Infection Control Policy for Dental Technologies
An effective infection control policy for dental technologies will ensure patient and practitioner safety during the provision of care.
Today’s oral health professionals work with a variety of rapidly changing technologies during patient care. Understanding how to manage technology in the dental setting requires knowledge and application of infection prevention guidelines, including manufacturer instructions for use. Knowledge of these instructions is a critical part of an effective infection control policy, because if not carried out in accordance with manufacturer recommendations, items could pose a risk of cross-transmission. Typical asepsis compliance failures regarding technology items include improper cleaning, disinfection and sterilization.
In light of bloodborne pathogens, such as the hepatitis B virus (HBV) and hepatitis C, disease transmission in dentistry has always been a concern. The emergence of human immunodeficiency virus (HIV) in the 1980s brought this issue to the forefront. The U.S. Centers for Disease Control and Prevention (CDC) and Occupational Safety and Health Administration (OSHA) provide evidence-based rules, recommendations and guidance regarding safety in the provision of care.
Oral health professionals should be well versed in CDC and OSHA guidelines when working with technology-related equipment. Two OSHA standards that dental offices must comply with are the OSHA Bloodborne Pathogen Standard and Hazard Communication Standard.1,2 The OSHA Bloodborne Pathogen Standard mandates protection for all dental team members who may be exposed to blood and other potentially infectious materials.1 Employers are required to provide protection for employees through the use of personal protective equipment, such as gloves, masks, protective eyewear and protective garments (e.g., lab coats and their laundering). In addition, they must offer HBV vaccination, and establish safe work practices, including safety devices (e.g., sharps containers) and annual training.1
The OSHA Hazard Communication Standard mandates protection from chemical hazards in the workplace. Employers must provide training and personal protective equipment for employees handling chemicals, including surface disinfectants, cleaning agents and dental materials.2 All chemicals must contain a safety data sheet that provides information on handling, including methods for storage and disposal, and first aid instructions in the event of accidental exposure.2
The CDC Guidelines for Infection Control in Dental Health-Care Settings — 2003 contain comprehensive guidance related to all aspect of infection prevention.3 The CDC published companion documents in 2016 outlining recommendations and basic expectations for safe care.4
PATIENT CARE ITEMS
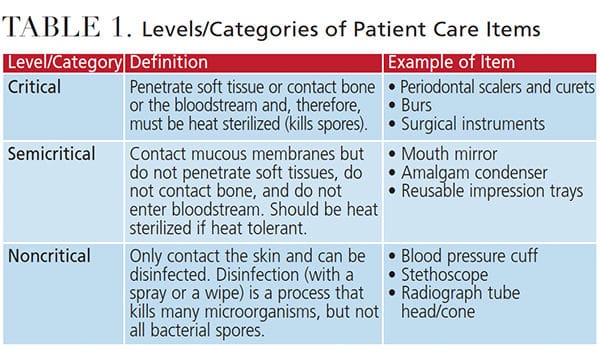
As oral health professionals, it is important to understand CDC guidelines when working with technology. Dental instruments are categorized into three levels related to sterilization: critical, semicritical and noncritical.3 Critical instruments (such as curets or burs) penetrate soft tissue or contact bone or the bloodstream and, therefore, must be heat sterilized. Semicritical instruments (e.g., mouth mirrors or amalgam condensers) contact mucous membranes, but do not penetrate soft tissues — however, if heat tolerant, they should also be sterilized. Table 1 outlines these categories. Sterilization refers to a process that kills all living microorganisms — including bacterial spores — through heat, gas or chemical methods.5 Noncritical items (e.g., X-ray tube heads or blood pressure cuffs) only contact the skin and can be disinfected. Disinfection with a spray or a wipe will kill many microorganisms, but not all bacterial spores.5
SURFACES AND EQUIPMENT
It is essential for providers to understand the instrument reprocessing cycle, as well as proper handling of surfaces in the operatory, many of which include technology-related equipment that may not be heat tolerant (i.e., sterilizable). There are two types of environmental surfaces in the dental operatory: clinical contact surfaces and housekeeping surfaces. The former include items frequently touched during treatment (e.g., instruments, equipment, switches and handles) that require sterilization or disinfection between patients, depending on the level of sterilization (critical, semicritical or noncritical). Housekeeping surfaces (such as floors and countertops) are not directly touched during treatment and require only routine cleaning.3
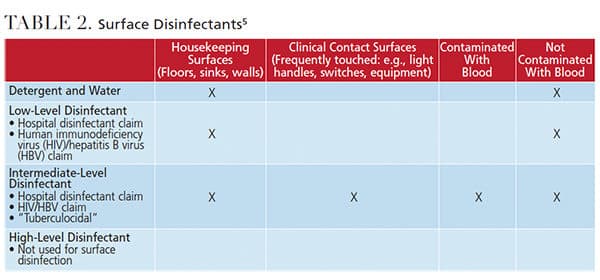
Surface disinfectants are used on both clinical contact and housekeeping surfaces. Many products are available and choosing one requires researching the three types of surface disinfectants: low level, intermediate level and high level (Table 2).3 Different types of surfaces in various settings require different levels of disinfection. For example, hospital rooms differ from dental operatories in which splash and spatter occur more frequently.
Low-level disinfectants carry a “hospital disinfectant” claim, meaning the product is used on noncritical housekeeping surfaces not contaminated by blood or other bodily fluids.3 Low-level disinfectants are effective against the test organisms Staphylococcus aureus, Salmonella and Pseudomonas, and are effective against HBV and HIV.3 Hospitals generally use low-level disinfectants on most surfaces because they are not generating aerosols and spatter.
Intermediate-level disinfectants also carry a “hospital disinfectant” claim and include the same characteristics as low-level disinfectants, but these agents are also effective against the hearty test organism Mycobacterium tuberculosis. This means it is considered “tuberculocidal” and is effective for use on items or surfaces contaminated with blood or other bodily fluids.3 It is wise to use intermediate-level disinfectants on all surfaces in the dental operatory because aerosols, blood, spatter and other fluids may not be visible. Using one product improves staff compliance and can be cost effective. As long as the label claims tuberculocidal activity, it is considered an intermediate-level disinfectant.
High-level disinfectants (such as glutaraldehyde) are not used on surfaces, but are indicated for use on semicritical items that cannot be heat sterilized.3
Surface disinfectants require cleaning or removing blood or other bodily fluids first by a spray-and-wipe or wipe-and-discard method.3 Next, the surface must be sprayed or wiped again and left wet or damp for the manufacturer’s recommended contact time (which varies). Failure to perform both steps results in surfaces that are not disinfected.
BARRIER Protection
Barriers made from impervious plastic are alternatives or adjuncts to surface disinfection. Barriers are placed on clean surfaces and changed between patients. The surface underneath does not need to be disinfected as long as the barrier was not compromised or visibly soiled.3,4 Barriers are one-time-use items recommended for difficult-to-clean items (such as chair controls, radiograph tube heads and exposure buttons).3 A combination of barriers and surface disinfection are often recommended by manufacturers of technology-related equipment.
CHALLENGES
Making sense of infection prevention guidelines with electronic devices is challenging. Adequate knowledge regarding the safety of each product is critical. As noted, following manufacturer instructions for use and their updates for each device are an important part of an effective infection control policy. These should provide detailed instructions on the cleaning and maintenance required to safely protect the device and abide by asepsis protocols. Striking a balance between proper infection control and protection of the device is key.
The U.S. Food and Drug Administration also provides suggestions on the labeling and instructions for use of medical devices.6 It asserts that instructions should include details on disassembly, cleaning, rinsing, disinfection/sterilization, drying, reassembly of the device, and use of disposable sheathing for other portions of the device. These processes may be unique to the brand used in the dental office.
SEMICRITICAL DEVICES
Each device has its own instructions regarding sterilization or disinfection, and oral health professionals should stay abreast of updates to infection control guidelines. While heat sterilization is required for semicritical items whenever possible, many devices have a combination of critical and noncritical components that cannot withstand heat sterilization.
Computer Mouse and Keyboard: Electronic health record management necessitates the frequent use of mice and keyboards, raising the risk that they will become contaminated. Reynolds et al7 found that 80% of infections occurred due to cross-contamination of hand-to-hand contact and other object-to-hand contact. Al-Ghamdi et al8 determined that up to 91% of office mice and keyboards were contaminated. Hartmann et al9 found 26 contaminated samples from keyboards and mice, compared with 18 contaminated samples from other fomites in patient rooms. The keyboard and mouse contain the majority of microorganisms and pose greater risks for cross-contamination.9 They represent easily contaminated surfaces and should have barriers placed before and after use, and they must be disinfected between uses.
Care must be taken when disinfecting mice and keyboards to avoid electrical shock and maintain the electrical components, but disinfectants are effective for this application. Rutala et al10 found that after testing six disinfectants (all containing chlorine, alcohol or phenol, and quaternary ammonium) “all disinfectants, as well as sterile water control, were effective at removing or inactivating more than 95% of test bacteria.” Disinfection did not damage the mice or keyboards in this study.10 Using disinfectants per manufacturer instructions regarding the correct contact time, and avoiding saturating or soaking the delicate electronic components are critical to maintaining equipment while adhering to proper infection control policy.
Radiography Equipment: Plastic barriers should be used — along with a disinfectant containing less than 17% alcohol — on handheld radiography units.11 A study by Hokett et al12 reported contamination rates of up to 51% underneath barriers covering digital sensors. Malta et al13 also demonstrated that disinfection of X-ray sensors and components were not common practice in dental offices; this report was based on 78 intraoral and extraoral X-ray systems that tested positive for cocci, Gram-negative bacilli and fungi. Intraoral cameras are used in similar fashion, suggesting they pose the same cross-contamination risk. Digital sensors and intraoral cameras should be barrier protected, followed by the use of an intermediate-level disinfectant,4 and wiping the intraoral camera lens with gauze and isopropyl alcohol.14,15
Handpieces: Some cordless handpieces have a removable head, and heat sterilization is recommended after each use. The remaining parts of the unit should be disinfected with an intermediate-level disinfectant. Handpieces connected to the dental unit air line and waterline should be heat sterilized between uses.3
Lights, Lasers and Other Devices: Near-infrared transillumination lights that use electromagnetic technology to create images are gaining popularity.16 One product has a plastic guiding sheath that is heat tolerant and autoclavable. The rest of the unit must be disinfected with an intermediate-level disinfectant, and the direct lens can be wiped with isopropyl alcohol and gauze.16 Another caries detection device uses a low-powered pulsating laser with a disposable plastic tip and sleeve that allow the laser to move through the plastic and not affect the quality of the light. The handle may be disinfected with an intermediate-level disinfectant approved for use on plastics. A computer aided design/computer aided manufacturing device that captures images to produce “same-day crowns” is frequently found in dental practice. The mirror sleeve is removable and heat tolerant (i.e., autoclavable). The camera body itself should be disinfected by wiping with an intermediate-level disinfectant (as opposed to spraying).17 Handheld oral cancer screening devices or scopes are used in the detection of oral cancer and abnormal lesions. These may be offered with single-use lens caps, and the rest of the unit can be disinfected with an intermediate-level disinfectant.
Cordless endodontic obturation systems combine a downpack heat source with a backfill extruder,18 and disposable or autoclavable tips, custom plastic sleeves, and intermediate-level disinfection are recommended. Curing lights require unique barriers specific to the manufacturer’s recommendations to ensure light quality and curing capability. The use of any intermediate-level disinfectant is acceptable. To avoid electrical shock, all devices connected to a wall outlet must be unplugged prior to any disinfectant application, and none of these devices should be directly sprayed with any disinfectant.
BEST PRACTICES
Impervious plastic barriers are effective in preventing cross-contamination of devices. The 2003 CDC guidelines emphasize that barriers are effective for difficult-to-clean items, such as electronic devices.3 Barriers alone might not be adequate to prevent cross-contamination, however, and may provide clinicians with a false sense of protection of semicritical items. A study by Choi19 found perforation rates of 44% to 51% after use of a single, 0.04-mm-thick plastic barrier during intraoral digital sensor use. This warranted further disinfection with an intermediate-level disinfectant. The use of a 0.08-mm plastic barrier provided a superior perforation rate, although the thickness irritated soft tissues.19 Thicker barriers make capturing digital images more difficult; in addition, they may inhibit the device’s effectiveness and increase patient discomfort.19 Imaging that depends on clear, direct contact with the tissues or teeth cannot be compromised due to the barrier, so thinner barriers are advised. Barriers are recommended for all semicritical devices, and the most effective thickness is a low-density polyethylene.19 Choosing the correct barrier with consideration to patient comfort is vital.
A combined approach including the use of barriers and intermediate-level disinfection appears to be best practice with regard to technology-related equipment. The keys to an effective infection control policy are proper training and compliance with manufacturer instructions for use. The protection and maintenance of expensive equipment are essential to longevity and function; however, clinicians must keep the asepsis of each type of equipment in mind and follow prudent infection control guidelines. The Organization for Safety, Asepsis and Prevention provides excellent resources to help practitioners adhere to CDC guidelines and OSHA regulations. With the goal of ensuring patient and provider safety, oral health professionals may want to visit the “Frequently Asked Questions” section at osap.org for additional information.
REFERENCES
- Occupational Safety and Health Administration. Bloodborne Pathogens Standard 29 CFR. 1910.1030. Available at: osha.gov/pls/oshaweb/owadisp.show_document?p_table=standards&p_id=1005 Accessed September 13, 2018.
- Occupational Safety and Health Administration. Hazard Communication Standard 29 CFR 1910.1200. Available at: osha.gov/pls/oshaweb/owadisp.show_document?p_table=standards&p_id=10099. Accessed September 13, 2018.
- Kohn WG1, Collins AS, Cleveland JL, et al. Guidelines for infection control in dental health-care settings — 2003. MMWR Recomm Rep. 2003;52(RR-17):1–61.
- U.S. Centers for Disease Control and Prevention. Summary of Infection Prevention Practices in Dental Settings: Basic Expectations for Safe Care. Available at: cdc.gov/oralhealth/infectioncontrol/guidelines/index.htm. Accessed September 13, 2018.
- Miller CH, Palenik CJ. Infection Control and Management of Hazardous Materials for the Dental Team. 4th ed. St. Louis: Mosby, Elsevier; 2010.
- U.S. Food and Drug Administration. Reprocessing Medical Devices in Health Care Settings: Validation Methods and Labeling by the Food and Drug Administration. Available at: fda.gov/downloads/medicaldevices/deviceregulationguidance/guidancedocuments/ucm25 3010.pdf. Accessed September 13, 2018.
- Reynolds KA, Watt PM, Boone SA, Gerba CP. Occurrence of bacteria and biochemical markers on public surfaces. Int J Environ Health Res. 2005;15:225–234
- Al-Ghamdi AK, Abdelmalek MA, Ashshi AM, Faidah H, Shukri H, Jiman-Fatani AA. Bacterial contamination of computer keyboards and mice, elevator buttons and shopping carts. African J Microbiology Res. 2011;5(23):3998–4003.
- Hartmann B, Benson M, Junger A, et al. Computer keyboard and mouse as a reservoir of pathogens in an intensive care unit. J Clin Monit Comput. 2004;18:7–12.
- Rutala WA, White MS, Gergen MF, Weber DJ. Bacterial contamination of keyboards: efficacy and functional impact of disinfection. Infect Control Hosp Epidemiol. 2006;27:372–377.
- NOMAD. Frequently Asked Questions: FAQ Portable X-Ray. Available at: http://aribex.com/support/faq-portable-x-ray/. Accessed September 13, 2018.
- Hokett SD, Honey JR, Francisco R, Baisden MK, Hoen MM. Assessing the effectiveness of direct digital: radiography barrier sheaths and finger cots. J Am Dent Assoc. 2000;131:463–467.
- Malta CP, Damasceno NL, Ribeiro RA, Silva CSF, Devito KL. Microbiological contamination in digital radiography: evaluation at the radiology clinic of an education institution. Acta Odontal Latinoam. 2016;29:239–247.
- Schick by Sirona. Cleaning and Disinfecting Sensors. Available at: schickbysirona.com/items.php?itemid=7669. Accessed September 13, 2018.
- Schick by Sirona. Cleaning and Disinfecting Cameras. Available at: schickbysirona.com/items.php?itemid=8749. Accessed September 13, 2018.
- Dexis. CariVu User Manual and Manufacturer’s Guidelines. Available at: dexis.com/carivu. Accessed September 13, 2018.
- Sirona. Omnicam User Manual. Available at: http://manuals.sirona.com/en/digital-dentistry/cerec-chairside-solutions/cerec-ac-with-cerec-omnicam.html. Accessed September 13, 2018.
- Patterson Dental. System B™ Cordless Obturation System — Cordless Fill Device. Available at: pattersondental.com/Supplies/ItemDetail/071137363. Accessed September 13, 2018.
- Choi JW. Perforation rate of intraoral barriers for direct digital radiography. Dentomaxillofac Radiol. 2015;44:20140245.
Featured image by WAKILA/ISTOCK/GETTY IMAGES PLUS
The authors have no commercial conflicts of interest to disclose.
From Decisions in Dentistry. October 2018;4(10):17–19.