 SOPHIE WALSTER/ISTOCK/GETTY IMAGES PLUS
SOPHIE WALSTER/ISTOCK/GETTY IMAGES PLUS
Impact of Self-Care on Periodontal Therapy Outcomes
An investigation of patient self-care performance in periodontal case management.
This course was published in the January 2021 issue and expires January 2024. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Discuss the authors’ proposed approach to case management and periodontal treatment for the patient described in this report.
- Explain the concept of cause-related therapy, and the clinical benefits of complying with individualized self-care recommendations.
- Describe the suggested use and effects of diode lasers as an adjunct in nonsurgical periodontal therapy.
Even in the presence of periodontal and peri-implant disease, motivated patients who demonstrate appropriate self-care can positively influence their oral health conditions, the prognosis of the case, and, above all, achieve clinical improvements leading to successful outcomes. While dentists can delegate auxiliary staff to teach personalized home hygiene methods, it is important during the first visit for dentists to explain the importance of following the personalized recommendations for effective biofilm removal, and the need to adhere to the prescribed frequency of recall visits.
Especially in complex periodontal cases, it is important to communicate that if patients agree to recommended etiological therapy and meticulously follow prescribed self-care routines, improvement of their periodontal condition is highly likely. However, this does not exclude the possible need for surgical management to achieve the therapeutic objectives and a stable clinical condition. After all, the human oral cavity hosts a microbial biofilm primarily composed of bacteria, as well as viral components.1,2 Today, as never before, we understand the potential pathogenic role of viruses and bacteria.
CASE REPORT
This case report provides evidence of how adhering to individually tailored oral hygiene recommendations can help improve the clinical outcome of periodontal therapy. Following a comprehensive periodontal evaluation in 2013, a 48-year-old female was diagnosed with Stage III, Grade B generalized periodontal disease. In order to mediate moderate to severe biofilm-induced inflammation, cause-related therapy (or initial preparation) was proposed to the patient.3–7 Cause-related therapy is the most appropriate term to describe treatment intended to remove the primary etiologic factor — in this case, bacterial biofilm.

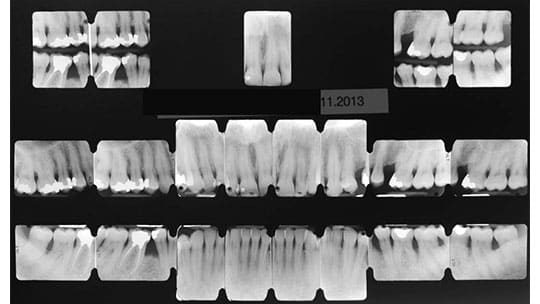
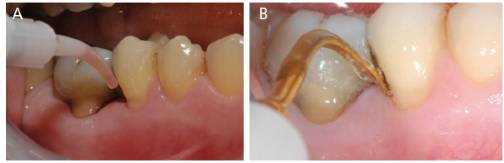
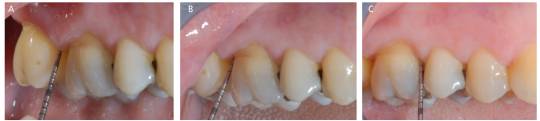
Commonly, three appointments of full-mouth periodontal instrumentation are required (Figure 1). The initial appointment is allotted two hours to conduct a complete radiographic evaluation (Figure 2), establish a relationship with the patient, enhance motivation to comply with individualized self-care strategies, and perform debridement. The remaining visits during this early stage of therapy are allotted one hour each.
Following circumferential probing, 980-nm diode laser-assisted periodontal nonsurgical instrumentation was performed throughout the oral cavity at three closely scheduled appointments.
The purpose of laser-assisted therapy is to decontaminate the subgingival sites and weaken the chemical bond between the calcified deposits and root surface, thus facilitating subsequent mechanical instrumentation.3 In addition, the laser provides an analgesic effect prior to subgingival instrumentation.8,9 Specifically, use of the diode laser results in reversal of the sodium-potassium pump on all cell membranes, which induces pulpal analgesia for approximately 20 minutes.8
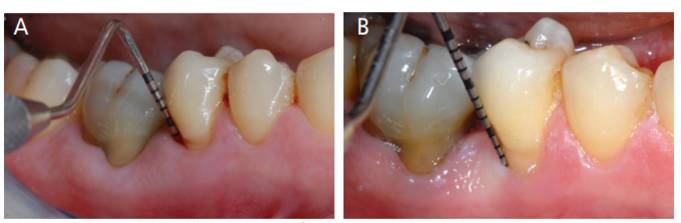
The laser optic fiber is thinner than any periodontal probe, enhancing effective calculus detection; this allows the clinician to collect data on the distribution, morphology and magnitude of calcified deposits. Based on the needs of each site, the clinician is able to plan subsequent instrumentation in a highly specific and customized manner.3–7 An accurate assessment of each site’s debridement needs should always precede and follow subgingival instrumentation. Toward this goal, the diode laser can replace the periodontal probe when evaluating the clinician’s performance,3–7 and, at the same time, provide a bactericidal effect10–12 that reduces the local microbial load and aids healing.
LASER PROTOCOL
The treatment in each site takes 30 seconds, preceded and followed by use of 10 ml hydrogen peroxide 3% or 10 volumes irrigation.13–16 Used prior to and following nonsurgical periodontal debridement with ultrasonic and manual instrumentation, a 980-nm diode laser was utilized with the following parameters:
- Power: 2.5 W — average 0.7 W – 10 kHz (10,000 Hz)
- Mode: micro-pulsed (ps) Time Ton = 30 microseconds (μs) Time Toff = 70μs 30%.
- Fluence: 120 J/cm2
- Time: 30 seconds/pocket (30” x 3)
- Fiber: 0.400 mm (400 μ), not activated
- Local anesthesia had not been administered
The procedure is replicated in each pocket over three visits. Thus, laser use — alternating with conventional instrumentation — is recommended.3 The initial treatment was performed at three closely scheduled appointments: Visit 1 (baseline), Visit 2, and Visit 3 (30 days since baseline). In this case, a fourth debridement appointment was scheduled four months after the baseline visit.
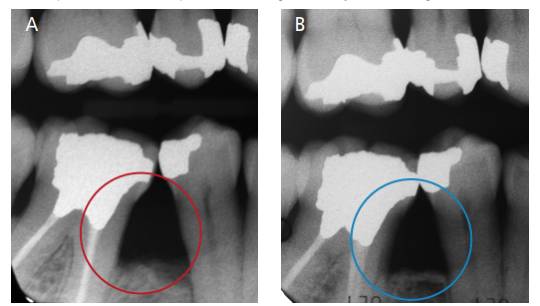
The patient was placed on a quarterly maintenance program. Case reevaluation at the one-year and multiyear follow-up appointments confirmed a significant and generalized improvement in probing data (Figure 3A through Figure 10C). Compared to the baseline X-rays, subsequent radiographs demonstrate the quality of bone mineralization (Figures 5A and 5B, and Figures 9A and 9B).
REEVALUATION AND FOLLOW-UP
From the second year on, case clinical stability was routinely assessed at maintenance visits, which had been reset at every four months. It is the clinician’s responsibility to update the diagnosis at each recall appointment by circumferential probing prior to nonsurgical periodontal instrumentation.3
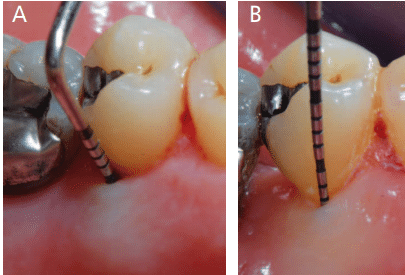
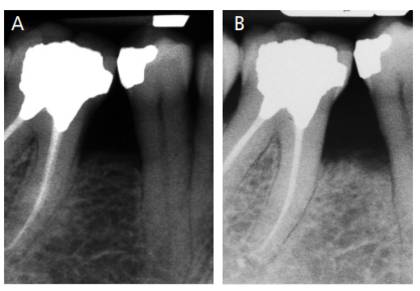
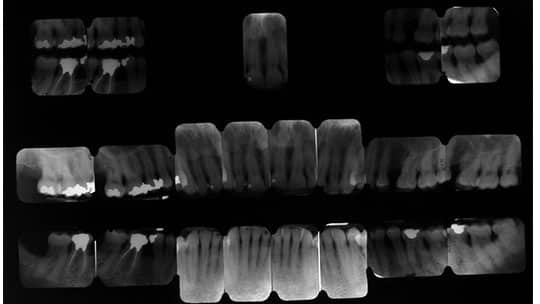
At a four-year follow-up appointment, a 7 mm probing depth was observed that was associated with bleeding on the mesiobuccal aspect of the first right maxillary molar. A periapical radiograph (Figure 7A) was obtained, along with a full-mouth X-ray series (Figure 8). A radiolucency was evident in the periapical radiograph (Figure 7A), indicative of a bone defect associated with a suspected bifurcation involvement.
In comparison, most sites maintained a condition of clinical stability, verified both by circumferential probing of the entire oral cavity and full-mouth radiographic assessment. Compared to the baseline radiograph (Figure 9A), after four years of follow-up the comparative interproximal bitewing image demonstrates better-represented bone architecture (Figure 9B).
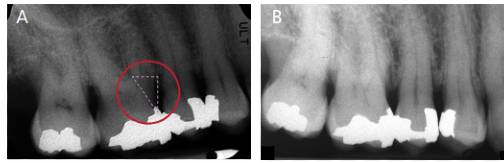
four years posttreatment from baseline. The dashed triangle seen in the red circle shows an arrow-shaped radiolucency, indicative of bifurcation involvement at the mesial site (A). Taken eight months later, periapical imaging shows an improvement in bone mineralization (B).
At the same four-year follow-up appointment, and following every radiographic diagnostic assessment, surgical management of the detected critical condition was proposed to the patient. Thus far, it had been possible to obtain a significant improvement in most sites using a laser-assisted nonsurgical approach. Continuous monitoring and diagnostic updating had identified an area that warranted surgical access due to its anatomical characteristics. Consequently, periodontal surgery was scheduled immediately after the next recall appointment. Before dismissing the patient, the clinician reinforced self-care oral hygiene methods and prescribed a disclosing solution to better highlight the biofilm to be removed from the future surgical site. The operator also stressed that long-term outcomes of any therapy, including surgery, could be positively influenced by the patient’s self-care performance.
Four months later, a prophylaxis appointment was scheduled approximately one week before surgery in the right posterior maxilla. The clinician updated the diagnosis by circumferential probing before nonsurgical periodontal instrumentation, noting that no bleeding upon probing or probing values greater than 3 mm were evident — even in the area where a justified surgical treatment was planned. These results were confirmed through additional probing.
It was natural to ask the patient what she had explicitly done in the previous four months. The answer was firm and assertive. She had followed the self-care recommendations scrupulously, three times a day: in the morning, and after lunch and dinner. In addition, at least twice a day the patient used an interdental brush dipped in chlorhexidine gel, as recommended.
The entire team congratulated her for a meticulous self-care regimen that resulted in postponement (for the time being) of surgical site management. The patient was dismissed after scheduling the next four-month recare appointment, at which time the right posterior maxillary area was reevaluated, also with a periapical radiograph (Figure 7B) taken eight months after the one shown in Figure 7A. As of this writing, the case has a three-year follow-up from the clinical instability episode, and seven years from the initial examination and treatment. The clinical case imaging was updated at the patient’s January 2020 recall visit (Figures 10A through 10C).
CONCLUSION
Since her initial evaluation in 2013, the patient in this case report has always shown good biofilm control. At each visit, the entire dental team reinforced self-care instructions and the importance of closely adhering to the personalized maintenance program. Although unpredictable, such a result may be possible with the initial therapy sessions first described, along with diagnostic updates conducted at each regularly scheduled recare appointment.
Surely, the patient’s self-care compliance in this specific case proved to be the single most significant factor affecting periodontal case management and a successful clinical outcome.
REFERENCES
- Mosaddad SA, Tahmasebi E, Yazdanian A, et al. Oral microbial biofilms: an update. Eur J Clin Microbiol Infect Dis. 2019;38:2005–2019.
- Schwiertz A. Microbiota of the Human Body Implications in Health and Disease. AG Switzerland: Springer International Publishing; 2016.
- Roncati M. Nonsurgical Periodontal Therapy Indications, Limits, and Clinical Protocols With the Adjunctive Use of a Diode Laser. Milano, Italy: Quintessence Publishing; 2015.
- Roncati M, Gariffo A. Three years non surgical periodontal treatment protocol to observe clinical outcomes in ≥ 6 mm pockets: a retrospective case series. Int J Periodontics Restorative Dent. 2016;36:189–197.
- Lerario F, Roncati M, Gariffo A, et al. Non-surgical periodontal treatment of peri-implant diseases with the adjunctive use of diode laser: retrospective controlled clinical study. Lasers Med Sci. 2016;31:1–6.
- Roncati M, Gariffo A, Barbieri C, Vescovi P. Ten year non surgical periodontal treatment protocol with the adjunctive use of diode laser monitoring clinical outcomes in ≥ 6 mm pockets: a retrospective controlled case series. Int J Periodontics Restorative Dent. 2017;37:647–654.
- Roncati M. The Importance of Dedicated Maintenance Therapy for Long-Term Success. In: Nevins M, Wang H. Implant Therapy: Clinical Approaches and Evidence of Success. 2nd ed. Chicago: Quintessence Publishing; 2019:475–489.
- Liang R, George R, Walsh LJ. Pulpal response following photo-biomodulation with a 904-nm diode laser: a double-blind clinical study. Lasers Med Sci. 2016;31:1811–1817.
- Heidari M, Fekrazad R, Sobouti F, et al. Evaluating the effect of photobiomodulation with a 940-nm diode laser on post-operative pain in periodontal flap surgery. Lasers Med Sci. 2018;33:1639–1645.
- Schulte-Lünzum R, Gutknecht N, Conrads G, Franzen R. The impact of a 940 nm diode laser with radial firing tip and bare end fiber tip on Enterococcus faecalis in the root canal wall dentin of bovine teeth: an in vitro study. Photomed Laser Surg. 2017;35:357–363.
- Gokhale SR, Padhye AM, Byakod G, Jain SA, Padbidri V, Shivaswamy S. A comparative evaluation of the efficacy of diode laser as an adjunct to mechanical debridement versus conventional mechanical debridement in periodontal flap surgery: a clinical and microbiological study. Photomed Laser Surg. 2012;30:598–603.
- Roncati M, Gariffo A. Systematic review of the adjunctive use of diode and Nd:YAG lasers for non-surgical periodontal instrumentation. Photomed Laser Surg. 2014;32:186–197.
- Caccianiga G, Cambini A, Donzelli E, Baldoni M, Rey G, Paiusco A. Effects of laser biostimulation on the epithelial tissue for keratinized layer differentiation: an in vitro study. J Biol Regul Homeost Agents. 2016;30(Suppl 1):99–105.
- Caccianiga G, Rey G, Fumagalli T, Cambini A, Denotti G, Giacomello MS. Photodynamic therapy (association diode laser/hydrogen peroxide): Evaluation of bactericidal effects on periodontopatic bacteria: an in vitro study. Eur J Inflamm. 2012;10(Suppl):102–106.
- Caccianiga G, Rey G, Paiusco A, et al. Oxygen high level laser therapy is efficient in treatment of chronic periodontitis: a clinical and microbiological study using PCR analysis. J Biol Regul Homeost Agents. 2016;30(Suppl 1):87–97.
- Caccianiga G, Rey G, Baldoni M, Paiusco A. Clinical, radiographic and microbiological evaluation of high level laser therapy, a new photodynamic therapy protocol, in peri-implantitis treatment; a pilot experience. Biomed Res Int. 2016;2016:6321906.
From Decisions in Dentistry. January 2021;7(1): 32–35.




