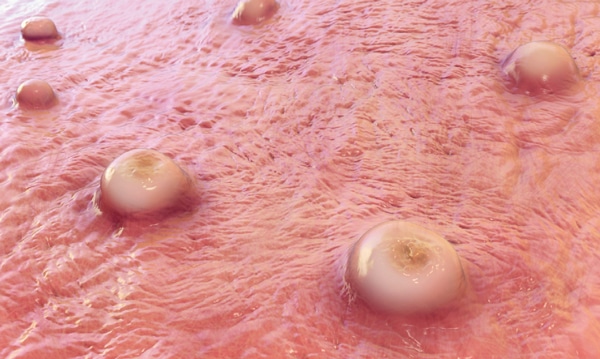
Monkeypox lesions are typically well circumscribed, deep seated, and often develop umbilication (Figure 1 and Figure 2). They are relatively at the same size and stage of development on a single site of the body, and most commonly present on the face, palms, and soles of the feet. Lesions are often painful until the healing phase — by the end of the second week — when they become itchy and form crusts.9 Table 1 describes the stages of monkeypox in more detail.

PREVENTION
To prevent contracting monkeypox, individuals should avoid contact with an animal or individual infected with the virus. Regularly cleaning hands with soap and water or an alcohol-based hand sanitizer, especially after encountering an individual or animal who is infected, is key. Avoid contact with clothes, bedsheets, towels and other items or surfaces touched by the rash or respiratory secretions of an infected individual. It is important to wash compromised clothes, towels, bedsheets and eating utensils with warm water and detergent. Clean and disinfect any contaminated surfaces and dispose of contaminated waste appropriately.10 Healthcare professionals can isolate infected patients from others who could be at risk for infection and should use personal protective equipment (PPE) when caring for patients.11
The current spread of monkeypox is through close, personal and often skin-to-skin contact, including sexual activity. The public should understand that the spread of monkeypox may occur at places such as raves, parties, clubs and festivals. Spaces where minimal or no clothing is worn and where intimate sexual contact occurs also have a higher likelihood of spreading the monkeypox virus.12
VACCINES
Because monkeypox is closely related to the virus that causes smallpox, the smallpox vaccine can protect against monkeypox if administered before exposure. Data from Africa suggest the smallpox vaccine is at least 85% effective in preventing monkeypox.11 Several vaccines to prevent smallpox also offer some protection. A newer vaccine developed for smallpox — MVA-BN, also known as Imvamune, Imvanex or JYNNEOS — was approved in 2019 for preventing monkeypox, but is not yet widely available.13 Individuals who have been vaccinated against smallpox will have some protection against monkeypox. The original smallpox vaccines are no longer available, and people younger than 50 are unlikely to have been vaccinated. Smallpox vaccination ceased in 1980 after smallpox was believed to have been eradicated.13
An antiviral medication developed to treat smallpox (tecovirimat) was approved by the European Medicines Agency for the treatment of monkeypox in January 2022.13 Tecovirimat (also known as TPOXX or ST-246) is approved by the U.S. Food and Drug Administration (FDA) for treating smallpox in adults and children. However, its use for other Orthopoxvirus infections, including monkeypox, is not yet FDA approved.14
In the United States, ACAM200 and JYNNEOS are the two vaccines approved to prevent smallpox. In certain groups, such as individuals with serious immune system problems, complications from ACAM2000 can be severe. This vaccine has the potential for more side effects and adverse events than the newer JYNNEOS vaccine. The ACAM2000 vaccine is administered as a live vaccinia virus preparation that is inoculated into the skin by pricking the skin surface. Following a successful inoculation, a lesion will develop at the site of the vaccination (i.e., a “take”). The virus growing at the site of this inoculation lesion can be spread to other parts of the body or even to other people. Individuals who receive vaccination with ACAM2000 must take precautions to prevent the spread of the vaccine virus and are considered vaccinated within 28 days.
Approved by the FDA for preventing monkeypox,10 JYNNEOS is administered as a live virus that is nonreplicating; vaccination consists of two subcutaneous injections four weeks apart. There is no visible “take” and, as a result, no risk for spread to other parts of the body or other people. Individuals who receive JYNNEOS are not considered vaccinated until two weeks after they receive the second dose of the vaccine.
The CDC recommends that exposed individuals receive the vaccine within four days from the date of exposure. If given between four and 14 days after the date of exposure, vaccination may reduce the symptoms of disease, but may not prevent it. The CDC also recommends that individuals exposed to monkeypox virus and who have not received the smallpox vaccine within the last three years should get vaccinated.11
In June 2022, Germany’s Standing Committee on Vaccination recommended that anyone exposed to monkeypox should receive two doses of the smallpox vaccine Imvanex. Those who have never received a smallpox immunization should receive two doses, 28 days apart, while one dose will suffice for those who have already been vaccinated against smallpox. Due to limited supply, Imvanex should be made available first to people who were exposed to the virus in the previous 14 days. The second priority group includes those at increased risk of contracting monkeypox, such as men who have sex with changing male partners, as more than 130 monkeypox cases have been diagnosed in Germany among this population.15
INFECTION PREVENTION MEASURES
Infectious and communicable diseases are a continuous occupational risk for oral health professionals and can lead to illness, disability and loss of work time. Dental practices must maintain the highest level of infection control to prevent disease transmission, and standard precautions should be applied for all patient care. Any patient suspected of having monkeypox should be directed to his or her primary care provider, health department clinic, or urgent care center for evaluation. Similar to other contagious lesions, dental care should be avoided until all lesions have crusted, those crusts have separated, and a fresh layer of healthy skin has formed underneath (Table 2).16
Since COVID-19, oral health professionals have become even more adept with infection prevention measures and use of PPE. The CDC’s Guidelines for Infection Control in Dental Health-Care Settings — 2003 provide comprehensive recommendations for preventing and controlling infectious diseases and managing the health and safety of providers and patients.17 Aerosol-producing procedures or equipment may resuspend dried material from lesions and should be avoided.16 Clinicians should wear gowns, gloves, eye protection, and KN95 or N95 respirators.
Monkeypox virus falls into the Tier 1 emerging viral pathogen category: an enveloped virus that is easiest to inactivate. Thus, high-level disinfectants are indicated for use. These agents damage the lipid envelope, resulting in the virus no longer being infectious.18 Clinicians can visit the U.S. Environmental Protection Agency’s website to determine which disinfectant is effective in destroying the emerging virus and follow the manufacturer’s directions for use.19
Clinicians can also use the CDC’s DentalCheck mobile application, developed directly from the agency’s Infection Prevention Checklist for Dental Settings: Basic Expectations for Safe Care, to create checklists to periodically assess protocols to ensure the facility meets minimum requirements for safe care.18 The infection control coordinator or compliance officer can use this annually or semi-annually to assess administrative policies and procedure.18
RESOURCES FOR CLINICIANS
The Advisory Committee on Immunization Practices (ACIP) recommends that individuals whose jobs may expose them to Orthopoxviruses, such as monkeypox, should receive either the ACAM2000 or JYNNEOS vaccine. This is known as pre-exposure prophylaxis (PrEP). Clinical laboratory personnel who perform testing to diagnose Orthopoxviruses, research laboratory workers who directly handle cultures or animals contaminated or infected with Orthopoxviruses, certain healthcare and public health response team members designated by public health authorities, and healthcare personnel who administer ACAM2000 or anticipate caring for patients with monkeypox should receive PrEP.14 At this time, most clinicians and laboratory workers in the United States who are not performing Orthopoxvirus testing to diagnose Orthopoxviruses, including monkeypox, are not advised to receive Orthopoxvirus PrEP.
The CDC, in conjunction with the ACIP, provides recommendations on who should receive smallpox vaccination in a nonemergency setting. Currently, vaccination with ACAM2000 is recommended for laboratory personnel working with certain Orthopoxviruses and military personnel. In 2021, the ACIP voted to recommend JYNNEOS PrEP as an alternative to ACAM2000 for individuals at risk for exposure to Orthopoxviruses.14
SUMMARY
All healthcare personnel should remain informed and current regarding the monkeypox virus and preventive measures to avoid transmission. Oral health professionals should be knowledgeable about infectious diseases and provide guidance to their patients. By maintaining the highest standard of infection prevention and care, and in accordance with CDC guidelines, dental teams can continue to effectively prevent the spread of transmissible diseases, such as monkeypox.
REFERENCES
- Weinstein RA, Nalca A, Rimoin AW, Bavari S, Whitehouse CA. Reemergence of monkeypox: prevalence, diagnostics, and countermeasures. Clin Infect Dis. 2005;41:1765–1771.
- Sklenovská N, Van Ranst M. Emergence of monkeypox as the most important orthopoxvirus infection in humans. Front Public Health. 2018;6:241.
- U.S. Centers for Disease Control and Prevention. About Monkeypox. Available at: https://www.cdc.gov/poxvirus/monkeypox/about/index.html. Accessed November 4, 2022.
- U.S. Centers for Disease Control and Prevention. Monkeypox: Frequently Asked Questions. Available at: https://www.cdc.gov/poxvirus/monkeypox/faq.html. Accessed November 4, 2022.
- U.S. Centers for Disease Control and Prevention. Monkeypox: Past U.S. Cases and Outbreaks. Available at: https://www.cdc.gov/poxvirus/monkeypox/outbreak/us-outbreaks.html. Accessed November 4, 2022.
- U.S. Centers for Disease Control and Prevention. U.S. Monkeypox Outbreak 2022: Situation Summary. Available at: https://www.cdc.gov/poxvirus/monkeypox/response/떖/index.html. Accessed November 4, 2022.
- World Health Organization. Monkeypox. Available at: https://www.who.int/news-room/fact-sheets/detail/monkeypox. Accessed November 4, 2022.
- U.S. Centers for Disease Control and Prevention. Monkeypox: How it Spreads. Available at: https://www.cdc.gov/poxvirus/monkeypox/transmission.html. Accessed November 4, 2022.
- U.S. Centers for Disease Control and Prevention. Monkeypox: Clinical Recognition. Available at: https://www.cdc.gov/poxvirus/monkeypox/clinicians/clinical-recognition.html. Accessed November 4, 2022.
- U.S. Centers for Disease Control and Prevention. Monkeypox: Prevention. Available at: https://www.cdc.gov/poxvirus/monkeypox/prevention.html. Accessed November 4, 2022.
- U.S. Centers for Disease Control and Prevention. Monkeypox and Smallpox Vaccine Guidance. Available at: https://www.cdc.gov/poxvirus/monkeypox/clinicians/smallpox-vaccine.html. Accessed November 4, 2022.
- U.S. Centers for Disease Control and Prevention. Social Gatherings, Safer Sex, and Monkeypox. Available at: https://www.cdc.gov/poxvirus/monkeypox/specific-settings/social-gatherings.html. Accessed November 4, 2022.
- World Health Organizations. Monkeypox: Questions and Answers. Available at: https://www.who.int/news-room/questions-and-answers/item/monkeypox. Accessed November 4, 2022.
- U.S. Centers for Disease Control and Prevention. Guidance for Tecovirimat Use Under Expanded Access Investigational New Drug Protocol during 2022 U.S. Monkeypox Cases. Available at: https://www.cdc.gov/poxvirus/monkeypox/clinicians/Tecovirimat.html. Accessed November 4, 2022.
- AP News. German Panel Recommends Vaccines After Exposure to Monkeypox. Available at: https://apnews.com/article/health-germany-smallpox-af95c70ee85d1463ef23b21f18d2477c. Accessed November 4, 2022.
- New York City Department of Health. Monkeypox (MPV). Available at: https://www.nyc.gov/site/doh/health/health-topics/monkeypox.page. Accessed November 4, 2022.
- Kohn WG, Collins AS, Cleveland JL, et al. Guidelines for infection control in dental health-care settings—2003. MMWR Recomm Rep. 2003;52(RR-17):1–61.
- U.S. Centers for Disease Control and Prevention. Infection Prevention Checklist for Dental Settings: Basic Expectations for Safe Care. Available at: https://www.cdc.gov/oralhealth/infectioncontrol/pdf/safe-care-checklist-a.pdf. Accessed November 4, 2022.
- U.S. Environmental Protection Agency. Disinfectants for Emerging Viral Pathogens: List Q. Available at: https://www.epa.gov/pesticide-registration/disinfectants-emerging-viral-pathogens-evps-list-q#search. Accessed November 4, 2022.
From Decisions in Dentistry. December 2022;8(12)34-36,39.




