 ROSSHELEN / ISTOCK / GETTY IMAGES PLUS
ROSSHELEN / ISTOCK / GETTY IMAGES PLUS
Flavonoids’ Effects on Oral Health
Current research suggests flavonoids’ bactericidal and antiviral action may help promote oral and systemic health.
This course was published in the October 2022 issue and expires October 2025. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
AGD Subject Code: 150
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Describe flavonoids’ possible bactericidal action.
- Explain potential benefits of flavonoid use in oral care products.
- Identify food sources of flavonoids and dietary recommendations for flavonoid-rich foods that may provide oral health benefits.
Many plant products, such as fruits, grains, tea, wine, barks and roots, possess bioactive ingredients known as flavonoids — a class of polyphenol organic compounds that is part of a larger group of plant-based phytochemicals.1 In recent years, flavonoids have garnered attention in dentistry and other sciences because they have shown potential health benefits, including antiviral and bactericidal action.2 Specifically, they may alter the structural integrity of bacterial cell membranes, inhibit peptidoglycan synthesis in cell walls, and prevent biofilm formation.3 For example, dental plaques are formed by cariogenic microorganisms that reside in a biofilm in a susceptible host. Streptococcus species account for a large portion of these, and, in some cases, Staphylococcus, Actinomycetes, Lactobacillus and Enterococcus may also be found in the oral cavity. These bacteria may interact with other microorganisms present in the oral cavity, such as Candida albicans, which can cause oral thrush.4,5
Many of these microorganisms perform beneficial functions, including the conversion of dietary nitrates into nitrites which are further converted to nitric acid and serve as vasodilators and inhibitors of cariogenic bacteria.6 However, the disruption of microbial homeostasis due to diet or immune response triggers the onset of diseases such as dental caries.4 Thus, foods or products that provide flavonoids may help promote oral and systemic health.
A growing interest in plant-based products has drawn increased attention to flavonoids beyond dietary sources and has led patients to consider toothpaste and mouthrinses made with alternative ingredients, including herbs and spices that contain polyphenols.7 Concerns surrounding possible side effects or sensitivities associated with traditional oral care products and an increase in antibiotic-resistant strains of many bacteria have also drawn attention to the bacteriostatic effects of herbal medicines and their possible benefits in the oral cavity.8–10
Furthermore, overall growth in the oral care industry and home-based oral care remedies has been fueled, in part, by the COVID-19 pandemic, which led more patients to choose at-home remedies to decrease exposure to COVID-19 in dental settings. To some extent, this trend is expected to persist, suggesting that flavonoids and other natural ingredients may play a continued role in oral hygiene — both through dietary sources and oral care products that patients choose.11
TOOTHPASTE AND MOUTHRINSE
The antimicrobial activity of flavonoids has been explored in numerous studies and in real-world applications. A study by Forouzanfar et al12 examined the performance of saffron, a spice with flavonoids and antioxidant properties,13 against a placebo among patients experiencing marginal gingivitis. Participants were assigned to use either the saffron toothpaste or an unspecified placebo, and pocket depth, gingival index, plaque index and bleeding on probing were examined before and after four weeks of use. The group that used saffron toothpaste experienced significant improvement in gingival indices and bleeding on probing in comparison with the placebo group. No further details about the study methodology were provided, limiting comparisons with other research.
In vitro tests studied the effects of toothpastes made from the medicinal plants Syzygium aromaticum (clove), Dennettia tripetala (pepper fruit) and Jatropha curcas (physic nut), which all contain phenols, flavonoids, alkaloid and saponins, on oral pathogenic microorganisms such as Escherichia coli, Bacillus subtilis, S. aureus, S. epidermidis, Micrococcus luteus, S. mutans, S. pyogenes, L. acidophilus and C. albicans. The plant-based toothpastes inhibited bacterial growth, particularly when more than one plant was included (e.g., clove combined with pepper fruit and/or physic nut), possibly due to synergistic effects of their bioactive compounds. The authors concluded these biomolecules may be safe and effective toothpaste ingredients.14 Similarly, a study by Uju and Obioma15 showed that clove, tobacco and bitter kola all have inhibitive effects on S. mutans, with the most effective being clove extract. The researchers suggest that clove extract be considered for use in toothpaste. Despite the flavonoids provided by tobacco, its use can have adverse effects on oral health; thus, efforts have been made to eliminate tobacco and nicotine from toothpastes and powders.16
Faria et al17 compared mouthrinses produced with extracts from the flavonoid-containing plants Calendula officinalis (pot marigold) and Camellia sinensis (tea plant) against a traditional oral rinse featuring chlorhexidine gluconate. The plant-based mouthrinses demonstrated some bacteriostatic effects against microbes that had attached to sutures after molar extraction, but their performance did not exceed that of chlorhexidine. However, while chlorhexidine has been shown to impair cell proliferation needed for wound repair, extracts of green tea and pot marigold flowers have demonstrated the ability to enhance healing.17 Additionally, a recent review of flavonoids showed they are also effective in preventing and reducing the severity of periodontitis when delivered to patients through mouthrinses, toothpaste and food products.18 They may reduce inflammation and periodontitis by targeting immune signaling that occurs in response to plaque biofilms.
ENDODONTIC AND POSTSURGICAL USES
After oral surgeries, bacteria in plaque may gain access to circulating blood, increasing the risk of septic inflammation of the endocardium.19,20 In an in vitro experiment using cardiomyocyte cells, the flavonoid myricetin inhibited inflammatory responses and cell signaling pathways that result from exposure to peptidoglycan, a cell wall component of plaque bacteria.19 The authors concluded that future research should examine the use of myricetin for treating periodontal disease and infective endocarditis. Similarly, licorice, when used alone or in root canal medications, exhibited a significant bactericidal effect against E. faecalis — a bacterium associated with oral infections — inhibiting its growth more effectively than the traditional calcium hydroxide medicament.21 The mechanisms behind the bactericidal effects of flavonoids include plasma membrane disruption, as well as inhibition of bacterial toxins and peptidoglycan, adenosine triphosphate, electron transport chain and nucleic acid synthesis.22
In endodontics, natural herbal products, such as licorice ethanolic extracts, have been used in root canal therapy for cleaning and disinfecting because they have exhibited antibacterial properties against several oral pathogens. Baicalein, a flavonoid found in the root of the Scutellaria baicalensis plant, promotes remineralization and alkaline phosphatase activity, angiogenic factors, and morphogenetic proteins in human dental pulp cells.23 Other uses of natural products (including grapefruit, tangerine, lime and lemon oils) in root canal therapy include serving as solvents for softening gutta-percha, and maintaining the viability of human periodontal ligament cells when used as a storage medium for avulsed teeth (aloe vera, for example).23
Additionally, a study by Soltani et al9 demonstrated that green tea, another flavonoid-rich plant, was effective in reducing gingival bleeding following oral surgery. After mandibular molar extractions, researchers used either a sterile gauze or a sterile gauze permeated with green tea to reduce gingival bleeding. The results showed that gingival bleeding took twice as long to resolve in the control group compared to subjects treated with green tea gauze.
USE OF PROPOLIS AS A MEDICAMENT
The ability of propolis to limit dental plaque accumulation has contributed to its use as an ingredient in mouthrinses and toothpaste. Propolis is a naturally occurring resinous substance obtained by bees mixing beeswax with substances from various plant species.24 Flavonoids are major phytochemicals in propolis, although other phenolic compounds are also present.25
Both Brazilian green and red propolis are effective in treating oral diseases.26 Propolis may reduce gingival inflammation.24,25 It can also be used as a canal irrigation solution or intracanal medicament in endodontic treatment. As a storage medium, it has the potential to keep periodontal ligament cells viable for six hours or longer, and may be more effective than traditional products, such as milk, balanced salt solution or serum. (However, it may be more difficult to obtain propolis and prepare a solution, so milk may be preferred.) Propolis is also capable of inhibiting osteoclast activity, inducing the production of tubular dentin, and reducing pulpal inflammation.26
During the treatment of malocclusions with narrowed maxilla in orthodontics, bone remodeling takes place within the palatine suture.27 Research with rats found that systemic delivery of a propolis solution increased bone remodeling within the palatine suture, as well as the number of osteoblasts.24 When constructing restorative dentin, propolis can be used to reduce the permeability of the dentin; it also provides advantages over calcium hydroxide in direct pulp capping.23,24 In addition, it facilitates the healing of surgical wounds and is used in socket wounds after tooth extractions to foster quick epithelial repair.24,26,28
ADDITIONAL APPLICATIONS
The use of flavonoids in other branches of dentistry has also been studied. An experiment by Yang et al29 showed that when used with ethanol-wet bonding, epigallocatechin-3-gallate — a type of catechin in green tea — promoted stronger bonding strength and stability of etch-and-rinse adhesives on dentin. This suggests that additional research is needed to assess the feasibility of widespread clinical use.
Numerous flavonoids (including proanthocyanidin, naringin and quercetin) have demonstrated efficacy in remineralizing root caries.30 In demineralized dentin, these flavonoids increased the modulus of elasticity and tensile strength, with proanthocyanidin working best — likely due to its ability to improve the cross-linking ability of the dentin collagen matrix.27 Further study to explore practical uses in clinical settings is warranted.
FLAVONOID INTAKE
Though controlled studies on the effects of specific flavonoid-rich foods on oral health are limited, results to date have supported flavonoids, perhaps due to their antioxidant, anti-inflammatory, anti-adhesive and antibacterial properties.18 For example, Loon et al8 concluded that bromelain extract from pineapples suppressed the growth of S. aureus, perhaps due to the presence of flavonoids, bromelain and vitamin C.
Due to their anti-adhesive properties, cranberries may also prevent caries by inhibiting biofilm formation. Research with cranberry juice found that it interfered with adhesion of Streptococcus bacteria in biofilms, possibly through interactions between cranberry components and hydrophobic proteins on the outside of bacterial cells.31
Tea is another rich source of flavonoids that may promote oral health due to its antibacterial action.32 Given the presence of flavonoids in tea, Kawarai et al33 compared the difference in the effects of Assam tea and green tea against S. mutans. Assam tea showed stronger biofilm inhibition against S. mutans than green tea. This may be due to the presence of galloylated catechins at high concentrations in Assam tea, coupled with smaller amount of polysaccharides — which, together, may work to inhibit biofilm formation.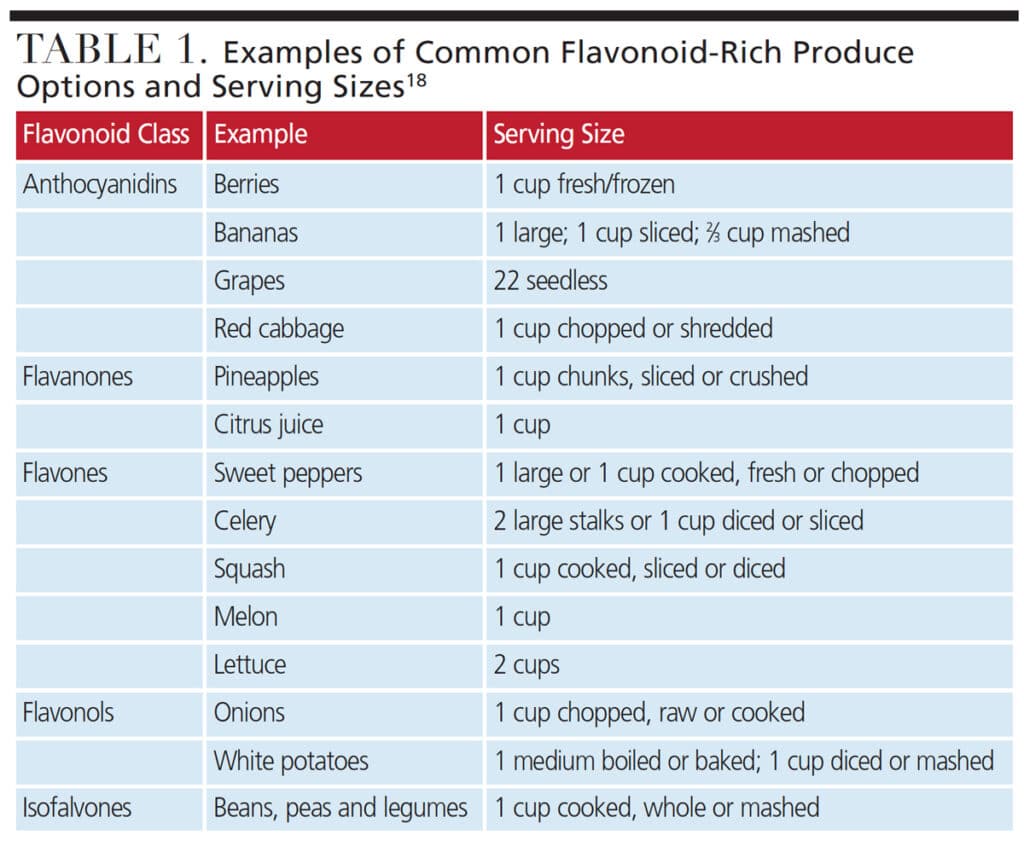
DIETARY RECOMMENDATIONS
Huang et al34 analyzed the 2013–2016 National Health and Nutrition Examination Survey data with the Phenol Explorer Database to estimate the amount of polyphenols consumed by American adults. After phenolic acids, flavonoids were consumed the most (379.1 ± 10.7 mg/day). Flavan-3-ols were the most consumed flavonoid subclass reported in this study, with a mean intake of 223.2 ± 7.8 mg/day. Surveys of flavonoid intake in the United States have reported tea, wine, beer, citrus fruits and apples as important sources of flavonoids among adults.34,35
As noted, research on the intake of specific flavonoid-rich foods and their role in oral health is limited, but promising. For example, the antibacterial benefits of bromelain extract from pineapple8 may also be present when the fruit is consumed. Indeed, research with school-age children found a decreased incidence of viral and bacterial infections and improved granulocyte counts in those who consumed one to two servings of pineapple daily.36
Further studies are also warranted to clarify the mechanisms that appear to help cranberries inhibit biofilm formation, as well as the serving size needed to provide benefits. Given that current dietary guidelines recommend consumption of two servings of fruit daily for a 2000-calorie diet,37 cranberry juice and canned pineapple could be included as part of a healthy diet that may provide additional oral health benefits.
Dental providers and other health professionals can help promote flavonoid consumption by educating patients about the benefits that fruits, vegetables and other flavonoid-rich foods provide (Table 1). In light of the growing interest in natural oral care products (which often contain flavonoids), dental teams can also discuss these options with patients as additional research about their effectiveness becomes available.
CONCLUSION
Although emerging evidence suggests flavonoids may provide oral health benefits, more studies are needed to understand how other types of plant chemicals contribute to the effectiveness of flavonoids and potential benefits of natural oral care products. Future work should also evaluate the possibility and implications of supplement/drug interactions.38,39
As research in these areas continues and scientific understanding grows,40 oral health professionals will be better able to offer evidence-based education to patients seeking guidance on nutrient-rich foods and natural oral care products.
REFERENCES
- Panche A, Diwan A, Chandra S. Flavonoids: an overview. J Nutr Sci. 2016;5:1–15.
- Kumar S, Pandey AK. Chemistry and biological activities of flavonoids: an overview. ScientificWorldJournal. 2013;2013:162750.
- Górniak I, Bartoszewski R, Króliczewski J. Comprehensive review of antimicrobial activities of plant properties. Phytochem Rev. 2019;18:241–272.
- Gutiérrez-Venegas G, Gómez-Mora J, Meraz-Rodríguez M, et al. Effect of flavonoids on antimicrobial activity of microorganisms present in dental plaque. Heliyon. 2019; 5:3–13.
- Nishikawara F, Nomura Y, Imai S, et al. Evaluation of cariogenic bacteria. Eur J Dent. 2007;1:31–39.
- Kilian M, Chapple M, Hannig P, et al. The oral microbiome — an update for oral healthcare professionals. Br Dent J. 2016;221:657–666.
- Natural products drive oral hygiene market growth. Available at: https://www.dentistrytoday.com/natural-products-drive-oral-hygiene-market-growth/. Accessed September 2, 2022.
- Loon Y, Satari M, Dewi W. Antibacterial effect of pineapple (Ananas comosus) extract toward Staphylococcus aureus. Padjadjaran J Dent. 2018;30:1–6.
- Soltani R, Haghighat A, Fanaei M. Evaluation of the effect of green tea extract on the prevention of gingival bleeding after posterior mandibular teeth extraction: a randomized controlled trial. Evid Based Complement Alternat Med. 2014;2014:1–4.
- Capodagli G, Lee S, Boehm K, et al. Structural and functional characterization of methicillin-resistant Staphylococcus aureus’ class IIb fructose 1,6-bisphosphate aldolase. Biochemistry. 201453:7604–7614.
- Grand View Research. Oral Care Market Size, Share and Trends Analysis Report by Product (Toothbrush, Toothpaste, Mouthwash/Rinse, Denture Products, Dental Accessories), by Region, and Segment Forecasts, 2022–2030. Available at: https://www.grandviewresearch.com/industry-analysis/oral-care-market. Accessed September 2, 2022.
- Forouzanfar A, Mokhtari M, Kamalinezhad M. Evaluation of toothpaste containing aqueous saffron stigma extract on gingival indices in patients with marginal generalized plaque-induced gingivitis. Iran J Med Sci. 2016;41(Suppl 3):S31.
- Karimi E, Oskoueian E, Hendra R, et al. Evaluation of Crocus sativus L. stigma phenolic and flavonoid compounds and its antioxidant activity. Molecules. 2010;15:6244–6256.
- Oluwasina O, Ezenwosu I, Ogidi C, et al. Antimicrobial potential of toothpaste formulated from extracts of Syzygium aromaticum, Dennettia tripetala and Jatropha curcas latex against some oral pathogenic microorganisms. AMB Expr. 2019;9:1–13.
- Uju D, Obioma N. Anticariogenic potentials of clove, tobacco and bitter kola. Asian Pac J Trop Med. 2011;4:814–818.
- Agrawal S, Ray R. Nicotine contents in some commonly used toothpastes and toothpowders: a present scenario. J Toxicol. 2012;2012:1–11.
- Faria R, Cardoso L, Akisue G, et al. Antimicrobial activity of Calendula officinalis, Camellia sinensis and chlorhexidine against the adherence of microorganisms to sutures after extraction of unerupted third molars. J Appl Oral Sci. 2011;19:476–482.
- Fernández-Rojas B, Gutiérrez-Venegas G. Flavonoids exert multiple periodontic benefits including anti-inflammatory, periodontal ligament-supporting, and alveolar bone-preserving effects. Life Sci. 2018;209:435–454.
- Rosas-Martínez M, Gutiérrez-Venegas G. Myricetin inhibition of peptidoglycan-induced COX-2 expression in H9c2 cardiomyocytes. Pre Nutr Food Sci. 2019;24:202–209.
- McDonald J. Acute infective endocarditis. Infect Dis Clin North Am. 2009;23:643–664.
- Badr A, Omar N, Badria F. A laboratory evaluation of the antibacterial and cytotoxic effect of liquorice when used as root canal medicament. Int Endod J. 2010;44:51–58.
- Górniak I, Bartoszewski R, Króliczewski J. Comprehensive review of antimicrobial activities of plant flavonoids. Phytochem Rev. 2018;18:241–272.
- Almadi E, Almohaimede A. Natural products in endodontics. Saudi Med J. 2018;39:124–130.
- Więckiewicz W, Miernik M, Więckiewicz M, et al. Does propolis help to maintain oral health? Evid Based Complement Alternat Med. 2013;2013:1–8.
- Ahangari Z, Naseri M, Vatandoost F. Propolis: chemical composition and its applications in endodontics. Iran Endod J. 2018;13:285–292.
- De Carvalho C, Fernandes W, Mouttinho T, et al. Evidence-based studies and perspectives of the use of Brazilian green and red propolis in dentistry. Eur J Dent. 2019;13:459–465.
- Epasinghe D, Yiu, C, Burrow M, et al. Effect of flavonoids on the mechanical properties of demineralised dentine. J Dent. 2014;42:1178–1184.
- Sankari S, Babu N, Rani V. Flavonoids — clinical effects and applications in dentistry: a review. J Pharm Bioallied Sci. 2014;6(Suppl 1):S26–S29.
- Yang H, Guo J, Deng D, et al. Effect of adjunctive application of epigallocatechin-3-gallate and ethanol-wet bonding on adhesive-dentin bonds. J Dent. 2016;44:44–49.
- Epasinghe D, Yiu C, Burrow M. Effect of flavonoids on remineralization of artificial root caries. Aust Dent J. 2015;61:196–202.
- Yoo S, Murata R, Duarte S. Antimicrobial traits of tea and cranberry-derived polyphenols against Streptococcus mutans. Caries Res. 2009;23:643–664.
- Wang H, Ren D. Controlling Streptococcus mutans and Staphylococcus aureus biofilms with direct current and chlorhexidine. AMB Expr. 2017;7:1–9.
- Kawarai T, Narisawa N, Yoneda S, et al. Inhibition of Streptococcus mutans biofilm formation using extracts from Assam tea compared to green tea. Arch Oral Biol. 2016;68:73–82.
- Huang Q, Braffett B, Simmens S, et al. Dietary polyphenol intake in U.S. adults and 10-year trends: 2007–2016. J Acad Nutr Diet. 2020;120:1821–1833.
- Bai W, Wang C, Ren C. Intakes of total and individual flavonoids by U.S. adults. Int J Food Sci Nutr. 2014;65:9–20.
- Cervo M, Llido L, Barrios E, et al. Effects of canned pineapple consumption on nutritional status, immunomodulation, and physical health of selected school children. J Nutr Metab. 2014;2014:1–9.
- Office of Disease Prevention and Health Promotion. Dietary Guidelines for Americans, 2020–2025. Available at: https://www.dietaryguidelines.gov/sites/default/files/떔-12/Dietary_Guidelines_for_Americans_떔-2025.pdf. Accessed September 2, 2022.
- Boullata J. Natural health product interactions with medication. Nutr Clin Pract. 2005;20:33–51.
- Dippenaar J. Herbal and alternative medicine: the impact on anesthesia. South African J Anaesth Analg. 2015;21:8–13.
- Catapano-Martinez D, Boyce M, Garland M. The protective role of polyphenols in oral health. Decisions in Dentistry. 2018;4(3):33–36.
From Decisions in Dentistry. October 2022;8(10):42-45.




