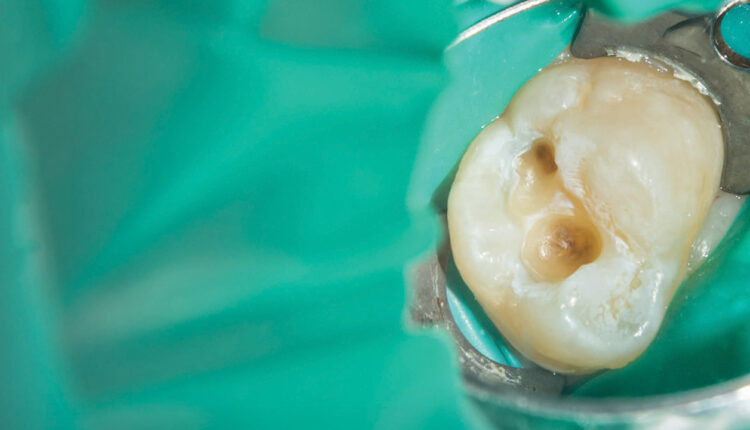
Clinical Treatment of Deep Caries
Adopting a minimally invasive approach in managing deep caries lesions will help avoid complications with pulpal involvement.

Although the standard of care taught in some dental schools and emphasized by many regional dental board examinations supports the removal of all carious tooth structure, selective caries removal will reduce issues with pulpal exposure.1 In addition, long-term evidence has demonstrated that bonded restorations placed over carious dentin can arrest the clinical progression of lesions.2 This article will provide evidence-based recommendations and examine key clinical considerations when treating deep caries lesions. Guidelines for deep caries treatment have been previously proposed by Alleman and Magne3 for biomimetic approaches to caries therapy, and Hernandez and Kolker4 for conservative approaches to treating deep caries.
Caries is defined as a disease that causes a shift in the dental biofilm to acid-producing microorganisms which, ultimately, leads to mineral loss from hard dental tissue.5 As the caries lesion progresses into dentin, the dentin tissue is first attacked by acid byproducts from microorganisms and eventually inhabited by the acid-producing microorganisms themselves.6 Therefore, a cross section of carious dentin has distinct histological and structural zones that represent how far along the caries process has progressed within the dentin (Figure 1).6
Clinically, the transitions between these histological zones may be difficult to detect, but they may be more easily described by the pioneering work of Kuboki et al7 and Fusayama and Terachima8 as the “outer carious dentin” (also called “infected dentin”), and the “inner carious dentin” (also called “affected dentin”). The two layers were differentiated by a staining dye and it was determined the infected dentin contained irreversible destruction of collagen cross-linking7 and was unable to undergo remineralization.8 It was later reported the infected dentin had 1300 times greater bacterial concentrations than the nonstained affected dentin.9
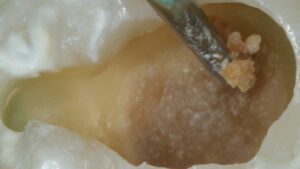
CARIOUS DENTIN DIFFERENTIATION
Many general dentists consider dentin hardness as the most important criteria for caries excavation.10 Infected dentin is soft due to the lack of mineral content or collagen network. Its consistency can be likened to cottage cheese (Figure 2). Affected dentin is firmer than infected dentin due to the presence of the collagen network and some mineral content. Its consistency may be referred to as leatherlike (Figure 3). Healthy dentin is often described as hard dentin or sound dentin.
Because the terms used to describe dentin hardness can be subjective, manipulation with dental instruments can help differentiate soft (infected), firm (affected), and hard (healthy) dentin.5 Soft dentin can be deformed by pressing an instrument into it, and it can easily be removed by gently scraping its surface with minimal force. Firm, leatherlike dentin does not deform when an instrument is placed into it, but it can be scraped off with medium pressure. Hard dentin requires a strong force to engage the dentin, and it cannot be removed without a sharp cutting edge or bur. A “scratchy” sound can be heard when dragging an explorer across hard dentin.

Aside from the use of tactile sensation, some clinicians rely on alternative methods to identify and remove soft dentin. A recent survey reported the majority of general dentists use a metal bur (69%) or hand excavator (66%) to remove deep caries, but some use ceramic (4%) and polymer (1%) burs.10 Polymer (or plastic) burs have a hardness that is lower than sound dentin, but harder than carious dentin. Although the reduced hardness of the bur and less aggressive blade design help prevent removal of sound dentin, polymer burs have been reported to leave remnant infected dentin.11 Made from zirconia, ceramic burs have been shown to remove caries as efficiently as metal burs.11 Similar to metal burs, however, they are harder than sound dentin, and their ability to discriminate between infected and affected dentin is unproven.11
Another method of identifying carious dentin is the use of caries detecting dyes. These dyes are typically composed of a propylene glycol solvent and either a red or green dye. The dyes have been shown to stain demineralized organic matrix, not the actual bacteria.12 Circumpulpal dentin and the dentin-enamel junction (DEJ) have also been shown to stain with these dyes (owing to a reduced level of mineralization).13 Therefore, injudicious use of caries detecting dyes will not truly differentiate affected and infected dentin, and may promote unnecessary removal of sound or affected dentin or even expose the pulp. Some evidence suggests that lightly stained dentin (termed “pink-haze” with the use of a red dye) represents affected dentin.14
EXCAVATION LEVEL
The decision of how much infected dentin to remove is a balance between removing enough carious dentin to obtain a favorable bond and effective seal for the restoration, while maintaining sufficient dentin over the pulp to ensure pulpal health. The long-taught dogma of complete caries removal until hard dentin is achieved — even at the expense of pulpal exposure — is being challenged with more conservative, selective caries-removal protocols.5 Selective caries removal has a 77% lower incidence of exposing the pulp than complete caries removal; however, there is insufficient evidence to determine if there is a decrease in signs and symptoms of pulpal disease with this approach.1 The argument for leaving carious dentin over the pulp is to avoid introducing caries-infected dentin into the pulp during excavation,15 and preserve a dentin thickness of at least 0.25 mm to prevent injury to the underlying odontoblasts.16 A 10-year study demonstrated that bonded restorations placed over carious dentin can arrest their progression.2
Prior to initiating a procedure in which a deep lesion is to be removed, a pulpal diagnosis should be obtained. Restorative treatment may be initiated with a normal pulp (immediate dissipation of cold sensation when stimulus is removed) or reversible pulpitis (sharp, quick pain that ends when cold stimulus is removed). Teeth with irreversible pulpitis (lingering pain to hot or cold beyond five seconds, or spontaneous pain) or pulpal necrosis (no response to vitality testing) require endodontic treatment.17
ONE- OR TWO-STEP PROCESS
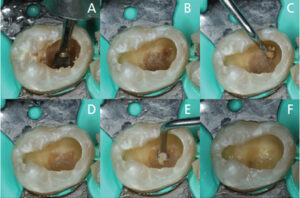
Deep caries may be selectively removed in either a one- or two-step process. In a one-step process, the lesion on the pulpal floor is removed until firm, leatherlike dentin is encountered. This firm, affected dentin may be remineralized18 and serves to protect pulpal health. The exception to this stopping point is if the lesion extends radiographically beyond the inner third of the dentin. For very deep caries, soft, infected dentin may be left over the pulpal floor, as a permanent restoration will seal remnant bacteria from any source of nutrition, causing bacterial death or dormancy (Figures 4A through 4F).2,19
In a two-step process, the lesion is removed until soft dentin is achieved, after which a provisional restoration is placed. At six weeks to a year after placement of the provisional restoration, the patient’s pulp vitality and caries risk are reassessed, the caries is reexcavated to achieve firm dentin, and the tooth is permanently restored.4 Some evidence suggests the one-step caries removal technique may be more effective than the two-step technique.1,19,20 The one-step technique has resulted in 77% fewer pulpal exposures than complete caries removal, whereas the two-step technique has led to 55% fewer exposures.1
Although leaving affected or infected dentin over the pulpal floor can be advantageous, achieving a periphery of sound, hard dentin and enamel is necessary to achieve optimal bonding and an effective restorative seal. Bond strength to affected dentin is 33% lower than when bonding to hard dentin, and the bond to infected dentin is 78% lower.21 Bonding to caries-infected dentin is ineffective due to its low cohesive strength from mineral loss and collagen degradation. By the same token, bonding to caries-affected dentin is inferior to bonding to hard dentin due to deposition of mineral content within its dentinal tubules, which cause an altered etch pattern and decreased resin penetration.11 Therefore, the peripheral area of the preparation should be composed entirely of hard dentin. Some authors have recommended a width of 1 to 2 mm of hard dentin.3
One area of carious tissue removal that may pose a clinical challenge is at the DEJ. Once a lesion is cavitated and the dentin is colonized with bacteria, the lesion can spread along the DEJ. At the periphery of the lesion, sound enamel may actually help shelter bacteria, creating sound enamel undermined by carious dentin.11 When achieving a peripheral zone of hard dentin, there may be parts of the margin in which normal enamel is lined by carious dentin. The clinician must choose between removing normal enamel with carious dentin, or removing only the dentin at the DEJ — leaving some unsupported enamel (Figure 5, Figure 6 and Figure 7). In their study, Kidd et al22 concluded that caries tissue at the DEJ should be removed if it is soft, but not necessarily stain-free.
INDICATIONS FOR A LINER
If pulpal exposure has occurred via iatrogenic damage or trauma, a direct pulp cap may be performed with a calcium hydroxide or calcium silicate liner. There is some evidence to suggest calcium silicate liners, such as mineral trioxide aggregate, perform better than calcium hydroxide liners.23 As noted previously, caries lesions approaching the pulp should not be exposed in order to prevent bacterial contamination of the pulp. When carious dentin is left near the pulp, some clinical guidelines suggest a liner should be placed using an indirect pulp cap technique,4 while others recommend the direct application of an adhesively bonded restoration.3 If a calcium hydroxide or calcium silicate liner is placed, its purpose is to stimulate the release of growth factors and, consequently, the formation of reparative dentin to protect the pulp from thermal, electrical or chemical stimuli.24 A recent systematic review reported the use of calcium hydroxide liner had no positive effect on the clinical outcome in the treatment of deep caries.24

SUMMARY
In conclusion, embracing a minimally invasive approach in managing deep caries will help avoid complications with pulpal involvement. Current evidence suggests the endpoint for caries removal should be leathery, firm affected dentin on the pulpal floor, with a periphery of sound, hard dentin and enamel. If a deep lesion extends to the inner third of the dentin, a minimal amount of soft, infected dentin may be left on the pulpal floor to prevent pulpal exposure. While a liner may be placed over the deep pulpal dentin, it may not be necessary. Finally, a bonded restoration should be carefully placed to create an effective seal that will prevent further lesion progression.
KEY TAKEAWAYS
- Prior to initiating a procedure in which a deep lesion is to be removed, a pulpal diagnosis should be obtained.
- Selective caries removal will reduce issues with pulpal exposure.1
- Long-term evidence has demonstrated that bonded restorations placed over carious dentin can arrest lesion progression.2
- A cross section of carious dentin has distinct histological and structural zones that represent how far along the caries process has progressed within the dentin.
- Infected dentin is soft due to the lack of mineral content or collagen network. Its consistency can be likened to cottage cheese.
- Affected dentin is firmer than infected dentin due to the presence of the collagen network and some mineral content. Its consistency may be referred to as leatherlike.
- Healthy dentin is often described as hard dentin or sound dentin.
- Another method of identifying carious dentin is the use of caries detecting dyes, but injudicious use of dyes will not truly differentiate affected and infected dentin, and may promote unnecessary removal of dentin, or even expose the pulp.
- The decision of how much infected dentin to remove is a balance between removing enough carious dentin to obtain a favorable bond and effective seal for the restoration, while maintaining sufficient dentin over the pulp to ensure pulpal health.
REFERENCES
- Ricketts D, Lamont T, Innes NP, Kidd E, Clarkson JE. Operative caries management in adults and children. Cochrane Database Syst Rev. 2013;3:CD003808.
- Mertz-Fairhurst EJ, Curtis JW Jr, Ergle JW, Rueggeberg FA, Adair SM. Ultraconservative and cariostatic sealed restorations: results at year 10. J Am Dent Assoc. 1998;129:55–66.
- Alleman DS, Magne P. A systematic approach to deep caries removal end points: the peripheral seal concept in adhesive dentistry. Quintessence Int. 2012;43:197–208.
- Hernandez M, Kolker JL. Modern Conservative Approach to Treat Deep Caries Lesions. Available at: https:/ / www.oralhealthgroup.com/ features/ modern-conservative-approach-to-treat-deep-caries-lesions/ . Accessed January 7, 2019.
- Innes NP, Frencken JE, Bjørndal L, et al. Managing carious lesions: Consensus recommendations on terminology. Adv Dent Res. 2016;28:49–57.
- Frank RM. Structural events in the caries process in enamel, cementum, and dentin. J Dent Res. 1990;69(Spec No):559–566,discussion 634–636.
- Kuboki Y, Ohgushi K, Fusayama T. Collagen biochemistry of the two layers of carious dentin. J Dent Res. 1977;56:1233–1237.
- Fusayama T, Terachima S. Differentiation of two layers of carious dentin by staining. J Dent Res. 1972;51:866.
- Anderson MH, Loesche WJ, Charbeneau GT. Bacteriologic study of a basic fuchsin caries-disclosing dye. J Prosthet Dent. 1985;54:51–55.
- Koopaeei MM, Inglehart MR, McDonald N, Fontana M. General dentists’, pediatric dentists’, and endodontists’ diagnostic assessment and treatment strategies for deep carious lesions: a comparative analysis. J Am Dent Assoc. 2017;148:64–74.
- de Almeida Neves A, Coutinho E, Cardoso MV, Lambrechts P, Van Meerbeek B. Current concepts and techniques for caries excavation and adhesion to residual dentin. J Adhes Dent. 2011;13:7–22.
- Boston, DW, Graver HT. Histological study of an acid red caries‐disclosing dye. Oper Dent. 1989;14:186.
- Yip HK, Stevenson AG, Beeley JA. The specificity of caries detector dyes in cavity preparation. Brit Dent J. 1994;176:417–421.
- Iwami Y, Shimizu A, Narimatsu M, Kinomoto Y, Ebisu S. The relationship between the color of carious dentin stained with a caries detector dye and bacterial infection. Oper Dent. 2005;30:83–89.
- Bjørndal L, Reit C, Bruun G, et al. Treatment of deep caries lesions in adults: randomized clinical trials comparing stepwise vs. direct complete excavation, and direct pulp capping vs. partial pulpotomy. Eur J Oral Sci. 2010;118:290–297.
- Murray PE, About I, Lumley PJ, Franquin JC, Remusat M, Smith AJ. Cavity remaining dentin thickness and pulpal activity. Am J Dent. 2002;15:41–46.
- Bahcall J, Ashrafi S, Xie Q. The importance of obtaining a pulpal and periradicular diagnosis prior to restorative treatment. Compend Contin Educ Dent. 2019;40:27–31
- Miyauchi H, Iwaku M, Fusayama T. Physiological recalcification of carious dentin. Bull Tokyo Med Dent Univ. 1978;25:169–179.
- Thompson V, Craig RG, Curro FA, Green WS, Ship JA. Treatment of deep carious lesions by complete excavation or partial removal: a critical review. J Am Dent Assoc. 2008;139:705–712.
- Schwendicke F, Meyer-Lückel H, Dörfer C, Paris S. Failure of incompletely excavated teeth: a systematic review. J Dent. 2013;41:569–580.
- Yoshiyama M, Tay F, Torri Y, et al. Resin adhesion to carious dentin. Am J Dent. 2003;16:47–52.
- Kidd EA, Ricketts DN, Beighton D. Criteria for caries removal at the enamel-dentine junction: a clinical and microbiological study. Br Dent J. 1996;180:287–291.
- Hilton TJ, Ferracane JL, Mancl L; Northwest Practice-based Research Collaborative in Evidence-based Dentistry (NWP). Comparison of CaOH with MTA for direct pulp capping: a PBRN randomized clinical trial. J Dent Res. 2013;92(Suppl 7):16S–22S.
- da Rosa WL, Lima VP, Moraes RR, Piva E, da Silva AF. Is a calcium hydroxide liner necessary in the treatment of deep caries lesions? A systematic review and meta-analysis. Int Endod J. 2018: Epub ahead of print.
Nathaniel Lawson, DMD, PhD, discloses he has received grants, research support or honoraria from 3M, Ivoclar, Dentsply Sirona, DMG America, GC America, Kuraray, and Pulpdent. Augusto Robles, DDS, MS, has no commercial conflicts of interest to disclose.
From Decisions in Dentistry. February 2019;5(2):10—12,14.





Bacteria in the mouth use these remains as a source of their food and produce acids that have the potential to dissolve enamel and cause cavities. These cavities now become areas where food keeps getting lodged very often.