Etiology and Treatment of Dentinal Hypersensitivity
A comprehensive review of sensitivity and therapeutic approaches fro clinical and home use.
Dentinal hypersensitivity is one of the most common patient complaints heard in dental practice.1 Affecting up to 57% of patients, it seems to peak between the ages of 20 and 40.2 The prevalence of hypersensitivity is slightly higher among women,3 with canines and premolars of both arches most commonly involved.4 The pain is often chronic and marked by acute episodes.5
According to Holland et al,6 dentinal hypersensitivity is defined as a “short, sharp pain arising from exposed dentin in response to stimuli, which are usually thermal, evaporative, tactile, and osmotic or chemical that usually cannot be attributed to any other form, dental defect or pathology.” Usually, the pain is localized and of short duration. This differs from pulpal pain, which is protracted, dull, aching, poorly localized, and lasts longer than the applied stimulus.
The distress caused by hypersensitivity can range from minor to severe. Patients experiencing severe hypersensitivity may find eating and drinking difficult, especially when consuming hot or cold substances. The pain experienced by individuals with sensitivity is highly subjective, however, and the intensity is episodic in nature. Unfortunately, patients are rarely able to isolate the relevant tooth.7 In order to isolate the causes of sensitivity, clinicians typically rely on exposing the suspected tooth to an air blast or hot or cold liquid to elicit a response. The use of a rubber dam to isolate the tooth can be helpful in this process.
HYDRODYNAMIC THEORY
Hydrodynamic theory is the most commonly agreed upon cause of dentinal hypersensitivity. Kramer8 and Brännström9 confirmed and expanded on this theory that establishes a relationship between applied pressure, air blasts and chemical stimuli to dentin fluid shifts that occur in response to these stimuli.7 In the original research, Brӓnnstrӧm ground through the enamel into the midcoronal dentin of premolars in children whose teeth were going to be extracted for orthodontic purposes. The children’s cross-sectional dentin tubules were exposed to saliva for a week, which resulted in increased sensitivity. Initially, a smear layer covered these teeth, but, by the end of the week, it had disappeared, thus making the dentin increasingly hyperconductive.9 Dentin permeability varies and can quickly decrease.10
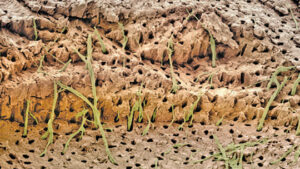
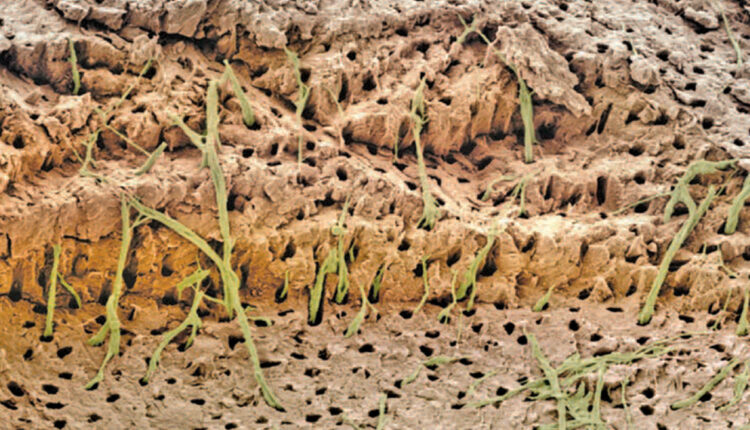
The evidence supporting hydrodynamic theory is based on in vivo studies in human and animal subjects. The distribution of nerves in dentinal tubules varies, with about 40% occurring over the pulp horns and a lesser percentage located in the cervical dentin. It appears that open dentinal tubules are necessary for exposed dentin to become sensitive (Figure 1, page 18); in fact, a patient’s sensitivity will increase in proportion to the number of large, open tubules present. Open tubules demonstrate high hydraulic conductance; conversely, fluid flow decreases if tubules are blocked. This provides a means for various treatment options. Magloire et al11 state that external stimuli result in dentinal fluid movement, and that odontoblasts and/or nerve complex reaction may be a distinctive mechanosensory system — providing a new role for odontoblasts as sensor cells. Information transfer between odontoblasts and axons may be the result of mediators in the gap space between odontoblasts and axons, as evidenced by nocioceptive-transducing receptors and trigeminal afferent fibers, and expression of reputed effectors by odontoblasts (Figure 2).11
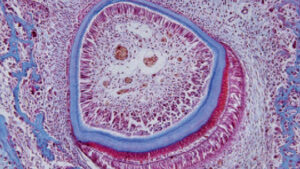
that lay down the dentin (thin blue layer), which makes up the bulk of the tooth. Enamel (thicker blue layer) is formed by ameloblasts, a layer of elongated cells around the outside of the enamel.
POSSIBLE CAUSES
Periodontal disease, gingival recession, cracked teeth, erosion, abrasion, abfraction and tooth fracture may cause hypersensitivity. All of these conditions result in exposed dentin, which creates an environment in which stimuli cause dentinal tubular fluid movement that activates nerve fibers, causing pain. The exposed dentin may result from removal of cervical cementum during scaling and root planing, finishing and polishing of restorations, or extreme toothbrushing — especially after the ingestion of acidic food or drinks. Regurgitation by patients with bulimia produces acid exposure, and subsequent brushing can lead to loss of tooth structure.1 Pain can be localized or general in nature, and may affect a variety of tooth surfaces, either together or individually.12
As noted, patients with periodontal disease are subject to dentinal hypersensitivity.13 Studies show the incidence of hypersensitivity increases one week following periodontal surgery, and resolves by eight weeks.14,15 Younger patients demonstrate more sensitivity compared to older adults, in whom sensitivity takes longer to resolve. Scaling and root planing can also cause sensitivity for several days following treatment.
Gingival recession results in exposure of root surfaces and possible sensitivity. Buccal bone provides most of the blood supply for the buccal gingiva, and any loss of buccal bone will result in a decrease in gingiva.14 Thin or fenestrated bone, tooth anatomy, tooth position or orthodontic movement may result in recession. Excessive toothbrushing with dentifrice can also cause recession.15 The signs and symptoms of cracked teeth may vary depending on the severity. Patients usually will experience acute pain with mastication, but, upon removal of the stimulus, the pain subsides. If the pain extends to the pulp or periodontal ligament, it will persist.16
Erosion is defined as a loss of enamel through chemical dissolution by acids that are not of bacterial origin. There are three types of erosion: extrinsic (e.g., diet, lifestyle or environment), intrinsic (e.g., gastric acid), and idiopathic.17 The unionized acid diffuses into the interprismatic areas of the enamel and dissolves the mineral in the subsurface area.18 In the initial stage, the tooth surface is dull due to demineralization, but the tooth is not hypersensitive because the dentin with open tubules is not exposed. Composite can be used to seal the enamel to restore the normal contour and prevent dentin exposure.19 Restoring the tooth will improve oral hygiene and reduce possible pulpal involvement, toothbrush/ dentifrice abrasion and acid erosion.20
Abrasion is the loss of tooth structure by mechanical forces from a foreign element, and it may cause sensitivity.21 Attrition is tooth-to-tooth contact, which results from occlusal function or parafunction — such as bruxism — and can cause loss of tooth structure on the occlusal surfaces and incisal edges.22
The role of abfraction in the etiology of dentinal hypersensitivity is controversial. Abfraction may occur when disproportionate cyclic, nonaxial tooth loading leads to cusp flexure and concentration of stresses in the susceptible cervical region of teeth. These cervical lesions, caused by occlusal stresses, lead to weakening of the cervical tooth structure and can cause enamel, cementum or dentin to chip away from the cervical aspect.23 Lee and Eakle24 first described lesions that may result from tensile stresses. They established that an abfraction lesion is often located at or near the fulcrum in the area with the greatest concentration of tensile stress; it is typically wedge-shaped and displays a size proportional to the degree and incidence of applied tensile force.
Others, however, contend there is limited evidence supporting abfraction as a primary cause of what are now referred to as noncarious cervical lesions. The noncarious processes that result in loss of hard tissue at the cervical area appear to be multifactorial, and may include abrasion, acidic challenges and possibly abfraction.23 While the available evidence suggests these lesions may develop from forces, including occlusal forces, acting alone or in combination,23 additional studies are warranted to determine the exact role of abfraction in noncarious lesions and any related sensitivity.
In addition to these sources, tooth whitening often causes dentinal hypersensitivity. The use of hydrogen peroxide or carbamide peroxide may infiltrate through enamel and dentin to the pulp. The glutathione peroxidase and catalase in the pulp do not have enough time to inactivate the hydrogen peroxide, which may cause sensitivity. It should also be noted that all bleaching gels are hypertonic and osmotically draw water from the pulp through the dentin and enamel to the whitening agent. This can potentially stimulate intradental nerves.25
TREATMENT OPTIONS
Clinicians can recommend a variety of treatments for at-home use or in office application (see Table 1 for a treatment option flowchart).26 The mechanism of action is typically nerve desensitization, protein precipitation, plugging dentinal tubules, sealing dentin, or ablating dentin with a laser. The most commonly used therapies include anti-inflammatory agents, protein precipitants, tubule-occluding agents and tubule sealants. The most conservative approach should be implemented initially, with more aggressive treatments suggested if relief is not achieved.27
TABLE 1. Strategies for the Treatment of Dentinal Hypersensitivity26 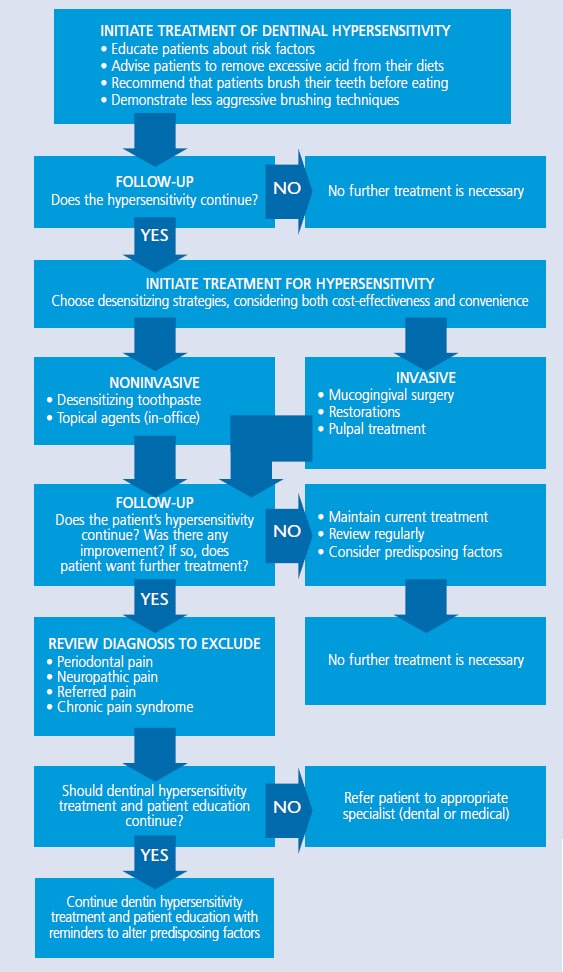 As a first line of defense against sensitivity, clinicians may wish to recommend a dentifrice with potassium salts (e.g., potassium nitrate, potassium chloride or potassium citrate), strontium salts (e.g., strontium chloride or strontium acetate), or fluoride. Potassium ions diffuse along the dentinal tubules, which block nerve action and dull the pain associated with hypersensitivity. Potassium salt dentifrices are effective, but may require two weeks of consistent use for patients to feel the effects.28 By comparison, the method of action found in strontium salt dentifrices or fluoride is tubule occlusion. Fluoride in various forms is an effective strategy for at-home relief of sensitivity. Sodium fluoride, stannous fluoride, and sodium monofluorophsophate all block dentinal tubules, reducing sensitivity. Prescription fluoride dentifrices and tray application may also be helpful.29Calcium phosphate technologies are another option for treating hypersensitivity. Amorphous calcium phosphate makes calcium and phosphate ions available in saliva to accelerate remineralization, and may help minimize whitening-induced sensitivity. It is available in gel and whitening products.29 In addition, products containing casein phosphopeptide-amorphous calcium phosphate (CPP-ACP) can be used to partially occlude dentinal tubules. Although pretreatment with a desensitizing agent will sometimes interfere with restorative bonding, a study by Borges et al30 found that paste with CPP-ACP did not negatively affect bond strength and, in some instances, improved it. Calcium sodium phosphosilicate can likewise aid in the infiltration and remineralization of tubules. The silica in the material performs as a nucleation position for the precipitation of calcium and phosphate.31 Tricalcium phosphate (TCP) is the most recent addition to the family of calcium phosphate technologies. Providing a slow release of calcium to the tooth surface, TCP is designed to boost the remineralizing effects of fluoride, which may also decrease sensitivity. It is available in a prescription dentifrice and fluoride varnish.32
As a first line of defense against sensitivity, clinicians may wish to recommend a dentifrice with potassium salts (e.g., potassium nitrate, potassium chloride or potassium citrate), strontium salts (e.g., strontium chloride or strontium acetate), or fluoride. Potassium ions diffuse along the dentinal tubules, which block nerve action and dull the pain associated with hypersensitivity. Potassium salt dentifrices are effective, but may require two weeks of consistent use for patients to feel the effects.28 By comparison, the method of action found in strontium salt dentifrices or fluoride is tubule occlusion. Fluoride in various forms is an effective strategy for at-home relief of sensitivity. Sodium fluoride, stannous fluoride, and sodium monofluorophsophate all block dentinal tubules, reducing sensitivity. Prescription fluoride dentifrices and tray application may also be helpful.29Calcium phosphate technologies are another option for treating hypersensitivity. Amorphous calcium phosphate makes calcium and phosphate ions available in saliva to accelerate remineralization, and may help minimize whitening-induced sensitivity. It is available in gel and whitening products.29 In addition, products containing casein phosphopeptide-amorphous calcium phosphate (CPP-ACP) can be used to partially occlude dentinal tubules. Although pretreatment with a desensitizing agent will sometimes interfere with restorative bonding, a study by Borges et al30 found that paste with CPP-ACP did not negatively affect bond strength and, in some instances, improved it. Calcium sodium phosphosilicate can likewise aid in the infiltration and remineralization of tubules. The silica in the material performs as a nucleation position for the precipitation of calcium and phosphate.31 Tricalcium phosphate (TCP) is the most recent addition to the family of calcium phosphate technologies. Providing a slow release of calcium to the tooth surface, TCP is designed to boost the remineralizing effects of fluoride, which may also decrease sensitivity. It is available in a prescription dentifrice and fluoride varnish.32
IN-OFFICE APPLICATION
Simple in-office therapies may be helpful in addressing sensitivity. Clinicians can apply a desensitizing prophylaxis paste formulated with 8% arginine and calcium carbonate to occlude tubules with plugs of arginine, calcium, phosphate and carbonate. It has been shown to endure normal pulpal pressure and acidic challenges, successfully minimizing dentinal tubule fluid flow and, thus, sensitivity,33 without harming bond strength.34
Fluoride varnish allows for the slow and continuous release of fluoride. Varnishes provide a natural resin-based vehicle for fluoride. Calcium fluoride is deposited on the tooth surface, resulting in the formation of fluorapatite. The addition of potassium oxalate causes the formation of acid-resistant calcium oxalate after reaction with the calcium of dentin.35 Extended-contact varnish is a photocured fluoride varnish that can be utilized to decrease dentinal hypersensitivity. It consists of a resin modified glass ionomer that incorporates glycerophosphate with fluoride release. It also encourages resin tag formation, allowing instantaneous and long-term occlusion of the tubules. Another varnish product combines 5% glutaraldehyde and 35% hydroxyethylmethacrylate. It acts as a biological fixative and creates a coagulation of plasma proteins in the tubules, blocking the openings. Capable of reducing sensitivity for at least three months,,sup>36 it can infiltrate acid-etched and moist dentin. These varnishes are compatible with adhesives, cements, and restorative and core build-up materials.37
Chlorhexidine varnish forms a mechanical barrier after drying, which reduces sensitivity, while providing an antiplaque and antibacterial action.38
Oxalates have also been shown to diminish dentinal permeability and block tubules.39 The oxalate reacts with the calcium ions in dentin to form calcium oxalate crystals in tubules and on the surface. The effect is reduced over time, however, as the crystals are removed by brushing and dietary acids. Etching improves the infiltration of calcium oxalate crystals into the tubules.39
Clinicians can also use a light-cured desensitizer that reduces sensitivity through the reaction of the monomer with calcium in the tooth and resin tag formation, resulting in tubule occlusion. A curing light subsequently polymerizes the resin layer. It can be used for immediate dentin sealing of prepared teeth.
INVASIVE TREATMENT OPTIONS
When less invasive therapies are not effective, bonding, grafting and laser treatment may provide relief from dentinal hypersensitivity. Bonding agents can be used to simultaneously desensitize and bond.40 Depending on the product used, a potential disadvantage to this approach is the need for phosphoric acid prior to placing the bonding agent, which may require the use of anesthesia.
An alternative is to use a self-etch adhesive. Another available bonding agent blocks dentinal tubules, and contains triclosan to minimize plaque formation.41 An alternative mode of desensitization is to cover exposed root surfaces with grafting. This should be considered prior to bonding techniques, however, because bonded restorations might preclude a successful graft.42 The use of Nd:YAG lasers provides thermal energy absorption on dentin, which may result in occlusion or narrowing of dentinal tubules.39,43
SUMMARY
Dentinal hypersensitivity is caused by exposed dentin in which stimuli trigger dentinal tubule fluid movement that activates nerve fibers to cause pain. The relationship between surface and intratubular precipitation and moderation of sensitivity is not straightforward. It is not the quantity of precipitate, but, rather, the quality, density, porosity, depth of penetration, and strength of attachment to the dentin that affects the results.40 Ultimately, treatment efficacy is determined by how long the diminution or elimination lasts.
Treatment decisions should be based on dentinal hypersensitivity severity and etiology. Some in-office treatments bring immediate relief that can be followed with a variety of at-home remedies. A combination of techniques may be warranted to provide long-term relief.
KEY TAKEAWAYS
-
- Hydrodynamic theory is the most commonly agreed upon cause of dentinal hypersensitivity.
- Open dentinal tubules demonstrate high hydraulic conductance, thus contributing to sensitivity; conversely, fluid flow decreases if tubules are blocked. This provides a means for various approaches to treatment.
- The prevalence of this condition is slightly higher among women,3 with canines and premolars of both arches most commonly involved.4
- Typically, pain from hypersensitivity is localized and of short duration; this differs from pulpal pain, which is protracted, dull, aching, poorly localized, and lasts longer than the applied stimulus.
- The use of a rubber dam can be helpful when attempting to isolate the tooth — or teeth — subject to hypersensitivity.
- Clinicians can recommend a variety of therapies for at-home use or in-office application. The most common treatments include anti-inflammatory agents, protein precipitants, tubule-occluding agents and tubule sealants.
- Treatment decisions should be based on sensitivity severity and etiology, and a combination of techniques may be warranted to provide effective relief.
REFERENCES
-
-
- Addy M. Dentine hypersensitivity: New perspectives on an old problem. Int Dent J.2002;52:367–375.
- Addy M. Etiology and clinical implications of dentine hypersensitivity. Dent Clin North Am. 1990;34:503–514.
- Flyn J, Galloway R, Orchardson R. The incidence of hypersensitive teeth in the West of Scotland. J Dent. 1985;13:230–236.
- Addy M, Mostafa P, Newcombe RG. Dentine hypersensitivity: The distribution of recession, sensitivity and plaque. J Dent. 1987;15:242–248.
- Dababneh RH, Khouri AT, Addy M. Dentine hypersensitivity — an enigma? A review of terminology, mechanisms, aetiology and management. Br Dent J.1999;187:606–611.
- Holland GR, Narhi MN, Addy M, Gangarosa L, Orchardson R. Guidelines for the design and conduct of clinical trials on dentine hypersensitivity. J Clin Periodontol. 1997;24:803–813.
- Li Y. Innovations for combating dentin hypersensitivity: current state of the art. Compend Contin Educ Dent. 2012;33(Suppl):10–16.
- Kramer IRH. The relationship between dentine sensitivity and movements in the contents of dentinal tubules. Br Dent J. 1955;98:391–392.
- Brännström M. The elicitation of pain in human dentine and pulp by chemical stimuli. Arch Oral Biol. 1962;7:59–62.
- Pashley DH. Dentin-predentin complex and its permeability: physiologic overview. J Dent Res. 1985;64(Suppl):613–620.
- Magloire H, Maurin JC, Couble ML, et al. Topical review. Dental pain and odontoblasts: facts and hypotheses. J Orofac Pain. 2010;24:335–349.
- Camilotti V, Zilly J, Nassar CA, Nassar PO. Desensitizing treatments for dentin hypersensitivity: a randomized, split-mouth clinical trial. Braz Oral Res. 2012;26:263–268.
- Chabanski MB, Gillam DG, Bulman JS, Newman HN. Prevalence of cervical dentine sensitivity in a population of patients referred to a specialist periodontology department. J Clin Periodontol. 1996;23:989–992.
- Uchida A, Wakano Y, Fukuyama O, Miki T, Iwayama Y, Okada H. Controlled clinical evaluation of a 10% strontium chloride dentifrice in treatment of dentin hypersensitivity following periodontal surgery. J Periodontol. 1980;51:578–581.
- Absi EG, Addy M, Adams D. Dentine hypersensitivity — the effect of toothbrushing and dietary compounds on dentine in vitro: an SEM study. J Oral Rehabil. 1992;19:101–110.
- Kahler W. The cracked tooth conundrum: terminology, classification, diagnosis, and management. Am J Dent. 2008;21:275–282.
- Bartlett DW. The role of erosion in tooth wear: aetiology, prevention and management. Int Dent J. 2005;55(Suppl):277–284.
- Zero DT, Lussi A. Erosion — chemical and biological factors of importance to the dental practitioner. Int Dent J. 2005;55:285–290.
- Lambrechts P, Van Meerbeek B, Perdigão J, Gladys S, Braem M, Vanherle G. Restorative therapy for the erosive lesion. Eur J Oral Sci. 1996;104:229–240.
- Grippo JO. Noncarious cervical lesions: the decision to ignore or restore. J Esthet Dent. 1992;4:55–64.
- Abrahamsen TC. The worn dentition — pathognomonic patterns of abrasion and erosion. Int Dent J. 2005;55(Suppl):268–276.
- McIntyre F. Restoring esthetics and anterior guidance in worn anterior teeth. A conservative multidisciplinary approach. J Am Dent Assoc. 2000;131:1279–1283.
- Michael JA, Townsend GC, Greenwood LF, Kaidonis JA. Abfraction: separating fact from fiction. Aust Dent J. 2009;54:2–8.
- Lee WC, Eakle WS. Possible role of tensile stress in the etiology of cervical erosive lesions of teeth. J Prosthet Dent. 1984;52:374–380.
- Swift EJ Jr. Tooth sensitivity and whitening. Compend Contin Dent Educ Dent.2005;26(Suppl):4–10.
- Canadian Advisory Board on Dentin Hypersensitivity. Consensus-based recommendations for the diagnosis and management of dentin hypersensitivity. J Can Dent Assoc. 2003;69:221–226.
- Al-Saud LM, Al-Nahedh HN. Occluding effect of Nd:YAG laser and different dentin desensitizing agents on human dentinal tubules in vitro: a scanning electron microscopy investigation. Oper Dent. 2012;37:340–355.
- Poulsen S, Errboe M, Hovgaard O, Worthington HW. Potassium nitrate toothpaste for dentine hypersensitivity. Cochrane Database Syst Rev. 2001;2:CD001476.
- Orchardson R, Gillam DG. Managing dentin hypersensitivity. J Dent Assoc. 2006;137:990–998.
- Borges BC, Souza-Junior EJ, da Costa Gde F, et al. Effect of dentin pretreatment with a casein phosphopeptideamorphous calcium phosphate (CPP-ACP) paste on dentin bond strength in tridimensional cavities. Acta Odontol Scand. 2013;71:271–277.
- Forsback AP, Areva S, Salonen JI. Mineralization of dentin induced by treatment with bioactive glass S53P4 in vitro. Acta Odontol Scand. 2004;62:14–20.
- Karlinsey RL, Mackey AC. Solid-state preparation and dental application of an organically-modified calcium phosphate. J Mater Sci. 2009;44:346–349.
- Panagakos F, Schiff T, Guignon A. Dentin hypersensitivity: effective treatment with an in-office desensitizing paste containing 8% arginine and calcium carbonate. Am J Dent. 2009;22(Suppl):3A–7A.
- García-Godoy A, García-Godoy F. Effect of an 8.0% arginine and calcium carbonate in-office desensitizing paste on the shear bond strength of composites to human dental enamel. Am J Dent. 2010;23:324–326.
- Camilotti V, Zilly J, Busato Pdo M, Nassar CA, Nassar PO. Desensitizing treatments for dentin hypersensitivity: a randomized, split-mouth clinical trial. Braz Oral Res. 2012;26:263–268.
- Sethna GD, Prabhulji MLV, Karthikeyan BV. Comparison of two different forms of varnishes in the treatment of dentine hypersensitivity: a subject-blind randomized clinical study. Oral Health Prev Dent. 2011;9:143–150.
- Dijkman GE, Jongebloed WL, de Vries J, Ogaard B, Arends J. Closing of dentinal tubules by glutaraldehyde treatment, a scanning electron microscopy study. Scand J Dent Res. 1994;102:144–150.
- Sköld-Larsson K, Sollenius O, Petersson LG, Twetman S. Effect of topical applications of a novel chlorhexidine-thymol varnish formula on mutans streptococci and caries development in occlusal fissures of permanent molars. J Clin Dent. 2009;20:223–226.
- Lan WH, Lee BS, Liu HC, Lin CP. Morphologic study of Nd:YAG laser usage in treatment of dentinal hypersensitivity. J Endod. 2004;30:131–134.
- Ide M, Morel AD, Wilson RF, Ashley FP. The role of a dentine-bonding agent in reducing cervical dentine sensitivity. J Clin Periodontol. 1998;25:286–290.
- Yu X, Liang B, Jin X, Fu B, Hannig M. Comparative in vivo study on the desensitizing efficacy of dentin desensitizers and one-bottle self-etching adhesives. Oper Dent. 2010;35:279–286.
- Douglas de Oliveira DW, Marques DP, Aguiar-Cantuária IC, Flecha OD, Gonçalves PF. Effect of surgical defect coverage on cervical dentin hypersensitivity and quality of life. J Periodontol. 2013;84:768–775.
- Orhan K, Aksoy U, Can-Karabulut DC, Kalender A. Low-level laser therapy of dentin hypersensitivity: a short-term clinical trial. Lasers Med Sci. 2011;26:591–598.
-
Featured photo courtesy of STEVE GSCHMEISSNER/SCIENCE SOURCE
The authors have no commercial conflicts of interest to disclose.
From Decisions in Dentistry. December 2016:2(12);18, 21-24.


