Demystifying Desquamative Gingivitis: Diagnosis and Management
The ability to properly diagnose desquamative gingivitis will improve outcomes for this patient population.
This course was published in the June 2020 issue and expires June 2023. The author has no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Describe desquamative gingivitis, along with its various clinical presentations and symptoms.
- Explain patient groups most commonly affected by desquamative gingivitis, and the percentage of cases diagnosed as lichen planes, mucous membrane pemphigoid or pemphigus vulgaris.
- Discuss diagnosis, testing, and treatment of desquamative gingival lesions.
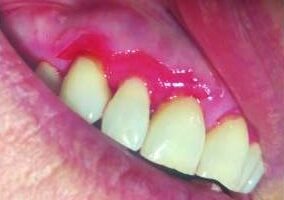
Desquamative gingivitis is a clinical term used to describe an erythematous, erosive and ulcerated appearance of the gingiva.1 Patients with desquamative gingival lesions will often complain of mucosal sloughing, gingival bleeding and oral discomfort, especially when consuming foods or beverages that may be acidic or spicy. Despite a combination of relatively uniform subjective complaints and objective clinical findings, clinicians should remember desquamative gingivitis does not represent a single diagnosis; rather, it is a descriptive term. In the majority of cases, a patient with desquamative gingivitis will likely be diagnosed with erosive lichen planus, mucous membrane pemphigoid (MMP) or pemphigus vulgaris (PV).1 This paper will discuss the clinical features, methods of diagnosis, and treatment protocols of each.
ORAL LICHEN PLANUS
Lichen planus is a chronic mucocutaneous condition seen most often in middle-aged adults, with a slight female predilection.2 Although it is sometimes categorized as an autoimmune condition, the exact etiology of lichen planus is unknown. It has been theorized it results from aberrant activation of CD4+ T-lymphocytes, which results in the characteristic clinical manifestations.2
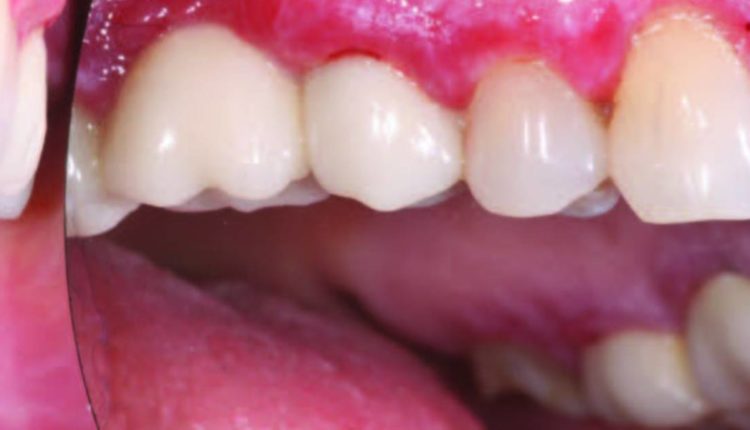
When lichen planus involves the oral mucosa, it may present with or without symptoms. Asymptomatic oral lichen planus is often reticular in appearance, while reticular lichen planus presents with white, lace-like striations known as Wickham striae.2 The buccal mucosa is most commonly affected (Figure 1A); however, any oral site can be involved. In some cases, lesions may be limited to the gingiva. Frequently, the lesions of oral lichen planus are bilateral and/or multifocal in appearance. The erosive form of lichen planus is seen less frequently than reticular lichen planus, however, it is more significant in that it is usually symptomatic and uncomfortable. Lesions of erosive lichen planus are frequently mixed red and white in appearance (Figure 1B), although in some patients an erythematous, ulcerated appearance will predominate. Even in these cases, focal Wickham striae can often be appreciated.
red and white lesions involving the alveolar mucosa.COURTESY DAVID MOISA, DDS, MS
MUCOUS MEMBRANE PEMPHIGOID
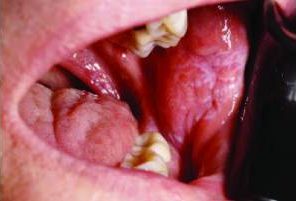
Also known as cicatricial pemphigoid, MMP is a chronic autoimmune disease of unclear etiology. It results from autoantibody production which targets hemidesmosomes and causes a subepithelial separation.3 Clinically, this results in the formation of vesicles or bullae that rupture, leading to ulceration and erythema (Figure 2). Similar to oral lichen planus, MMP is seen most commonly in middle-aged females, and is also considered to be a mucocutaneous disease.3
The intraoral presentation of MMP is also comparable to that of erosive lichen planus. Desquamative gingivitis is a frequent finding; in many cases, MMP lesions may be limited to the gingiva.4 Involvement of non-gingival sites may still be seen, and this does not exclude a diagnosis of MMP. Although blister formation may be seen, intraoral bullae will often rupture quickly, leading to the formation of mucosal erythema and ulceration. This condition has a positive Nikolsky sign, meaning that firm pressure applied on clinically normal tissue can induce blister formation.4
PEMPHIGUS VULGARIS
This is a severe, progressive autoimmune disease caused by autoantibodies directed against intraepithelial desmosomes.3 The resulting intraepithelial clefting produces the clinical lesions of PV, which appear as vesicles and bullae involving the skin and mucosa. Because the microscopic separation in PV occurs more superficially compared to MMP, the blisters that result are more prone to rupture and ulceration. This disease is seen most often in middle-aged adults; however, no clear gender predilection has been reported. It occurs with increased frequency in patients of Mediterranean, Ashkenazi Jewish or South Asian descent. The reported incidence is approximately 1 in 5 million.5
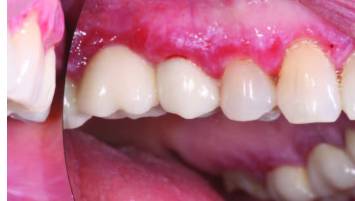
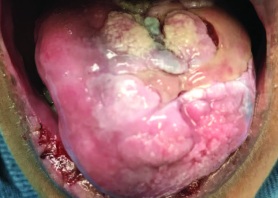
Oral lesions of PV present as fragile vesicles that quickly rupture, resulting in ulcerations with surrounding erythema. The ulcers can be quite large, and some authors have described them as having irregular, ragged borders (Figure 3).5 Any oral site may be involved, and the presentation of PV is often bilateral and/or multifocal. Involvement of the gingiva produces desquamative gingivitis similar to lichen planus and MMP. As with MMP, PV is a Nikolsky positive process. The oral lesions of PV often precede the development of cutaneous manifestations — in some cases by almost a year. They are also frequently slower to resolve than skin lesions.5 In extreme cases, patients with chronic, undiagnosed PV may present with weight loss because the lesions may interfere with the ability to maintain proper nutrition. Mortality from PV has been infrequently reported.
OTHER DIAGNOSTIC CONSIDERATIONS
Lichen planus, MMP and PV represent approximately 90% of the cases that clinically present as desquamative gingivitis.1,3 Additional conditions that may produce similar oral manifestations include lichenoid mucositis, systemic lupus erythematous, erythema multiforme, graft versus host disease, and paraneoplastic pemphigus, among others.3,6 While these entities are outside the scope of this discussion, they should be considered in the clinical differential diagnosis when deemed appropriate.
Due to the similarities in the clinical appearances of lichen planus, MMP and PV (as well as the other less frequently encountered chronic oral vesiculo-erosive processes), tissue biopsy remains the gold standard for diagnosis. Arriving at a single diagnosis is critical, because despite some overlap in treatment, these entities are associated with different long-term prognoses and extraoral manifestations. Additionally, the expected response to certain medications may be different, depending on the specific diagnosis.
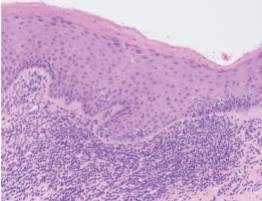
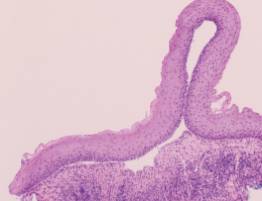
The histopathologic findings with routine hematoxylin and eosin staining are unique for lichen planus, MMP and PV, and are reflective of their presumed underlying etiologies. Lichen planus will demonstrate a dense, linear band of lymphocytes directly subjacent to the surface epithelium (Figure 4A). There is often migration of lymphocytes from the connective tissue into the epithelial layers (a process called exocytosis), which results in liquefactive degeneration of the basal (bottommost) layer of the epithelium.2,3 This microscopic presentation is consistent with the proposed cytotoxic T lymphocyte-induced etiology of lichen planus. Clinically, MMP will demonstrate a neat subepithelial separation, which is caused by the autoantibodies targeting the hemidesmosomes connecting the epithelium and underlying connective tissue (Figure 4B).3,4 An intraepithelial separation is observed in PV, resulting from autoantibodies directed against desmosomal attachment proteins within the epithelium (Figure 4C).3,5 With this type of separation, rounded-appearing acantholytic epithelial cells, known as Tzanck cells, may be seen.3,5
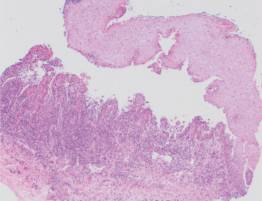
In theory, when these classic microscopic findings are identified on biopsy, a definitive diagnosis should be established. While this is often the case, the reality is that significant overlap of histologic features may be observed (Figure 5). For example, an artifactual separation of tissue — either during the biopsy procedure or when the specimen is being processed — can closely mimic a cleft of MMP or PV. Similarly, an inflammatory infiltrate resembling that seen in lichen planus can occasionally be observed in other conditions. For these and other reasons, a clinician receiving a biopsy report for a desquamative lesion may be disappointed to see a nonspecific diagnosis, along with a comment recommending either repeat biopsy or submission of tissue for direct immunofluorescence (DIF). This test uses fluorescent antibodies targeted against specific proteins to establish a diagnosis. With regard to oral vesiculo-erosive diseases, DIF can further confirm a diagnosis of MMP or PV, as these conditions have unique immunofluorescence profiles. Unlike a standard tissue biopsy, which is placed in formalin, DIF requires submission in a medium known as Michel’s solution.
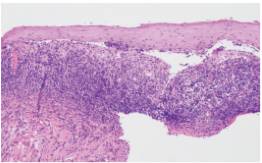
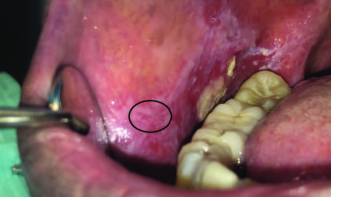
Selecting an appropriate biopsy site is critical for obtaining a good diagnostic sample. This can be extremely challenging in multifocal disease processes, such as lichen planus, MMP and PV. The most diagnostic tissue is not necessarily the worst looking area clinically, and these areas should be avoided if possible. Similarly, ulcerated areas have reduced diagnostic quality when compared to non-ulcerated lesional tissue. If an ulcerated site is to be biopsied, taking a perilesional sample from the edge of the ulceration, as opposed to the center, is necessary to obtain diagnostic tissue. The reason for this is an ulcer, by definition, lacks a surface epithelium, and diagnosis of these conditions relies on being able to analyze the relationship between the epithelium and underlying connective tissue. Therefore, a biopsy taken from the center of an ulcer will likely result in a diagnosis of nonspecific ulceration and inflammation. The best sites to biopsy are those that are mixed red and white in appearance; a portion of the lesion demonstrating a clinically visible Wickham striae-like pattern is of high diagnostic quality (Figure 6).
As noted, the majority of cases that appear as desquamative gingivitis are lichen planus, MMP or PV. Of these, lichen planus is seen most frequently. In cases of erosive lichen planus, identification of Wickham striae is a helpful, albeit nondiagnostic, clinical finding. Clinically, MMP may be diagnosed more often in patients presenting with desquamative gingivitis and without other oral lesions.
TREATMENT GUIDELINES
Administered as a rinse, gel or spray, topical steroids most often serve as the first line of treatment for desquamative gingival lesions. Due to the multifocal nature of these diseases, a steroid rinse is best able to provide coverage of all affected sites without the need for difficult or tedious application by the patient.3 If symptoms have not improved, stronger topical and/or systemic therapies can be prescribed. If the patient reports symptomatic relief, he or she should be encouraged to use the rinse only as needed for flare-ups. Additionally, if symptoms have mostly improved, but now can be localized to one or only a few sites, a topical steroid gel can be prescribed for direct topical application. Nonsteroidal therapies, such as tacrolimus and apremilast, are also emerging as potential treatments for chronic oral ulcerative processes refractory to topical steroids.2,7 If a diagnosis of PV is suspected, short courses of systemic steroids should be avoided, as this may lead to a rebound effect and worsening of the lesions after cessation of therapy.
As with any disease process, patient education is an essential part of effective management. Patients should be informed that lichen planus, MMP and PV are chronic conditions. In other words, one should not expect to see complete resolution of the clinical lesions with the prescribed medications, although reduction in severity of erythema and ulceration often occurs. The success of any particular treatment should be gauged by improvement in symptoms. In this vein, no treatment is recommended if a patient is asymptomatic. Patients should be informed that lesions associated with these conditions will naturally come and go, and not to be alarmed if lesions spread from one site to another. Although there may be many different environmental triggers for these conditions — including, but not limited to, increased physiological or psychological stress and certain types of foods or beverages — there are no items that patients must absolutely avoid. If and/or when patients experiences a worsening of symptoms, they need only use the prescribed medications until they have relief. Prolonged, continuous use of a topical steroid rinse should be discouraged, as it may dry the mouth and make patients more prone to developing a candidal infection.
One of the principal reasons determining a definitive diagnosis is critical is that each of these conditions has its own set of long-term sequalae and extraoral manifestations. Patients with lichen planus should be informed of the small, but increased risk for developing oral squamous cell carcinoma.8 They should also be educated about the importance of continued long-term follow-up with an oral and maxillofacial pathologist. Approximately 25% of patients with MMP will develop ocular lesions that can cause blindness if left untreated; therefore, referral to both an ophthalmologist and a dermatologist is necessary for appropriate management of any eye and skin lesions, respectively. Patients with PV should be referred to a dermatologist, even in the absence of any cutaneous lesions, as the oral manifestations of PV may precede the development of any skin lesions by up to a year. Management of PV often requires strictly regimented dosing of systemic steroids or other forms of systemic immunosuppressive therapy.
A summary of the pertinent clinical and histologic findings, as well as the possible treatment modalities for lichen planus, MMP and PV, can be found in Table 1.
CONCLUSION
When a patient presents with desquamative gingivitis, it represents the start of a potentially challenging, albeit hopefully rewarding, diagnostic journey for patient and provider alike. Chronic oral vesiculo-erosive lesions can often remain undiagnosed or improperly treated for years, leading to patient dissatisfaction and reduced quality of life. The ability to properly recognize this disease process and arrive at a singular definitive diagnosis is essential for improved patient outcomes and care.
REFERENCES
- Rogers RS, Sheridan PJ, Nightingale SH. Desquamative gingivitis: clinical, histopathologic, immunopathologic, and therapeutic observations. J Amer Acad Dermatol. 1982;7:729–735.
- Lavanya N, Jayanthi P, Rao UK, Ranganathan K. Oral lichen planus: an update on pathogenesis and treatment. J Oral Maxillofac Pathol. 2011;15:127–132.
- Cheng YL, Gould A, Kurago Z, Fantasia J, Muller S. Diagnosis of oral lichen planus: a position paper of the American Academy of Oral and Maxillofacial Pathology. Oral Surg Oral Med Oral Pathol Oral Radiol. 2016;122:332–354.
- Bagan J, Muzio LL, Scully C. Mucous membrane pemphigoid. Oral Dis. 2005;11:197–218.
- Black M, Mignogna MD, Scully C. Pemphigus vulgaris. Oral Dis. 2005;11:119–130.
- Muller S. Oral lichenoid lesions: distinguishing the benign from the deadly. Moder Pathol. 2017;30:S54–S67.
- Bettencourt M. Oral lichen planus treated with apremilast. J Drugs Dematol. 2016;15:1026–1028.
- Fitzpatrick SG, Hirsch SA, Gordon SC. The malignant transformation of oral lichen planus and oral lichenoid lesions: a systematic review. J Am Dent Assoc. 2014;145:45–46.
From Decisions in Dentistry. June 2020;6(6):40–43.





