
Five Misconceptions in Endodontics
Exploring commonly held myths will result in a deeper understanding of modern root canal treatment.
Endodontic therapy has evolved from a specialty procedure requiring tedious mechanical shaping of oftentimes-obscure root canal systems into one that employs advanced technology that allows patients to retain teeth that were once untreatable. This is the result of advances in materials, techniques and equipment. New root canal cleaning, shaping and obturation systems, along with enhanced magnification, illumination and reliable electronic apex locators are a few of the technological developments that have contributed to the improved delivery of endodontic treatment. Although these have allowed better outcomes and made treatment more predictable, endodontic success is not based solely on new techniques and devices. It is grounded in the knowledge and application of basic biological and mechanical principles.
This paper will discuss five misconceptions prevalent in endodontics. These false notions may interfere with optimal outcomes — even when the latest technology is utilized. Exploring these myths will hopefully clarify commonly held fallacies, resulting in a deeper appreciation and understanding of endodontic procedures.
MYTH 1: “But I don’t see anything radiographically”
While radiographs are an integral and essential part of root canal therapy (RCT), endodontic diagnosis is sometimes mistakenly based solely on a radiograph at the initial phase of treatment. Diagnosis is the critical first step of RCT and must include a thorough and systematic assessment of both subjective and objective information. The value of radiographic images alone is often overestimated. Because they are two-dimensional images of three-dimensional (3D) objects, conventional radiographs have inherent limitations — although this can be mitigated to some extent by taking more than one radiograph from different angles. Moreover, recent advances in technology allow 3D techniques, such as cone beam computed tomography (CBCT), to aid in the diagnosis of complex cases. However, imaging does not replace a medical and dental history and clinical examination of the patient as part of the diagnostic process.
Refer to the chapter on diagnosis in any endodontic textbook and in the description of objective tests note that radiographs are routinely listed as the last objective test. This is because the radiograph is only one aspect of the objective evaluation and needs to be interpreted in the context of history and other clinical tests. When used as the sole means of evaluation, a radiograph can lead to an incorrect diagnosis and, consequently, inappropriate treatment.
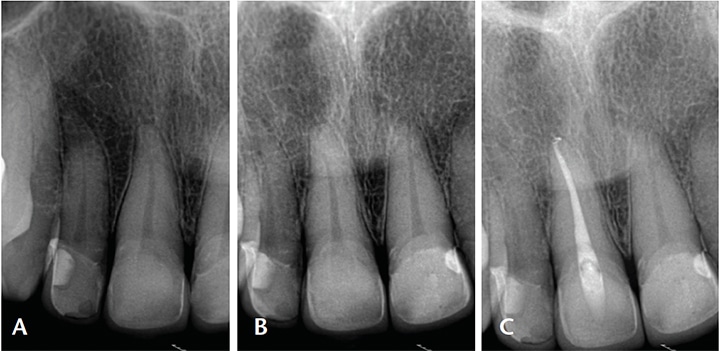
the symptoms resolved immediately after root canal treatment of tooth #8 (C).
In many instances, endodontic pathosis does not show up radiographically. It should be acknowledged that only hard tissues are visible radiographically; soft tissues, such as pulp, are not visible radiographically. Therefore, for a patient presenting with signs and symptoms of pulpitis, changes in the pulp will often not show radiographically. While the radiograph will reveal possible etiological factors, it will not identify the affected tooth. The patient whose radiographs are shown in Figures 1A and 1B presented with a chief complaint of throbbing pain from the maxillary right central and lateral incisors after porcelain veneers were placed on the four maxillary incisors. The patient could not localize the pain — and this is not uncommon in cases of irreversible pulpitits. It has been shown that patients are unable to accurately pinpoint the tooth that is the source of pain, especially when they have symptoms of irreversible pulpitits and no periapical symptoms.1,2
Objective clinical testing (including palpation, biting and pulp sensibility tests) revealed the maxillary right central incisor had irreversible pulpitis and symptomatic apical periodontitis. Although this was the source of the patient’s pain, this assessment was not evident radiographically. The patient’s symptoms resolved after RCT was completed on the maxillary right central incisor (Figure 1C).
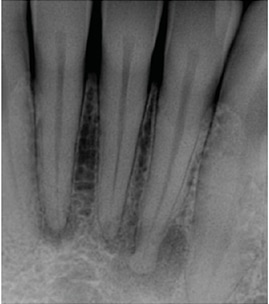
incisors with periapical radiolucencies.
The teeth tested vital and there is no
history of trauma. These lesions are
periapical osseous dysplasia.
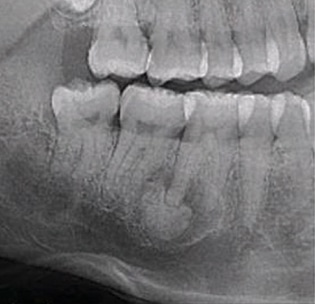
molar with a lesion around the distal root. The tooth is vital. This lesion is not of endodontic
origin.
Likewise, a necrotic pulp, or symptomatic apical periodontitis associated with a necrotic pulp or an irreversibly inflamed pulp will not be evident radiographically. If the inflammation only involves the periodontal ligament, no radiographic changes will be seen. However, the patient’s symptoms — verified with objective tests of palpation, biting or percussion — would confirm that apical periodontitis is present clinically, although it is not present radiographically.
As the inflammation progresses and results in bone resorption, a radiolucent area may develop periapically. It is important to recognize that lesions may develop in the bone before they are visible radiographically. The appearance of a lesion is dependent on structural variations in the apical region, bone density, radiographic angulation and contrast.3 In order to visualize an apical lesion, it is necessary to have approximately 30% to 50% of mineral bone loss.4,5 Therefore, pathosis may be present, but lacking radiographic manifestation.
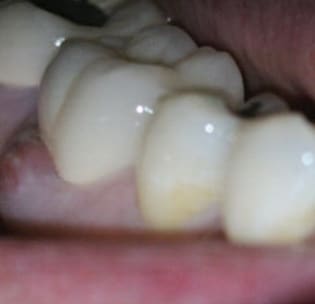
gingival abscess on the buccal.
Conversely, radiographic findings may be unrelated to pathological changes associated with the teeth in the area. Periapical cemental dysplasia may be misdiagnosed as a lesion of endodontic origin secondary to a necrotic and infected pulp. However, the diagnostic procedures would conclude that the pulp is vital. In this instance, other causes for the radiographic anomaly should be considered (Figure 2 and Figure 3).
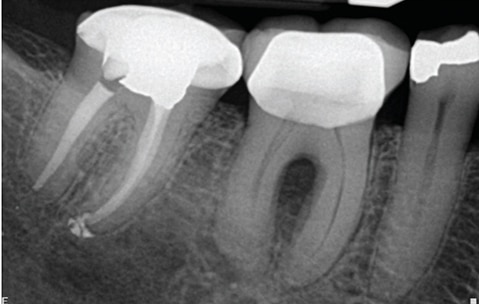
Another example of incomplete diagnosis based only on a radiograph is the endo-perio lesion. As seen in Figure 4, this patient presented with a gingival abscess buccal to the mandibular right first molar, deep periodontal probing with bleeding on the buccal aspect of tooth #30, and radiographic evidence of a lucency in the furcation (Figure 5). Without objective testing, this pathology may have been erroneously attributed to periodontal disease. However, the pulp was diagnosed as necrotic, as there was no response to cold testing. The endo ice cold test can be effective for endodontic testing of teeth with full-coverage restorations.6 Additionally, there was a logical etiology for the pulpal pathology, as the tooth had an existing restoration, signifying potential pulpal irritation in the past. In this case, proper treatment based on a thorough evaluation, not just radiographic evaluation, resulted in resolution of the patient’s symptoms and healing (Figures 6A and 6B).
The takeaway is that radiographic interpretation alone may have shortcomings and should only complement subjective and objective findings.
MYTH 2: “I do not treat molars, only anteriors and bicuspids”
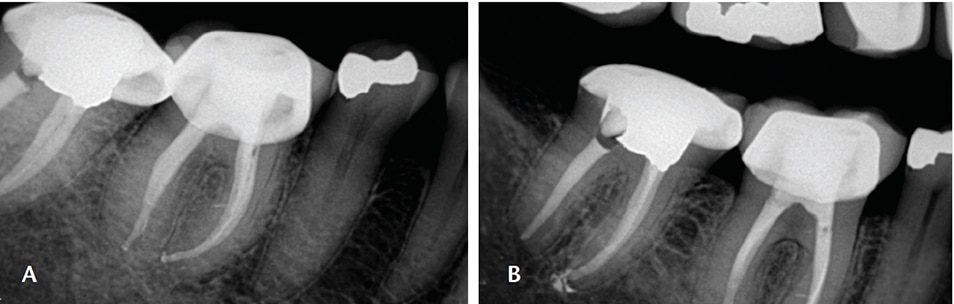
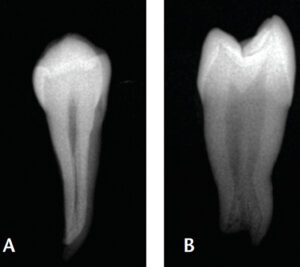
Case selection is an important part of treatment. Treating cases that are within the clinician’s comfort zone and level of expertise will help ensure optimal outcomes. Many general dentists limit their endodontic procedures to anterior teeth and bicuspids, opting to refer molars to the endodontist. It is important to realize that anterior and bicuspid teeth may have anatomy that varies from what is perceived from the radiographic image. Thus, it is important to recognize the limitations of radiographs in case selection, as well. Based on the mesial-distal view (Figure 7A), most dentists might conclude this tooth would be difficult to treat and might best be treated by an endodontist. However, the two-dimensional buccal (clinical) view of this tooth may not raise any concerns (Figure 7B). Note how the clinical scenario changes when the anatomical details are revealed in the mesial-distal view; although this view is not seen with conventional radiographs, an angled view may shed light on this complex configuration. A 3D CBCT image would reveal this complicated anatomy in greater detail. With the traditional buccal (clinical) view, the clinician would not be aware the canal bifurcates in the apical third and thus is a challenging case to treat. The pulp canal system is complex and canals may branch, divide and rejoin; therefore, it is important to have an appreciation for the pulp space anatomy prior to undertaking endodontic therapy (Figures 8A and 8B).
Maxillary anterior teeth are usually considered to have uncomplicated anatomy comprised of a single canal.7 The same cannot be presumed for mandibular anteriors or premolars. At least 25% of the time, mandibular incisors have two canals, while the incidence of two canals in mandibular first premolars occurs in 30% of cases.7 While there are uncomplicated mandibular incisors and premolars, it is important to appreciate that complex anatomy is not uncommon, and that mandibular incisors and premolars are often mistakenly considered to be simple teeth to treat endodontically.
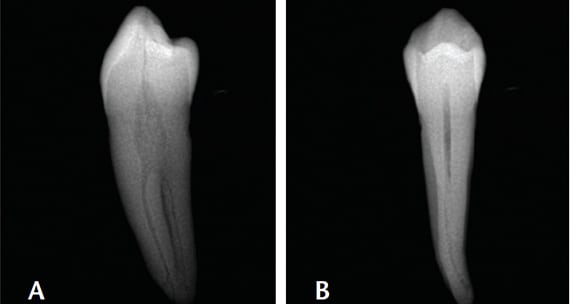
Due to their small size and radiographic appearance, mandibular incisors are perceived to have small, round canals that, in theory, should be easy to clean and shape with files. However, observe the discrepancy between the facial (clinical) view seen in Figure 9A and mesial view (Figure 9B). The amount of debris left in root canals after instrumentation is related to internal anatomic characteristics, such as an oval canal; in addition, broad, oval canals are challenging to clean and obturate effectively. This is illustrated in Figures 10A and 10B. Although it may appear radiographically that a single cone can adequately fill a “round” canal, this is evidently not the case. The inadequate obturation seen here is due to lack of proper shaping that resulted from the clinician’s lack of awareness that the canal is oval and initial access was insufficient.8
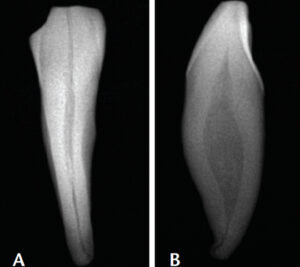
reality of a very broad canal (B).
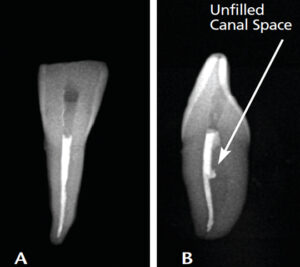
adequate on clinical view (A); however, the
mesial-distal view shows the inadequacies of
the obturation, which is secondary to
deficiencies in shaping the canal (B).
The main objective in RCT is the cleaning and shaping of the complete pulp space, followed by a bacteria-tight seal achieved by obturating the canal and timely placement of a sound coronal restoration. Compromising any of these steps will decrease the prognosis of a favorable outcome.
MYTH 3: “I need to establish working length as soon as I find the canal”
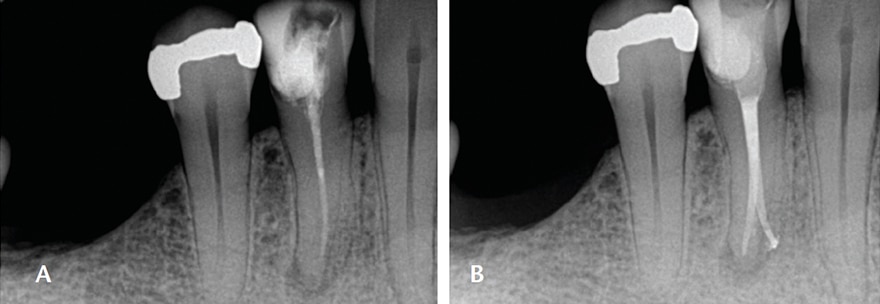
There is no benefit in establishing working length as soon as the canal is identified.9,10 It is advisable to establish the radicular access or coronal flare first for two reasons. First, this provides straight-line access into the coronal aspect of the canal. Straight-line access enables better tactile sense of the apical portion,11 better assessment of the canal configuration, and a better approach to cleaning and shaping the canal.12 Consider the maxillary bicuspid shown in Figures 11A and 11B. The buccal (clinical) view reveals a relatively straight root canal system. However, the perspective changes once the clinician examines the mesial view. Both canals are curved, with the buccal canal exhibiting significant curvature. Shaping the coronal portion first will create a more favorable condition to allow access to the apical portion of the canal and, consequently, working length. This is a rational and logical approach that will allow easier access to the apical portion of the canal. Attempting to reach working length prior to establishing the coronal flare may result in mishaps, such as ledges and transportations.12,13
The second reason to not establish working length immediately upon finding the canal is that as the canal is shaped, the working length changes.9,12 The most significant change occurs when shaping the coronal portion of the canal. This step is achieved with orifice openers and, when necessary, Gates-Glidden drills. The coronal flare helps improve access, ensuring that files have a smooth path to the first canal curvature. It has been shown that less change in working length occurs when it is established after coronal flaring.9 Establishing coronal flare first results in more consistent determination of working length.14 The shaping of the coronal portion of the canal has to be done at some point; achieving it initially results in fewer mishaps and less chance of changes in working length.
MYTH 4: “Let me just increase the working length slightly so that it looks correct radiographically”
The apical constriction has been established as the ideal termination point of shaping and filling the root canal system.15 This constriction has been reported as being 0.5 to 1.0 mm from the apical foramen.16 Radiographs have traditionally been used to determine working length to this level. However, there are shortcomings with the radiographic evaluation of working length. For example, difficulties arise due anatomical interferences and distortion due to angulation. Furthermore, the position of the apical constriction to the apical foramen, and position of the apical foramen to the anatomic or radiographic apex have been shown to fluctuate. The apical constriction is not consistently located 0.5 to 1.0 mm from the foramen.17
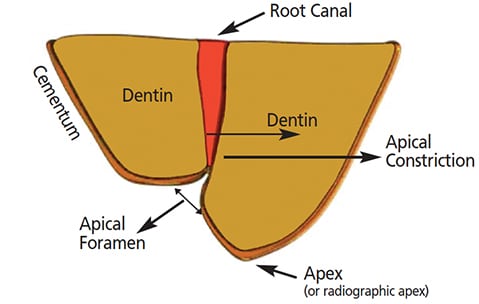
The apical foramen may be eccentrically located away from the anatomical or radiographic apex of the root (Figure 12).18 It may also be lateral to the apical extent of the root or on the buccal or lingual aspects. The accompanying images demonstrate the eccentric position of the apical foramen: While an attempt to place the working length file 1.0 mm from the radiographic apex (Figure 13A) resulted in the file protruding from the canal (Figure 13B), note how placing the file 1.0 mm from the observed apical foramen appears to be slightly short of the presumed working length on the radiograph (Figure 13C). Based on these challenges, the use of an electronic apex locator for length determination has been shown to be more accurate for assessing working length than radiographic methods.19,20
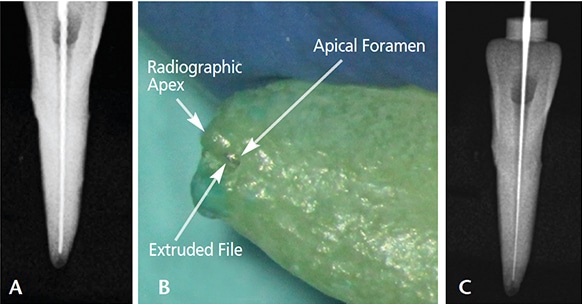
Other advantages of using apex locators are a decrease in radiation and increased efficiency. Although these devices are highly accurate, it is still advisable to take at least one radiograph to detect possible errors in the electronic measurement and to better assess canal morphology and deviations.19 Radiographs taken during RCT may add essential information about canal anatomy that could be missed if apex locators are used exclusively and no working radiographs are taken. When the working length established by the apex locator looks slightly shorter than 0.5 to 1.0 mm from the radiographic apex, it is most likely at the correct position, and increasing the working length, even slightly, would result in over-instrumentation and overfilling.21
MYTH 5: “I am going to prescribe antibiotics, just in case “
The routine use of antibiotics during the course of endodontic treatment is not supported by the principles of evidence-based dentistry and is counter to the recommendations of publications supported by the American Association of Endodontists22 and the American Dental Association.23 It has been proven that antibiotics do not relieve painful pulpitis and do not resolve localized periapical inflammation.24,25 Endodontic treatment removes the source of infection from the root canal system and this establishes a favorable condition for healing to take place. Furthermore, prescribing antibiotics prophylactically does not prevent flare-ups or reduce pain.26–28
Antibiotics are indicated when there are systemic signs of an infection, such as fever and malaise, an infection that is spreading, or cellulitis is present.29 Unsupported use of antibiotics also contributes to the development of antibiotic-resistant bacteria,30 which is a serious global health threat.31,32 Furthermore, antibiotics are not innocuous drugs (as some clinicians and many patients perceive), and their use involves risks. An estimated one in five emergency department visits for adverse drug events in the United States is for antibiotic-related adverse events.33 Clearly there are legitimate indications for antibiotics in dentistry; however, the decision to prescribe antibiotics should be based on a sound diagnosis and clear indications — not due to patient demands or a “just-in-case” mentality.
CONCLUSION
Advances in techniques, materials and equipment have allowed many general practitioners to expand their services to include endodontic procedures. While these technological developments have contributed to more predictable RCT, successful treatment remains grounded in the knowledge and application of basic biological and mechanical principles. The key to successful outcomes is for the clinician to recognize his or her skill and comfort levels. By choosing cases wisely and referring to the endodontist when appropriate, practitioners can help ensure the best possible care.
KEY TAKEAWAYS
- Radiographs should be used as an adjunct in diagnosis. Recognize their limitations and use them as one of the objective measures. Treatment should not be based solely on the radiographic appearance.
- Appreciate the complexity of mandibular anterior and bicuspid teeth. They are oftentimes considered simple to treat based on their two-dimensional radiographic appearance; However, these teeth have a high incidence of multiple canals, and, oftentimes, the canal shape is much broader than presumed.
- Ensure straight-line access has been established prior to measuring working length. Take the time to open the coronal portion of the canal to make it is easier and more suitable to establish working length.
- Electronic apex locators have been shown to provide a reliably accurate assessment of working length. In a small percentage of cases, however, these devices can produce errors. A corroborating radiograph is strongly recommended in all circumstances. When there are small discrepancies between the two, it is advisable to trust the apex locator.
- Prescribe antibiotics only when indicated. Antibiotics are not effective at preventing flare-ups and are not indicated for cases of irreversible pulpitis.
REFERENCES
- McCarthy PJ, McClanahan S, Hodges J, Bowles WR. Frequency of localization of the painful tooth by patients presenting for an endodontic emergency. J Endod. 2010;36:801–805.
- Falace DA, Reid K, Rayens MK. The influence of deep (odontogenic) pain intensity, quality, and duration on the incidence and characteristics of referred orofacial pain. J Orofac Pain. 1996;10:232–239.
- Halse A, Molven O, Fristad I. Diagnosing periapical lesions: disagreement and borderline cases. Int Endod J. 2002;35:703–709.
- Bender IB, Seltzer S. Roentgenographic and direct observation of experimental lesions in bone I. J Am Dent Assoc. 1961;62:152–160.
- Bender IB, Seltzer S. Roentgenographic and direct observation of experimental lesions in bone II. J Am Dent Assoc. 1961;62:708–716.
- Miller SO, Johnson JD, Allemang JD, Strother JM. Cold testing through full coverage restorations. J Endod. 2004;30:695–700.
- Vertucci FJ. Root canal morphology and its relationship to endodontic procedures. Endo Topics. 2005;10:3–29
- Siqueira JF, Arau ́jo MC, Garcia PF, Fraga RC, Dantas CJ. Histological evaluation of the effectiveness of five instrumentation techniques for cleaning the apical third of root canals. J Endod. 1997;23:499–502.
- Davis RD, Marshall JG, Baumgartner JC. Effect of early coronal flaring on working length change in curved canals using rotary nickel-titanium versus stainless steel instruments. J Endod. 2002;28:438–442.
- Goerig AC, Michelich RJ, Schultz HH. Instrumentation of root canals in molar using the step-down technique. J Endod. 1982;8:550–554.
- Stabholz A, Rotstein I, Torabinejad M. Effect of preflaring on tactile detection of the apical constriction. J Endod. 1995;21:92–94.
- Leeb J. Canal orifice enlargement as related to biomechanical preparation. J Endod. 1983;9:463–470.
- Qualtrough AJ, Dummer PM. Undergraduate endodontic teaching in the United Kingdom: an update. Int Endod J. 1997;30:234–239.
- Ibarolla JL, Chapman BL, Howard JH, Knowles KI, Ludlow MO. Effect of preflaring on root ZX apex locators. J Endod. 1999;25:625–626.
- Ricucci D and Langeland K. Apical limit of root canal instrumentation and obturation, part 2. A histological study. Int Endod J. 1998;31:394–409.
- Kuttler Y. Microscopic investigation of root apexes. J Am Dent Assoc. 1955;50:544–552.
- Green D. A stereomicroscopic study of 700 root apices of maxillary and mandibular posterior teeth. Oral Surg Oral Med Oral Path. 1960;13:728–733.
- Burch JG, Hulen S. The relationship of the apical foramen to the anatomic apex of the tooth root. Oral Surg Oral Med Oral Path. 1972;34:262–268.
- Martins JN, Marques D, Mata A, Carames J. Clinical efficacy of electronic apex locators: systematic review. J Endod. 2014;40:759–777.
- Kobayashi C, Suda H. New electronic canal measuring device based on the ratio method. J Endod. 1994;20:111–114.
- ElAyouti A, Weiger R, Lost C. Frequency of overinstrumentation with an acceptable radiographic working length. J Endod. 2001;27:49–52.
- Walton RE. Antibiotics: A Risky Prescription. Available at: aae.org/publications-and-research/communique/antibiotics–a-risky-prescription.aspx. Accessed July 10, 2017.
- Aminoshariae A, Kulild JC. Evidence-based recommendations for antibiotic usage to treat endodontic infections and pain. A systematic review of randomized controlled trials. J Am Dent Assoc. 2016;147:186–191.
- Keenan JV, Farman AG, Fedorowicz Z, Newton JT. A Cochrane systematic review finds no evidence to support the use of antibiotics for pain relief in irreversible pulpitis. J Endod. 2006;32:87–92.
- Sutherland S, Matthews DC. Emergency management of acute apical periodontitis in the permanent dentition: a systematic review of the literature. J Can Dent Assoc. 2003;69:160.
- Walton RE, Chiappinelli J. Prophylactic penicillin: Effect on post-treatment symptoms following root canal treatment of asymptomatic periapical pathosis. J Endod. 1993;19:466–470.
- Pickenpaugh L, Reader A, Beck M, Meyers WJ, Peterson LJ. Effect of prophylactic amoxicillin on endodontic flare-up in asymptomatic, necrotic teeth. J Endod. 2001;27:53–56.
- Henry M, Reader A, Beck M. Effect of penicillin on postoperative endodontic pain and swelling in symptomatic necrotic teeth. J Endod. 2001;27:117–123.
- Baumgartner JC. Antibiotics and the Treatment of Endodontic Infections. Available at: aae.org/uploadedfiles/publications_and_research/endodontics_colleagues_for_excellence_newsletter/summer06ecfe.pdf. Accessed July 10, 2017.
- Marra F, George D, Chong M, Sutherland S, Patrick DM. Antibiotic prescribing by dentists has increased: Why? J Am Dent Assoc. 2016;147:320–327.
- U.S. Centers for Disease Control and Prevention. Antibiotic Resistance Threats in the United States, 2013. Available at: http://www.cdc.gov/drugresistance/pdf/ar-threats-2013-508.pdf. Accessed July 10, 2017.
- Fluent MT, Jacobsen PL, Hicks LA. Considerations for responsible antibiotic use in dentistry. J Am Dent Assoc. 2016;147:683–686.
- Shehab N, Patel PR, Srinivasan A, Budnitz DS. Emergency department visits for antibiotic associated adverse events. Clin Infect Dis. 2008;47:735–743.
The author has no commercial conflicts of interest to disclose.
Featured Image by GEORGHANF/ISTOCK/GETTY IMAGES PLUS
From Decisions in Dentistry. August 2017;3(8):13–16, 18.



[…] Y. Five misconceptions in endodontics. Decisions in Dentistry. […]