Infection Control in Austere Practice Settings
Whether providing care in a field operatory or urban weekend clinic, clinicians must ensure that asepsis standards are met.
Oral health professionals are volunteering in increasing numbers as a way to give back to the community. In giving back, these clinicians are helping improve access to dental care for underserved populations and those who cannot afford routine care due to economic circumstances.1 This article will address some of the challenges of practicing outside of traditional settings while still adhering to the standard of care expected in the United States and as allowed by state licensure. Nontraditional settings include austere environments, such as a pavilion in Central America or a large-scale free dental clinic in an urban setting. Although the focus is on austere conditions, these principles apply to all settings because infection prevention is a key goal in all types of practice.
One word commonly used to describe austere practice settings is “mission” — whether the effort is underwritten by a religious group, professional organization, corporate sponsor or military operation. Volunteering to provide dentistry to underserved populations allows clinicians to do the most good for the most patients and deliver care not otherwise available. It also provides a chance to perform a high volume of work without regard for whether the patient can pay. Sometimes, there is an opportunity to do procedures not normally done in the clinician’s private practice (extractions, for example) by working with a mentor.
Practicing with seasoned colleagues allows the mission to be a learning experience, and return visits to similar locations will allow clinicians to follow up with local providers to see if any of their patients experienced postoperative complications or infections. In order to guard against the chance of postop infections, strict adherence to infection control principles must be maintained, along with an assurance that sterile instruments are used during treatment.1–3 Overcoming these challenges and adhering to the U.S. standard of care can go a long way in promoting good will, as the volunteer professionals intend.
PRE-TRIP PLANNING
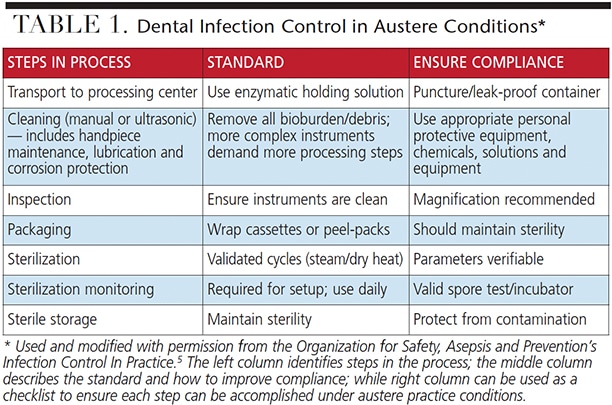
The first step in ensuring that infection control principles are followed is to plan the trip with infection control as the foundation of all aspects of care (Table 1). As an example, the type of dentistry anticipated often includes extractions. The standard of care is to use sterile instruments for each new patient encounter.4 Depending on the number of patients expected to be seen (usually between four and 10 per hour, per provider), the volume of instruments needed quickly adds up. Assuming that each extraction will require a minimum or four or five instruments, a clearer picture of why instrument processing and infection control should be a foundational element comes into focus.
Common instrument processing steps including transport of contaminated instruments, cleaning, decontamination and maintenance, inspection (for cleanliness, corrosion and breakage), packaging (Figure 1), sterilization (Figure 2), sterilizer monitoring and sterile storage. Each aspect should be considered in pre-trip planning.5
An initial decision as to whether to use instruments in kits or in loose groups held together in a sterilization pouch is a good starting point. This will begin to account for the workflow, personnel needed and packaging requirements (e.g., cassettes, wraps or bags); it will also dictate what types of bins will be needed to safely transport instruments from the operative area to the sterilization area.
It is also important to plan how instruments will be cleaned prior to sterilization. Will the process be automated, assisted with ultrasonics, or be done totally by hand? Unless the location is adjacent to a modern medical facility with adequate instrument processing capacity, automated washers will probably not be available. Instruments will most likely be manually cleaned, but ultrasonic cleaning will be a significant advantage over hand scrubbing alone. Ultrasonic cleaners are relatively simple to operate and lightweight; in addition, solutions or tablets can be used to enhance the ultrasonic action to better remove and neutralize the bioburden.
Space is limited on most dental missions; therefore, planning to use only a small amount of space is highly beneficial. This is another area in which basic principles apply. One-way workflows and low personnel traffic are key planning concepts to consider.6 Instrument drop-off near the ultrasonic unit will reduce instrument handling and the time personnel will spend in heavy utility gloves in hot environments. A space between the ultrasonic cleaner and sterilizer will be useful for inspecting instruments for cleanliness, corrosion and breakage, and for packaging instruments to go into the sterilizer. A sink located in this space will be useful for hand scrubbing instruments and rinsing instruments when necessary. Packaging helps instruments maintain sterility while stored in less-than-ideal conditions, such as high dust and humidity.7 Processing packaged instruments works best when using steam sterilization, dry heat or ethylene oxide.7–9
CONSIDER THE CLINICAL ENVIRONMENT
Sterilization is one of the most critical decisions affecting overall mission success. In the planning stages, consider how the sterilizer or sterilization method will perform in the chosen environment. Will the electrical power be adequate, or will an extra generator be needed for sterilization? Will the team have access to the necessary water (remembering that distilled water is not available in all countries)? Planning to use the simplest available means of sterilization is best. If only solid instruments are taken for treatment (e.g., scalers, forceps, elevators and retractors), dry-heat sterilization may be adequate — although most dry-heat sterilizers utilize long sterilization cycles (unless forced air circulation chamber technology is used). Regarding water quality, water purifiers/distillers may satisfy the requirements of some tabletop steam sterilizers. Whatever method is chosen, there must be a validated cycle that meets the instrument processing instructions.
It is advisable to practice instrument processing prior to the mission to make sure the equipment works with the power and water systems being used. Several full sterilization loads and spore tests should be completed to verify the system is functioning properly. Depending on mission timing and the number of instruments needed, the operation may require two or three sterilizers, several more water distillers and enough generator power to run them all — including all other dental equipment. Sterilization monitoring and spore testing will be necessary, too. The proper incubator for the spore test is essential, and rapid read-out tests will allow standards to be met for sterilization with minimum wait time (three negative spore tests are required to put any sterilizer into service after relocation or major repair).4

If the scope of the mission includes operative dentistry or endodontics, it introduces a new dimension of instrument processing — specifically, handpieces and dental units (Figure 3). Compared to solid metal instruments, handpieces present a more complex challenge in terms of sterilization and maintenance.10 Dental units pose their own list of requirements, such as waterline disinfection and maintenance, as well as suction decontamination and maintenance. The internal workings of a handpiece require a sterilization medium that penetrates the internal lumens and moving parts that generate the spin and torque of the bur. Because handpieces are heat tolerant, and liquid sterilants cannot be monitored like heat sterilization, it is not appropriate to sterilize handpieces using cold sterilization methods, such as glutaraldehyde.11 Steam and forced air dry heat are the most efficient ways to sterilize handpieces, and these methods can be accomplished in less than an hour per load in most cases. Many handpiece manufacturers’ instructions for use recommend specific methods for decontamination, cleaning and sterilization. This is another opportunity to plan ahead. In order to render handpieces (including slow-speed motors) sterile prior to use, these instructions must be followed between every patient.
When the mission is expected to include operative dentistry or endodontics, cassettes might be the most effective way to maintain an efficient workflow. This presents the opportunity to place wedges, matrix bands, gauze or endodontic files in the kit prior to sterilization (provided the instructions allow it). It is important to plan how many kits will fit into the sterilizer, along with some peel-packs or other packages, as this will dictate the number of patients that can be seen per hour or day. It will also dictate the number of kits required in anticipation of the number of patients to be treated. Rather than merely recording the high numbers of procedures and patients treated, after-action reports should document how the operation met its goals for infection control, safety and ethical standards.
STRIKE A BALANCE
Mission teams must strike a balance between the number of patients treated (e.g., 10 patients per hour: five operative and five extractions) and the volume of materials and equipment that can be staged or shipped to the location. In this example, if all 10 kits fit into one sterilization cycle that takes 45 minutes, in order to clean and package instruments and see the next hour’s patients, the processing cycle may take up to 90 minutes. Using this calculation, 15 operative and 15 extraction kits would be needed in order to continually see patients through an 8-hour day. In addition, there are advantages and disadvantages in expanding the scope of care if the intention is to move the practice location every day (or every several days). Moving too much equipment — either overnight or during one of the workdays — may limit the number of clinical days or severely stress the team. It is critical to plan for a reasonable amount of work each day, taking into account the fitness of the dental team.
ADDITIONAL CONSIDERATIONS
Other aspects of infection control to consider include consumable supplies, personal protective equipment, and staff protection and vaccinations. Consumable supplies will most easily be shipped in bulk and prepackaged onsite. Single-use packs are convenient, but may generate excess refuse compared to an aseptically packaged kit in a small plastic bag. This approach also makes it easier to customize the kit for the dental team and reduce the amount of extra materials in the kits. Single-patient burs are efficient for use in nontraditional settings, as these are available in small volume packages, can be separated for each patient and will remain sterile until use. In similar fashion, the volume of single-use anesthetic needles and anesthetic cartridges must be adequate for the number of patients anticipated (and packing extra needles and anesthetic is a wise idea). It is also crucial to make sure the anesthetic is not expired and stored according to the manufacturer’s recommendations.
Personal protective equipment for dental treatment and instrument processing should be used during mission dentistry, just as it is in traditional practice settings. Adequate supplies of scrubs or gowns, exam gloves, safety glasses (for patients and providers), masks and hair covers should be considered in mission planning.4 It is equally important to ensure that all staff are protected with the needed vaccinations for local endemic diseases and common outbreaks that may include hepatitis A, hepatitis B, typhoid and cholera. Malaria prophylaxis and mosquito netting may also be required to keep the mission team safe.12 Special consideration should be given to a contingency plan for postexposure prophylaxis for human immunodeficiency virus or hepatitis C in case of bloodborne pathogen-contaminated exposures or needlesticks. A competent medical authority to counsel and provide the needed medications within a few hours is the standard of care.13 This may be accomplished through some form of electronic messaging and telemedicine, but the inventory should include an adequate supply of the required medications.
Team dynamics, respect and consideration of staff members are critical elements of a successful mission. For example, while the leader may be enthusiastic to work a 12- or 14-hour day to treat as many patients as possible, due thought must be given to the many support functions that must be accomplished after treatment hours, such as set up for the next day or sterilization of more complex devices. It may only be possible to work eight hours and allow the entire team to fully recover for the next day’s work.
Many organizations, recommendations and checklists are available to assist dental professionals who wish to volunteer their time, and any first trip should probably be done as part of an experienced team that is familiar with the location and can provide care in a safe fashion. Once a few trips’ worth of experience is gained, expansion into other locations or new scopes of practice can be initiated. A thorough understanding of the logistics of mission dentistry — even with a narrow time frame and practice scope — is critical to any undertaking. Being able to provide safe care will add to the experience and sustain the concept of “first, do no harm.”
SUMMARY
Although the focus has been on dentistry in austere environments, the basic principles and concepts of infection control and instrument processing are easily applied to routine practice settings. Is the office layout designed and planned to support an efficient workflow? Are instrument-processing protocols strictly adhered to? Is there adequate space for cleaning, inspection, packaging and sterilization? Is the storage area designed to keep instruments sterile until they are used? Is sterilization monitoring part of routine practice? In light of these points, it is advisable to regularly reexamine the office’s infection prevention protocol and performance. At minimum, an annual evaluation will allow dental teams to see if there are efficiencies or opportunities to make the practice safer for patients and staff.
In conclusion, it should be obvious that infection control, instrument processing and adherence to standard practices are essential to any clinical environment. The key to the safe and successful provision of care in austere conditions is planning. This includes setting an achievable schedule, having the right equipment and supplies, caring for staff members and working as a team. Volunteering to provide dentistry in an austere environment is a rewarding experience. Dental teams can make it even more rewarding by adhering to strict ethical and infection control principles, knowing that the services provided to these patients pose a low risk of postoperative complications or infection.
KEY TAKEAWAYS
- Volunteering to provide dentistry to underserved populations allows clinicians to do the most good for the most patients and deliver care not otherwise available.
- In order to guard against the chance of pathogenic transmission or postoperative infections, strict adherence to infection prevention principles must be maintained when providing care in nontraditional settings.
- Space is limited on most dental missions; therefore, planning to use only a small amount of space is highly beneficial.
- It is advisable to practice instrument processing prior to the mission to make sure all the equipment works with the power and water systems being used.
- Team dynamics, respect and consideration of staff members are critical elements of a successful mission.
REFERENCES
- American Dental Association Foundation. International Dental Volunteer Organizations. Available at: internationalvolunteer.ada.org. Accessed July 11, 2017.
- Sfikas PM. Volunteering your services. J Am Dent Assoc. 1999;130:278–280.
- Han JS, Quinonez CR. Dental volunteerism: Is the current model working? J Can Dent Assoc. 2013;79:69.
- U.S. Centers for Disease Control and Prevention. Guidelines for Infection Control in Dental Health-Care Settings — 2003. Available at: cdc.gov/mmwr/preview/mmwrhtml/rr5217a1.htm. Accessed July 11, 2017.
- Organization for Safety, Asepsis and Prevention. Establishing a high quality instrument processing program: General aspects of instrument processing. Infection Control in Practice. 2017;16:1
- Miller CH, Palenik C. Infection Control and Management of Hazardous Materials for the Dental Team. 6th ed. St. Louis: Mosby; 2017:133.
- Association for the Advancement of Medical Instrumentation. ANSI/AAMI ST79, A1, A2, A3, A4 Consolidated Text: Comprehensive guide to steam sterilization and sterilization assurance in health care facilities. Available at: my.aami.org/aamiresources/previewfiles/ST79_Wa4_1310_preview.pdf. Accessed July 11, 2017.
- Association for the Advancement of Medical Instrumentation. ANSI/AAMI ST55:2010: Comprehensive guide to steam sterilization and sterilization assurance in health care facilities. Available at: my.aami.org/aamiresources/previewfiles/ST55_1014_preview.pdf. Accessed July 11, 2017.
- Association for the Advancement of Medical Instrumentation. ANSI/AAMI ST50:2004: Dry heat (heated air) sterilizers. Available at: webstore.ansi.org/Previews/PREVIEW_ANSI+AAMI+ST50-2004+(R2010).pdf. Accessed July 11, 2017.
- Parker HH, Johnson RB. Effectiveness of ethylene oxide for sterilization of dental handpieces. J Dent. 1995;23:113–115.
- Miller CH, Cleaning, sterilization, and disinfection: Basics of microbial killing for infection control. J Am Dent Assoc. 1993;124:48–57.
- Organization for Safety, Asepsis and Prevention. Guide for Safety and Infection Control for Oral Healthcare Missions. Available at: c.ymcdn.com/sites/osap.site-ym.com/resource/resmgr/Docs/dental_aid_guide.pdf. Accessed July 11, 2017.
- Kuhar DT, Henderson DK, Struble KA, et al. Updated U.S. public health service guidelines for the management of occupational exposures to human immunodeficiency virus and recommendations for postexposure prophylaxis. Infec Control Hosp Epidemiol. 2013;34:875–892.
The author has no commercial conflicts of interest to disclose. The views expressed are those of the author and do not reflect the official views or policy of the U.S. Department of Defense or its components.
From Decisions in Dentistry. August 2017;3(8):26–29.