 DANIIL DUBOV/ISTOCK/GETTY IMAGES PLUS
DANIIL DUBOV/ISTOCK/GETTY IMAGES PLUS
Current Perspectives on Articaine in Dental Anesthesia
This hybrid agent offers distinct benefits over other local anesthetic drugs.
This course was published in the March 2022 issue and expires March 2025. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Discuss articaine’s hybrid ester and amide formulation, and this agent’s prevalence in dental anesthesia.
- Explain clinical situations and patient populations suited for articaine administration, as well as the risk of paresthesia with these injections.
- Describe the benefits of articaine in the dental setting.
Beginning with the approval of lidocaine in 1948, all but one of the amide local anesthetic (LA) drugs in dentistry was borrowed from medicine. Articaine was developed specifically for dentistry in 1976 and subsequently borrowed by medicine. Since its introduction, its popularity has increased steadily.
Articaine was approved for use in the United States by the U.S. Food and Drug Administration (FDA) in 2000 as a 4% formulation with 1:100,000 epinephrine. In 2006, 4% articaine, 1:200,000 epinephrine was approved. More than two decades later, articaine is second only to lidocaine in worldwide use, where it is estimated that for every five cartridges of lidocaine used, three cartridges of articaine are used.1–3 As the first hybrid LA with both ester and amide properties, articaine’s developers placed an emphasis on dentistry’s requirement for a profound and durable LA with relatively rapid detoxification.3
Pharmacologically, articaine requires a greater concentration compared to 2% lidocaine to achieve its enhanced effectiveness. Unlike 2% lidocaine formulations, a 3% articaine, 1:200,000 epinephrine formulation provides reliably effective anesthesia of all teeth except the mandibular molars.4 A 4% articaine formulation, on the other hand, provides greater overall effectiveness of the entire dentition and supporting structures, and similar or greater durations compared to 2% lidocaine.4 While not superior to other LA drugs in all respects, recent systematic reviews suggest articaine demonstrates superiority in a number of ways.3,4 For example, in specific applications such as pulpitis and mandibular infiltration, articaine’s efficacy exceeds all of the other LAs by a significant margin.4
Guidelines regarding the concentration of epinephrine to use in specific situations are addressed in FDA-approved manufacturer statements. According to articaine package inserts: “For most routine dental procedures, [articaine] containing epinephrine 1:200,000 is preferred. However, when more pronounced hemostasis or improved visualization of the surgical field are required, [articaine] containing epinephrine 1:100,000 may be used.”5,6 The durable effectiveness of articaine, 1:200,000 epinephrine is very similar to the durable effectiveness of its 1:100,000 epinephrine formulation, while significantly reducing cardiovascular risk.
UNIQUE PHARMACODYNAMICS
Key properties of articaine include its liposolubility and vigorous protein binding, which promote efficacy and diffusibility.7–9 Because of the increased effectiveness of its 4% formulations, reduced volumes are possible compared to many other LA drugs. Enhanced diffusibility can decrease the number of injections, as well. For example, buccal infiltrations of articaine often provide palatal and lingual anesthesia without the need for separate palatal or lingual injections.10 Reducing doses and decreasing the number of penetrations are possible when using articaine.
In the presence of cardiac compromise, articaine is useful because of its 1:200,000 epinephrine formulation. Adequate volumes of anesthetic can be administered while significantly decreasing cardiac stimulation.
Articaine is approved for use in children age 4 and older.5,6 Both patients and clinicians benefit from articaine’s increased diffusibility in buccal infiltrations, where the need for block anesthesia and separate palatal and lingual injections is significantly reduced.10
UNIQUE PHARMACOKINETICS
Articaine’s rapid metabolism influences patient safety. Its half-life has been reported to be dose and age dependent; that is, the more administered and the older the patient, the longer the half-life — up to about 45 minutes, which provides a significant decrease in the exposure time of sensitive tissues to the drug.11 Lidocaine and prilocaine half-lives, for example, average 90+ minutes.11–13 With a dose-related elimination range of ~15 to 45 minutes in healthy individuals, articaine has distinct advantages over the other amides.11–13 While one cartridge of articaine (containing twice the number of milligrams) is somewhat more toxic than one cartridge of lidocaine, when comparing the drugs milligram to milligram, articaine is reported to have only 60% of the toxicity of lidocaine.12,13 Articaine’s unique metabolic pathway suggests potential advantages in liver dysfunction, geriatric dentistry, pediatric dentistry, as well as pregnancy and nursing.5,6
Pregnancy — Professional judgment — which includes incorporating the most recent FDA labeling rules14 regarding drugs in pregnancy and provider consults when indicated — should guide decisions when considering articaine use in pregnancy. Package inserts approved by the FDA generally state: “Articaine-epinephrine should only be given during pregnancy when benefits outweigh risks.”5,6 Articaine’s rapid elimination half-life has been suggested to provide advantages over other LA drugs in pregnancy because it decreases the fetus’ exposure to toxic effects.15 While this opinion may prevail in time, decisions to use articaine in pregnancy must also consider previously demonstrated fetal alterations in animals that occurred when articaine doses of four times maximum human recommendations were administered. While this represents a significant overdose, when comparing relative LA drug safety in pregnancy, it was noted that similar overdoses of lidocaine and prilocaine did not cause any fetal alterations.5,6
Nursing — When considering use in nursing mothers, articaine’s rapid elimination half-life is especially reassuring, as it has a distinct advantage over all the other drugs. This is a significant benefit that is addressed in current articaine package inserts, which note: “when using [articaine], nursing mothers may choose to pump and discard breast milk for approximately four hours (based on plasma half-life) following an injection of articaine [to minimize infant ingestion] and then resume breastfeeding.”5,6 There is currently no other LA drug available in dental cartridges where this statement applies.
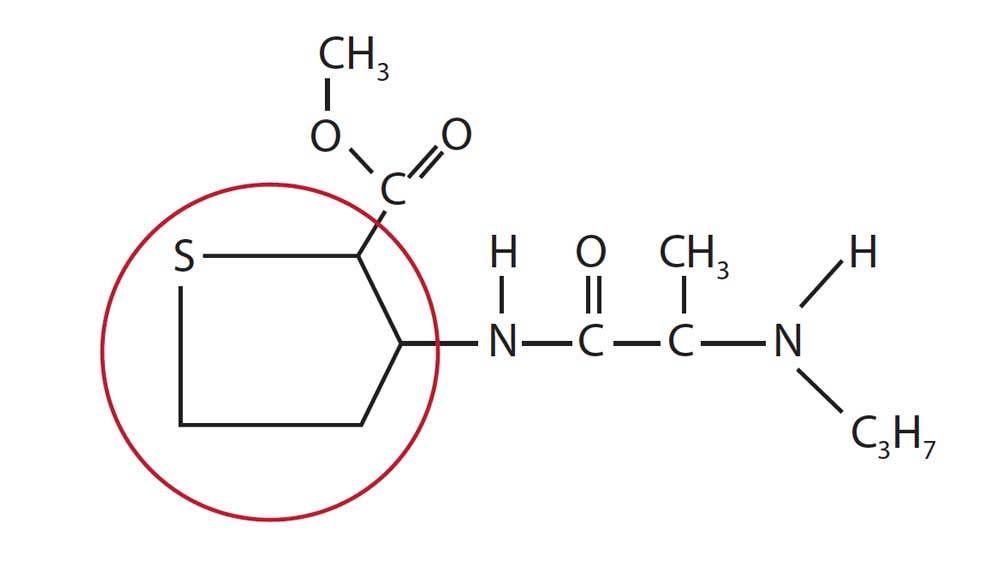
CLINICAL EFFECTIVENESS
As previously stated, in many cases articaine’s lipophilic properties make it possible to reduce doses and the number of penetrations to achieve anesthesia. Articaine’s thiophene ring (Figure 1), which increases its liposolubility, is thought to be responsible for much of its enhanced potency and diffusibility.16,17
Onset in Infiltration — Articaine has a rapid onset in infiltration, equal to or surpassing all other LA drugs. This has been reported to be anywhere from one to two minutes. The closest rival is mepivacaine at 90 seconds to two minutes, which, on occasion, may have a faster onset (but, overall, articaine’s onset is the fastest).16,17
Effectiveness in Infiltrations — In a 2021 systematic review with meta-analysis, articaine was found to be nearly three times (2.78) more effective than lidocaine in infiltration injections for routine dental treatment.4 This has been reported in both adult and pediatric patients, reducing the need for block anesthesia in many instances.4
Effectiveness in Inferior Alveolar Blocks — The same systematic review found articaine to be 1.5 times more effective than lidocaine in inferior alveolar (IA) blocks for routine dental treatment.4 Overall, considering both nerve blocks and infiltrations, articaine was found to be 2.17 times more effective compared to lidocaine.4
Effectiveness in Gow-Gates Blocks — The histology of the tissues within the infratemporal fossa and pterygomandibular triangle changes dramatically when contrasting the relatively lower injection heights of conventional IA nerve blocks to the higher injection heights of Gow-Gates blocks. The tissues at the level of the Gow-Gates nerve block are less dense and more lipid soluble in nature, which, together, favor articaine’s liposolubility and diffusibility.18
Effectiveness in Buccal Infiltrations — A number of studies have found articaine to be superior to lidocaine in buccal infiltrations for anesthesia and supplemental anesthesia of mandibular molars. In this technique, some suggest that lingual infiltrations in addition to buccal infiltrations may provide somewhat higher success rates, but information is limited.16 In the 2021 systematic review previously referenced, the following statement is made: “Articaine has a higher likelihood of anaesthesia success than lidocaine for mandibular molar buccal infiltration anaesthesia.”4
Palatal and Lingual Anesthesia Following Buccal Infiltrations — The relative diffusibility of articaine versus lidocaine was reported in a 2018 paper that examined relative concentrations of each drug in palatal tissues following buccal infiltration. Markedly higher concentrations of 4% articaine were found in palatal mucosa compared with 2% lidocaine solutions after buccal infiltrations.19
Success for palatal tissue anesthesia in scaling and root planing after buccal infiltration of articaine versus lidocaine was reported in a different 2018 study, in which the authors concluded, “The success rate for maxillary buccal infiltration to induce palatal anesthesia using articaine was 90% during scaling and root planing … and for [lidocaine] solution was 20% … The difference between the two agents was statistically significant.”20
In 100 children receiving articaine versus lidocaine for maxillary and mandibular primary molar extractions, both articaine and lidocaine were administered via buccal infiltration; 50 children received articaine and the other 50 received lidocaine. All children in the articaine group had sufficient anesthesia in palatal and lingual tissues during their procedures versus none of the children in the lidocaine group, all of whom required separate injections to anesthetize palatal and lingual tissues.10
Effectiveness in Pulpitis — A recent systematic review of all five LA drugs in cartridge form provides effectiveness ratings for each of the drugs. A total of 7981 studies were identified, of which 16 were analyzed to compare the drugs’ relative abilities to provide effective IA nerve blocks in patients with irreversible pulpitis of mandibular teeth. The rankings are considered to provide high-quality evidence of each drug’s effectiveness. A significant difference was observed in that articaine demonstrated much greater effectiveness in pulpitis than any of the other drugs.21 The average success rate for each of the drugs is provided in Table 1.
Articaine’s significantly greater effectiveness in pulpitis versus lidocaine is further supported by a 2020 umbrella review. Of the eight available systematic reviews comparing articaine to lidocaine efficacy in pulpitis, five were selected for analysis. The umbrella review concluded that “articaine is more effective than lidocaine for local anaesthesia of teeth with irreversible pulpitis undergoing root canal treatment.”22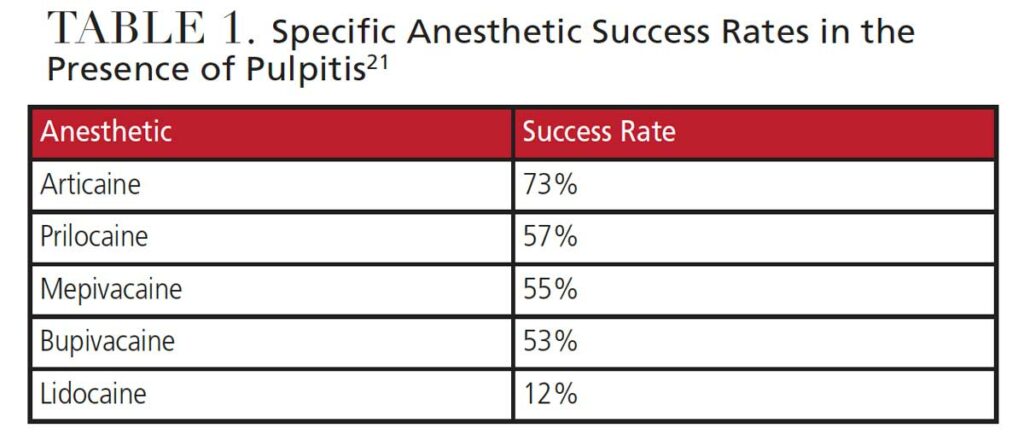
CURRENT PERSPECTIVES
Pediatric Use Under Age 4 — Information on articaine use in children under the age of 4 is limited. In clinical trials with 61 patients between the ages of 4 and 16, the safety and effectiveness of articaine were demonstrated.5,6 A 1989 retrospective survey in which 211 children under the age of 4 received 240 doses of articaine without reported adverse effects suggested that package inserts which stated there was no experience with children under the age of 4 could be modified on the basis of the survey.23 A subsequent 2010 systematic review, however, concluded there was insufficient data to support articaine use in very young children.9 Current package inserts in North America continue to state “the safety and effectiveness of [articaine HCL 4%] epinephrine 1:200,000 and 1:100,000 in pediatric patients below the age of 4 years have not been established.”5,6
Paresthesia — Paresthesia has been defined as lingering pain, numbness, increased sensitivity, and/or taste alterations resulting from nerve injury associated with LA injections.16 Despite widely conflicting opinions on the incidence of paresthesia associated with the use of articaine in IA and lingual nerve blocks, the long-term use of articaine in Europe since 1976 and more than two decades of use in the U.S. have not demonstrated exceptional levels of permanent paresthesia.24 Articaine’s market share in Germany, for example — with a population estimated at more than 83 million — has increased steadily since its inception to its level in 2021 of approximately 98%.3 The reported incidence of permanent paresthesia from articaine, according to the Pharmacovigilence Working Party of the European Medicines Agency, which includes Germany, is estimated to be one in 4.8 million.25
Although retrospective studies, case reports, and opinion pieces have associated articaine with a higher risk of paresthesia in the past, Malamed26 states “there is no scientific evidence that articaine is more neurotoxic than other local anesthetics.” In addition, a study of comparative cytotoxicity suggests “that among local anesthetics commonly used in dentistry, articaine … had the least neurotoxic effects … after an exposure corresponding to the dental practice.”27
The actual incidence of permanent paresthesia resulting from LA drugs, individually and collectively, is disputed, and, because of the rarity of permanent paresthesia, may likely remain unknown. Some authors have suggested rates as high as one in 140,000 for articaine, while others have suggested rates as low as one in 4.16 million.28
Despite a significant amount of encouraging research over the past two decades, whether or not to use articaine in IA blocks remains a matter of choice. Unfortunately, the potential causes of paresthesia are numerous, and some may not yet have been identified. Professional judgment is always advised.
Rethinking Injection Trauma — While some authors have referred to articaine’s 4% concentration and direct contact injury to nerves as the suspected primary causes of paresthesia, a somewhat different point of view may be inferred from other research.28–30 For example, a 2015 review notes, “Overall, when it comes down to scientifically sound research and data, no general clear evidence can be found to support the claim that articaine is associated with increased paresthesia because of its inherent characteristics … Based on the findings presented … procedural trauma appears to be a valid alternative explanation for the reported neurological complications.”30
Trauma from injection may be defined as physical injury to tissues during needle insertion and withdrawal, and possibly the impact of fluid dynamics during injection (specifically, increases in pressure when forcing solution out of cartridges rapidly). As noted, it has been suggested that procedural trauma may be a primary cause of paresthesia.30
A review of basic principles of fluid dynamics may provide an understanding of what occurs during dental injections with both manual syringes and computer-controlled LA delivery devices.16,31–34 The primary properties that impact fluid pressures exiting needle tips typically include flow velocity (rate), pressure (exerted either manually or computer controlled), and density (of tissues surrounding needle tips) as functions of time and space. When considering the nature of tissues at a given deposition site, the result may be a slow, gentle stream of anesthetic solution that does not trigger a pain response, or a high-pressure stream that results in trauma to surrounding tissues and elicits a pain response. Table 2 presents these variables related to different tissue types, adaptive capacity (flexibility) of those tissues, suggested injection rates, and the relative pressures generated within tissue spaces. All can impact tissue trauma during injections, as well as pain levels.16,33,34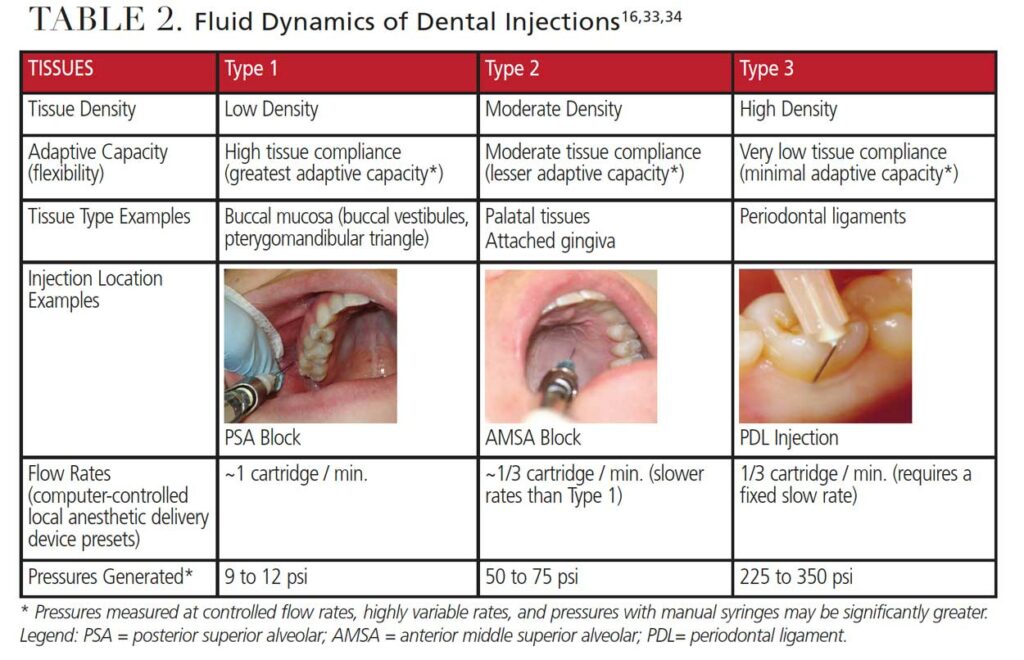
Kudo33 demonstrated patients perceive pain related to initial injection rates and intermittent fluid bursts generated when injections are initiated. The pain perception from bursts increases when small bore (higher gauge) needles are used.33
There is a significant relationship between the rate of injection and the rise in interstitial pressures when depositing anesthetic solution, which can affect its retention within the region. Gow-Gates and colleagues,31 along with Rood,32 discussed the implications of the injection hydraulics of rapid injection versus customarily recommended rates. Specifically, Gow-Gates and colleagues31 wrote that, “It is claimed that depositing 2 ml of an anesthetic solution at the lingula in 18.3 sec increases hydrostatic pressure from 14.5 to 469 mm Hg. … The resulting loss of the anesthetic through the pores of the capillary walls affects its concentration at the nerve membrane and may result in a partial or complete failure of the block.”
This significant imbalance between intra- and extra-vascular pressures increases the potential for uptake of anesthetic through capillary walls. Anesthetic solution intended for local inhibition of painful impulses instead may be forced into the systemic circulation, where it can quickly reach the more sensitive neurons of the central nervous system. This is possible, regardless of the LA drug selected. In addition, epinephrine will be forced into the circulation along with any LA drug that enters it, as opposed to constricting local vessels.
In addition to affecting success and safety, too much pressure can also increase pain, as demonstrated by Kudo,33 compromising safety when LA is forced into the circulation versus remaining in the tissues where the drug is deposited.29,30,32,33
SUMMARY
Decades of global research provide an extensive look at articaine, as well as paresthesia as an outcome of neurotoxicity and injection trauma. Moving forward, consideration of injection factors that may increase the potential for trauma continue to warrant attention.
Research focused on the pharmacology of articaine has confirmed what many clinicians already know: Articaine is an excellent drug with distinct benefits over other LA drugs. As the only hybrid local anesthetic available in dentistry, articaine is here to stay.
Bonus Web Content
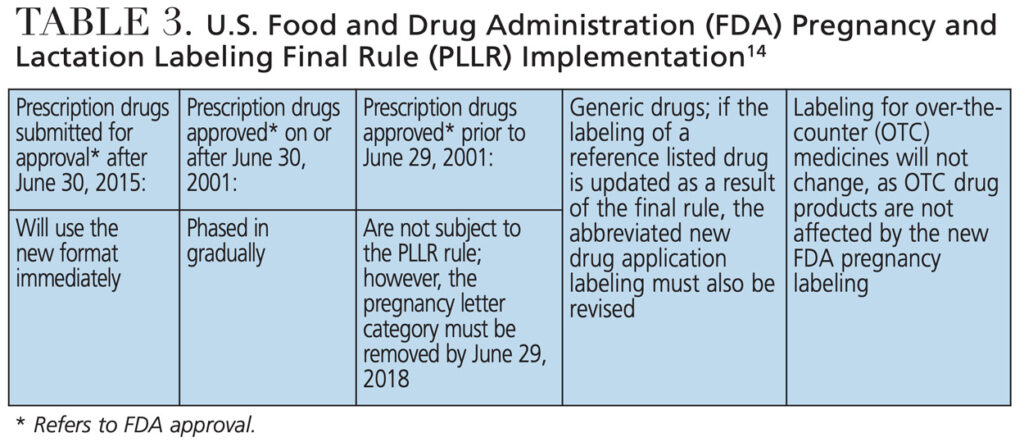
Update on Drug Labeling for Pregnancy and Lactation14
In 2015, the U.S. Food and Drug Administration implemented a change to the long-standing system for rating drug risks during pregnancy and lactation. In an effort to provide product information that is more meaningful to both patients and healthcare providers, the widely recognized system of letterbased risk categories (A, B, C, D and X) on prescription and biological drug labels was replaced with a new information format. This change was made to address concerns that the old letter system resulted in assumptions about the actual meaning of the letters that created confusion among patients and providers.
The new labeling system is designed to facilitate better patient-centered counseling and informed decision-making for pregnant women considering drugs recommended for their healthcare needs. Although the new labeling format guides improved individualized care decisions, it still does not provide a definitive “yes” or “no” answer in most cases. Clinical interpretation is still required on a case-by-case basis.
While the new Pregnancy and Lactation Labeling Final Rule (PLLR) went into effect on June 30, 2015, the implementation time-line of changes in product directions for use, including drug labels and package inserts, extends well beyond that date (Table 3). The A, B, C, D and X risk categories, in use since 1979, are now replaced with narrative presentations of information related to use of a drug during pregnancy and lactation, including: Risk Summary, Clinical Considerations for Use, and Supporting Data. A lactation subsection provides information about using the drug while breastfeeding, including the amount of drug in breast milk and potential effects on the breastfed infant, and a subsection on female and male reproductive potential with information about the need for pregnancy testing, contraception and infertility.
REFERENCES
- Malamed SF. Articaine 30 Years Later. Available at: https://www.oralhealthgroup.com/features/. Accessed February 11, 2022.
- Articaine — An Overview. Available at: https://www.sciencedirect.com/topics/medicine-and-dentistry/articaine/pdf. Accessed Accessed February 11, 2022.
- Malamed SF. Articaine, 20 Years Later. Available at: https://www.dentallearning.net/articaine-20-years-later. Accessed February 11, 2022.
- Martin E, Nimmo A, Lee A, Jennings E. Articaine in dentistry: an overview of the evidence and meta-analysis of the latest randomised controlled trials on articaine safety and efficacy compared to lidocaine for routine dental treatment. [Published correction appears in BDJ Open. 2021;11;7(1):29.] BDJ Open. 2021;7:27.
- Septodont. Articaine hydrochloride and epinephrine injection. Instructions for Use. Available at: www.septodontusa.com. Accessed February 11, 2022.
- Dentsply Pharmaceutical. Articadent — articaine hydrochloride and epinephrine bitartrate injection, solution. Instructions for Use. Available at: https://www.drugs.com/pro/articadent.html. Accessed February 11, 2022.
- Isen DA. Articaine: pharmacology and clinical use of a recently approved local anesthetic. Dent Today. 2000;19:72–77.
- Paxton K, Thome DE. Efficacy of articaine formulations: quantitative reviews. Dent Clin North Am. 2010;54:643–653.
- Katyal V. The efficacy and safety of articaine versus lignocaine in dental treatments: a meta-analysis. J Dent. 2010;38:307–317.
- Rathi NV, Khati AA, Agrawal AG, Baliga M, Thosar NR, Deolia SG. Anesthetic efficacy of buccal infiltration articaine versus lidocaine for extraction of primary molar teeth. Anes Prog. 2019;66:3–7.
- Oertel R, Ulrike E, Rainer R, Wilhelm K. The effects of age on the pharmacokinetics of the local anesthetic drug articaine. Reg Anesth Pain Med. 1999;24:524–528.
- Scully C. Scully’s Medical Problems in Dentistry. 7th ed. London: Churchhill Livingstone; 2014:51–96.
- Jastak JT, Yagiela JA, Donaldson D. Local Anesthesia of the Oral Cavity. Philadelphia: WB Saunders; 1995.
- U.S. Food and Drug Administration. Pregnancy and Lactation Labeling (Drugs) Final Rule. Available at: https://www.fda.gov/drugs/labeling-information-drug-products/pregnancy-and-lactation-labeling-drugs-final-rule. Accessed February 11, 2022.
- Malamed SF. Idaho State Dental Association. Presentation at the North Idaho Conference. Coeur d’Alene, Idaho. March 6, 2020.
- Bassett K, DiMarco A, Naughton D. Local Anesthesia for Dental Professionals. 2nd ed. Upper Saddle River, N.J.: Pearson; 2015.
- Malamed SF. Handbook of Local Anesthesia. 7th ed. St. Louis, Mo.: Mosby; 2020.
- Hawkins M, Bassett K, Royer R. Dental Local Anesthesia: Techniques and Key Features. Available at: https://www.dentallearning.net/ATF-ce. Accessed February 11, 2022.
- Al-Mahalawy H, Abuohashish H, Chathoth S, Al-Masoud N, Al-Jandan B. Articaine versus lidocaine concentration in palatal tissues after supraperiosteal buccal infiltration anesthesia. J Oral Maxillofac Surg. 2018;76:315.e1–315.e7.
- Ashwath B, Subramoniam S, Vijayalaskshmi R, Shanmugam M, Priya BM, Anitha V. Anesthetic efficacy of 4% articaine and 2% lignocaine in achieving palatal anesthesia following a single buccal infiltration during periodontal therapy: A randomized double-blind split-mouth study. J Anaesthesiol Clin Pharmacol. 2018;34:107–110.
- de Geus JL, Noguiera da Costa JK, Maran BM, Loguercio AD, Reis A. Different anesthetics on the efficacy of inferior alveolar nerve block in patients with irreversible pulpitis. J Am Dent Assoc. 2020;151:87–97.
- Nagendrababu V, et al. Is articaine more effective than lidocaine in patients with irreversible pulpitis? An umbrella review. Int Endod J. 2020;53:200–213.
- Wright GZ, Weinberger SJ, Friedman CS, Plotzke OB. The use of articaine local anesthesia in children under 4 years of age — a retrospective report. Anesth Prog. 1989;36:268–271.
- Stenver DI. Case number: 3200-1367. Adverse effects from anaesthetics used in relation with dental care with a special focus on anaesthetics containing articaine. Pharmacovigilance Working Party of the European Union. October 20, 2006.
- Haling F, Neff A, Ziebart T. Local anesthetic usage among dentists: German and international data. Anesth Prog. 2021;68:19–25.
- Malamed SJ. Dental Pain Control and Local Anesthesia: A 40-year journey. Available at: https://www.dentistrytoday.com/dental-pain-control-and-local-anesthesia-a-40-year-journey/. Accessed February 11, 2022.
- Malet A, Faure M-O, Deletage N, Pereire B, Haas J, Lambert G. The comparative cytotoxic effects of different local anesthetics on a human neuroblastoma cell line. Anesth Analg. 2015;120:589–596.
- Moore PA, Haas DA. Paresthesias in dentistry. Dent Clin North Am. 2010;
54:715–730. - Haas DA, Lennon D. A 21-year retrospective study of reports of paresthesia following local anesthetic administration. J Can Dent Assoc. 1995;61:319–320, 323–326, 329–330.
- Toma M, Berghahn M, Loth S, Verrengia B, Visani L, Velotti F. Articaine and paresthesia in dental anaesthesia: neurotoxicity or procedural trauma? Available at: https://www.oralhealthgroup.com/features/articaine-and-paresthesia-in-dental-anaesthesia-neurotoxicity-or-procedural-trauma/. Accessed February 11, 2022.
- Kafalias MC, Gow-Gates GAE, Saliba GJ. The Gow-Gates technique for mandibular block anesthesia: A discussion and a mathematical analysis. Anesth Prog. 1987;34:142–149.
- Rood JR. The pressures created by inferior alveolar injections. Br Dent J. 1978;
144:280–282. - Kudo M. Initial injection pressure for dental local anesthesia: effects on pain and anxiety. Anesth Prog. 2005;52:95–101.
- Hochman MN, Friedman MJ, Williams W, Hochman CB. Interstitial tissue pressure associated with dental injections. Quintessence Int. 2006;37:469–476.
From Decisions in Dentistry. March 2022;8(3)25-29.




