CE Sponsored by Colgate P4 Medicine’s Paradigm for Periodontal Health
The concept of P4 medicine — participation, prediction, prevention and personalization — offers new tools to manage the various stages of periodontal disease.
This course was published in the June 2019 issue and expires June 2022. The author has no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
The concept of P4 medicine — participation, prediction, prevention and personalization — offers new tools to manage the various stages of periodontal disease
INTRODUCTION
As part of the evolution toward individually tailored health care, the P4 concept considers the complex biological interactions underlying health and disease states; it is comprehensive, holistic, quantifies wellness and demystifies disease — all with the goal of supporting individual health. Under P4 medicine, patients and health care providers work as a team to promote health throughout the lifespan. This requires practitioners — including dental teams — to embrace a multilevel interdisciplinary approach and harness emerging technologies to ensure optimal care.
Based on P4 principles, the new periodontal classification system emerging from the 2017 World Workshop on the Classification of Periodontal and Peri-implant Diseases and Conditions was supported by an unrestricted educational grant from Colgate-Palmolive. We are equally delighted to sponsor this article, which brings to life the P4 concept adapted for periodontal therapy. Emphasizing the advantages of using this model for chronic disease management, the author notes P4 periodontics involves empowering patients and reinforcing oral health literacy; important points considering the success of periodontal therapy relies on patients adopting and maintaining an effective daily oral care routine.
Ultimately, using the P4 attributes of prediction, prevention, personalization and participation can help spark thoughtful discussions between patients and providers that will support Whole Mouth Health. I hope you will find this seminal article a valuable resource for improving patient care.
—Matilde Hernández, DDS, MS, MBA
Scientific Affairs Manager Professional Oral Care
Colgate Oral Pharmaceuticals
Adapting the tenets of P4 medicine to oral health care provides practitioners with exciting new ways to manage patients through all stages of periodontal disease
The concept of P4 medicine emerged in the early 2000s as the proposed “future of medicine.”1,2 It dictates that management of chronic diseases should not be “reactive,” but should incorporate the principles of the 4Ps: participation, prediction, prevention and personalization. P4 medicine is a holistic and integrative approach to health and disease management, empowering clinicians and patients to actively participate in care. It incorporates the concepts of personalized (or precision) medicine and takes into account lifespan disease experience, new diagnostic and risk assessment tools, patient participation in care, and utilization of technology to advance all aspects of care.

In this paper, periodontology and the new periodontal classification of disease are used as examples of how P4 medicine — or, in this case, P4 periodontics — can be incorporated into the management of chronic oral health conditions. Due to space constraints, this discussion will focus on management of individuals through various stages of periodontal disease. However, it is recognized and accepted that P4 medicine and P4 periodontics embrace all aspects of health care, ranging from global populations and communities to microlevels of individual body systems.
Recently, a landmark publication detailed how oral health is a continuing public health burden, with approximately 50% of the world’s population experiencing tooth decay, periodontal disease and tooth loss.3 Despite improvements in our understanding of oral diseases and how they might be prevented, this burden has remained static over the last 25 years. While the FDI World Dental Federation has a stated goal to reduce the burden of oral disease by 2020,4 it seems this may be an elusive target. Therefore, we need to rethink our preventive and treatment strategies for the management of chronic oral conditions. In light of the complexities of chronic oral conditions, this commentary will focus on plaque-associated periodontal disease as an example of how changes in our thinking for management is shaping future strategies and treatment paradigms.
STAGES OF PERIODONTAL HEALTH AND DISEASE
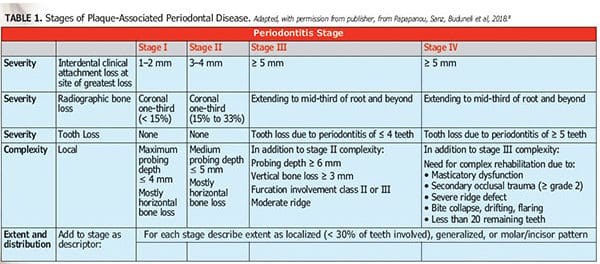
The new classification system arising from the World Workshop on Classification of Periodontal and Peri-Implant Diseases recognizes that plaque-associated periodontal disease represents a spectrum of conditions ranging from periodontal health to advanced, dentition-threatening disease.5 For the first time, periodontal health has been clearly defined.6,7 With an accepted definition of periodontal health, it is now possible to define plaque-associated periodontal disease through a sequential series of stages (Table 1) and grades of severity.
With a focus on well-being and disease prevention, periodontal health is an achievable goal that can be maintained throughout life. The focus is on whole-body wellness, as well as the prevention of chronic disease and its associated risk and modifying factors. Contemporary periodontal care should seek to identify how the disease transitions through life, and how to reverse its course at the earliest identifiable stage. Within the P4 medicine concept, maintaining (or returning individuals to) health will no longer be solely up to individual clinicians, but will involve interactions with all health professionals through a “multisector” approach, as well as embracing and harnessing all available technological advances.
The new classification of periodontal disease is somewhat based on the principles of P4 medicine, inasmuch as plaque-associated periodontal disease can be classified into stages and grades according severity, rate of progression, and modifying risk factors.8 The trajectory relates to the rate at which the disease has developed and age of the patient; it can be graded as slow, moderate or rapid. This fits into the P4 model, as it takes into account the predictive, preventive and personalized aspects of periodontal disease. The only aspect lacking is the participatory component of P4 medicine. However, as will become evident later, this important aspect should also be factored into P4 periodontics because an assessment of a patient’s ability to participate in ownership of his or her periodontal condition will significantly influence disease prevention and management.
An important concept in P4 medicine is that the trajectory, or development, of chronic disease can be related to allostasis (a state of equilibrium) and subsequent loss of homeostasis as a result of increasing allostatic load. Allostasis is an active process whereby the body adapts to internal and external stressors. These processes are evident during the development of gingivitis. For example, periodontal health may be maintained even in the presence of the external stressor of dental plaque. However, if the allostatic load increases, balance between host and microbial challenge may be altered, resulting in development of gingivitis and, if further dysregulated, periodontitis.
These concepts form the basis of a P4 medicine continuum for periodontal disease, whereby treatment options can be either proactive or reactive, and these directly relate to patient- or clinician-mediated management strategies. Within this model, allostatic load is influenced by external stressors, some of which patients can control, while others can only be controlled by treatment (Figure 1). Thus, the stages of periodontitis can be defined, in part, on the basis of allostatic load and individual responses — and these can be elegantly incorporated into the concepts of P4 medicine.
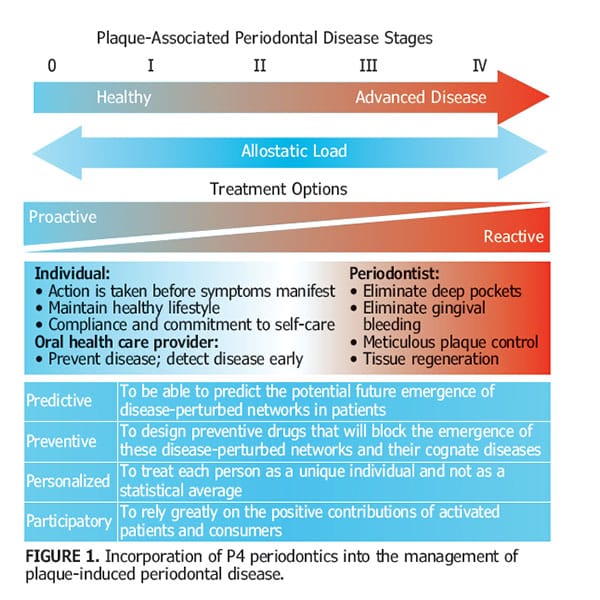 Stage 0. Periodontal Health — allostasis or general homeostasis: Periodontal health is a state free of inflammation (gingivitis and periodontitis) that permits an individual to function normally and not suffer adverse consequences (mental or physical) as a result of past periodontal damage.7 Accordingly, four levels of periodontal health have been proposed, depending on the state of the periodontium (structurally and clinically sound or reduced) and the relative treatment outcomes:
Stage 0. Periodontal Health — allostasis or general homeostasis: Periodontal health is a state free of inflammation (gingivitis and periodontitis) that permits an individual to function normally and not suffer adverse consequences (mental or physical) as a result of past periodontal damage.7 Accordingly, four levels of periodontal health have been proposed, depending on the state of the periodontium (structurally and clinically sound or reduced) and the relative treatment outcomes:
- Pristine periodontal health in a structurally sound and uninflamed periodontium
- Well maintained clinical periodontal health on a structurally and clinically sound (intact) periodontium
- Periodontal disease stability on a reduced periodontium
- Periodontal disease remission/control on a reduced periodontium8
Thus, two forms of health can be recognized: one is a state that has never progressed to disease, and the other is a reversal from a disease state to one of health. In the latter, periodontal health is attained by controlling inflammation and infection, reducing predisposing factors, and controlling any modifying factors. These processes result in either periodontal disease stability on a reduced periodontium, or periodontal disease in a state of remission or control on a reduced periodontium. In periodontal health, risk factors and allostatic load remain constant threats that must be continually monitored.
Stage I. Initial (early/mild) Periodontitis (allostatic load commences): Detectable signs and symptoms of gingivitis are apparent, along with early signs of destruction of the periodontal tissues, such as early bone loss. Most likely, the patient is unaware of these changes and not experiencing any difficulties. This stage represents the initiation of clinical signs and symptoms associated with increased risk for further disease development.
Stage II. Moderate Periodontitis (initial exposure to changing allostatic load): Periodontal destruction has progressed and clinical attachment loss up to 3 to 4 mm may be noted, along with bone loss up to the coronal one-third of root surfaces. Probing depths may be as great as 5 mm, and the nature of bone loss appears to be generally horizontal. Variable rates of progression in Stage II can be noted, and these are largely regulated by other stressors (predisposing and modifying factors known to modify the clinical manifestation of periodontitis). Unless detected clinically, treatment may not always be available. If treatment is provided, it is usually reactive to the symptoms.
Stage III. Severe Periodontitis with potential for additional tooth loss (increasing allostatic load): Periodontal destruction has now progressed and clinical attachment loss greater than 5 mm may be noted, along with bone loss extending to the middle-third of root surfaces. Furcation involvement of grade I and II may be seen, and tooth loss attributable to periodontitis is limited to four teeth or less. By this stage, reactive care is generally undertaken. It is important to note that such reactive therapy involves treating the symptoms of chronic periodontitis and sometimes disregards addressing the allostatic conditions (e.g., unhealthy lifestyle, external stressors, or predisposing and modifying factors) that most likely led to the condition’s development.
Stage IV. Severe Periodontitis with potential for loss of the dentition (complete allostatic overload): Periodontal destruction has progressed and clinical attachment loss greater than 5 mm may be noted, along with bone loss extending beyond the middle-third of root surfaces. Furcation involvement of grade I and II may be seen, and tooth loss attributable to periodontitis involves five or more teeth. Additional functional disturbances are noted, including loss of masticatory function, secondary occlusal trauma and bite collapse. Progression to this stage is potentially a terminal phase of disease expression. While traditional reactive treatment can be provided, additional therapy and complex rehabilitative procedures will be needed to restore reasonable function.
While it is difficult to undergo a spontaneous reversal in all but the initial stages of periodontal disease, it is possible that — with appropriate lifestyle changes and advanced periodontal regenerative therapy — a patient could revert from stages II and III back to periodontal health, albeit on a reduced periodontium, or in a state of disease remission transition. Thus, with application of the P4 principles at all stages of the disease (possibly except Stage IV), it is possible to return to periodontal health.
PRINCIPLES OF P4 PERIODONTICS
If we accept that our current reactive periodontal therapy model is of limited value, new paradigms should be explored that enable management of the various stages of periodontal disease and associated risk factors. In this context, the P4 model has been proposed as a novel approach to contemporary management of chronic diseases.1,2 Management of plaque-associated periodontal disease should be no exception, and thus the term P4 periodontics has been coined.
In order to deliver P4 periodontics, four factors that are pathognomic of plaque-associated periodontitis must be taken into account. These include understanding the severity, activity, control and responsiveness of the disease. While we currently do not have all the tools needed to accurately define these features, the future holds considerable promise for technologies that will assist in monitoring periodontal disease.
Severity: At present, severity of periodontal disease is determined using some very “blunt instruments.” These include visual assessment of bleeding on probing, crude measurement of pocket depth and loss of attachment, tooth mobility and furcation involvement, and, generally, two-dimensional radiography. Clearly, there is need for more sophisticated tools to determine disease severity, and these could include judicious use of three-dimensional radiography, microbiome assessments, and proteomic analysis of gingival punch biopsies, as well as salivary and gingival crevicular fluid analyses for biomarkers of disease. Identifying disease severity aids appropriate treatment decisions to control the disease’s effect on periodontal tissues.
Activity: The determination of periodontal disease activity is a crucial factor in understanding how biological processes drive disease progression that ultimately leads to tissue destruction. The aim of therapy is to stop or reduce progression, with the goal of preventing the disease from progressing to severe periodontitis. Unfortunately, one of the great shortcomings in periodontal disease diagnostics is the inability to determine disease-active from disease-inactive sites due to the lack of well-validated disease-activity markers.
Control: The aim of controlling plaque-associated periodontal disease is to manage it as an opportunistic infection that is modified by the host inflammatory response. Thus, it is important to control inflammation and infection. In addition to controlling the disease, it is necessary to identify and reduce (or manage) both predisposing and modifying factors. Once the disease has been stabilized, further control is managed through continuous assessment and supportive periodontal care. Due to the lack of good biomarkers for disease activity or severity, objective markers for disease control are elusive and clinicians must rely on current subjective assessments, such as reduction in bleeding on probing, plaque control, reduced pocket probing depths, and gain of clinical attachment levels.
Responsiveness: The literature notes that among patients with periodontitis, up to 20% (or more) prove unresponsive to conventional periodontal therapy.9–11 On the other hand, some 80% do respond to treatment — albeit to varying degrees and rates. Understanding individual responsiveness to periodontal treatment will become critical in the context of personalized treatment plans based on an individual’s microbiome, inflammasome and allostatic load.
INTEGRATION INTO PERIODONTAL CARE
P4 medicine allows an integrated patient-centered and systems-based model for management of chronic disease. Because plaque-induced periodontitis is a classic chronic disease, it is appropriate to consider how P4 medicine may be adapted for periodontal therapy. By considering the life course of periodontitis, each of the 4Ps can be modeled into its management (Table 2). For example, two “life courses” of periodontitis experience can be described: One is slowly progressing, and the other is more severe, undergoing a period of significant activity that is associated with tissue destruction.12 Regardless of the course, it is possible to identify where identification, prevention, personalization and individual participation in disease control fit into the lifespan trajectory of periodontitis.
Predictive Periodontics: If precursors of the emergence of periodontal disease (Stage I) can be identified, interventions that control biologic pathways leading to disease can be proactive and, presumably, prevent tissue destruction. Thus, in terms of prevention, prediction of disease is a crucial element that should focus on risk factors and risk factor phenotype. In the P4 model, this is not only restricted to well-known risk factors (such as smoking and uncontrolled diabetes), it will also require assessing the allostatic load, genetic inflammation phenotype (inflammasome), and microbiome to identify risk of disease development. This will involve an array of health professionals who will interact through data management systems to feed information into a system-based protocol. In turn, this will allow identification of healthy individuals without any signs or symptoms of periodontitis, but with risk potential for disease development.
Preventive Periodontics: Periodontology has always recognized the importance of prevention, and this has focused largely on emphasizing daily plaque control. However, the question arises as to how effective this approach has been. For example, it has been noted that despite the dramatic increase in use of oral hygiene aids, clinical efforts to provide oral hygiene instruction, and the associated general improvement in oral hygiene levels in the community, the incidence of severe periodontitis remains largely unaffected.13 Prevention of plaque-associated periodontal disease will therefore require a more comprehensive understanding of disease etiopathogenesis. While significant inroads have been made into this field, it is still in its infancy with regard to establishing biological system networks, defining how risk factors influence these networks, and identifying protective factors that prevent disease establishment or progression. Thus, by preventing progression at stages 0 and I, it may be possible to dampen the effects of known risk factors and, hence, periodontitis may not develop or could even be reversed. A simple model for managing inflammation to control infection and thus drive gingivitis back to health has been proposed by Bartold and Van Dyke.14,15
Personalized Periodontics: It could be argued that clinical periodontics already practices personalized periodontics. We recognize that individuals present with different sets of clinical signs, symptoms, risk factors and modifying factors that require individualized treatment. However, generic treatments remain the mainstay of traditional therapy; these include a philosophy that aims to eliminate deep pockets and gingival bleeding, while achieving meticulous plaque control. In practical terms, this is rarely achieved. Indeed, the focus on anti-infective therapy alone has been questioned because it fails to effectively manage up to 20% (or more) of patients with periodontitis.9 This is because generic periodontal therapy makes a number of assumptions that tend to manage the “average” patient. As a result, this one-size-fits-all approach can prove quite successful for some individuals — but not all. Clearly, a missing link in current management strategies is our inability to fully incorporate the complexities of human biology, host responses to the environment, and genetic control of these responses into patient stratification.
Patient stratification is the process whereby a disease (such as periodontitis) is understood to be a complex condition arising from multiple pathways — and as a result of multiple risk factors — that can act in isolation or in an additive manner. This forms the basis of personalized periodontal therapy that focuses on a number of questions, including:
- What are an individual’s major risk factors for periodontitis?
- How can we identify individual risk for developing periodontitis?
- How can we identify individual risk for progression of periodontitis?
- How do we determine individual treatment response and outcomes?
A crucial element in personalized periodontics will be to identify the so-called “nonresponder” to generic treatments. It is important to identify these patients and target individual phenotypes/genotypes to better control their periodontal disease. To accomplish this, periodontology will need to develop sophisticated systems biology communication technology to access individual genome, microbiome and inflammasome data. Furthermore, this information will need to be managed so that patients’ complex, diverse and collated data can be incorporated across the P4 periodontics platform.
Participatory Periodontics: An often overlooked, but important, aspect of P4 periodontics is how to empower patients with their own data so they take a higher level of ownership of their periodontal and systemic health. A common question from patients with newly diagnosed periodontitis is, “What can I do to improve this situation?” A standard recommendation has been to improve oral hygiene and perhaps address lifestyle issues that may be negatively impacting the condition. Although patients may wish to participate in their periodontal care, clinicians find this an ongoing challenge. There is no doubt that for successful and long-term management of periodontal disease, the patient must be an integral part of treatment strategies that include a predictive and preventive approach. In the future, participatory periodontics will require new data collection processes involving self-tracking that can be utilized to assist individuals in working collaboratively with oral health professionals to maintain (or attain) periodontal health.
Consistent with P4 medicine, participatory periodontics requires greater incorporation of health literacy into patient care. This is the degree to which individuals have the capacity to obtain, process and understand basic health information and services so they can make appropriate health care decisions.16
In today’s era, there is an app for everything. Technological advances, such as Fitbits, sleep trackers, posture monitors and blood glucose monitoring (to name a few), allow people to monitor almost everything in relation to their health and health care. According to a 2014 survey, 31% of U.S. consumers use a quantified self tool to track their health and fitness, food, diet, sleep and/or mood.17 Oral health is not being left behind, with numerous dental apps and websites available for use.18 This is all about gathering information for both patients and practitioners. With patients more involved than ever in their health care, this presents opportunities to develop new levels of patient participation in care. In the P4 model, such participation will allow a new level of information flow between the patient and provider for all stages of health and disease.
CONCLUDING COMMENTS
Current management protocols for plaque-associated periodontal conditions are reactive to the disease signs and symptoms. The P4 approach in medicine and periodontal care recognizes that chronic disease should be managed in a more proactive manner, with the goal of achieving healthy aging. It embraces four key aspects of health care, leading to a truly holistic approach. Using the themes of prediction, prevention, personalization and participation, patients and health care providers work as a team to promote health and well-being — throughout life and across the health continuum, from health to advanced disease. The basis of this model is data gathering and taking a systems approach through the use of sophisticated and ever-evolving technologic advances, multilevel interdisciplinary interactions, and a fully participating patient.
REFERENCES
- Flores M, Glusman G, Brogaard K, Price ND, Hood L. P4 medicine: How systems medicine will transform the healthcare sector and society. Per Med. 2013;10:565–576.
- Hood L. Systems biology and P4 medicine: past, present, and future. Rambam Maimonides Med J. 2013;4:e0012.
- Kassebaum NJ, Smith AG, Bernabé E, et al. Global, regional, and national prevalence, incidence, and disability-adjusted life years for oral conditions for 195 countries, 1990–2015: A systematic analysis for the global burden of diseases, injuries, and risk factors. J Dent Res. 2017;96:380–387.
- Glick M, Monteiro da Silva O, Seeberger GK, et al. FDI Vision 2020: Shaping the future of oral health. Int Dent J. 2012;62:278–291.
- Caton JG, Armitage G, Berglundh T, et al. A new classification scheme for periodontal and peri-implant diseases and conditions — Introduction and key changes from the 1999 classification. J Periodontol. 2018;89(Suppl 1):S1–S8.
- Lang NP, Bartold PM. Periodontal Health. J Clin Periodontol. 2018;45(Suppl 20):S9–S16.
- Chapple IL, Mealey BL, Van Dyke TE, et al. Periodontal health and gingival diseases and conditions on an intact and a reduced periodontium: Consensus report of workgroup 1 of the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions. J Periodontol. 2018;89(Suppl 1):S74–S84.
- Papapanou PN, Sanz M, Buduneli N, et al. Periodontitis: Consensus report of workgroup 2 of the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions. J Periodontol. 2018;89(Suppl 1):S173–S182.
- Kornman KS. Contemporary approaches for identifying individual risk for periodontitis. Periodontol 2000. 2018;78:12–29.
- Hirschfeld L, Wasserman B. A long-term survey of tooth loss in 600 treated periodontal patients. J Periodontol. 1978;49:225–237.
- McFall WT Jr. Tooth loss in 100 treated patients with periodontal disease. A long-term study. J Periodontol. 1982;53:539–549.
- Kornman KS, Giannobile WV, Duff GW. Quo vadis: what is the future of periodontics? How will we get there? Periodontol 2000. 2017;75:353–371.
- Bartold PM, Seymour GJ, Cullinan MP, Westerman B. Effect of increased community and professional awareness of plaque control on the management of inflammatory periodontal diseases. Int Dent J. 1998;48:282–289.
- Bartold PM, Van Dyke TE. Periodontitis: a host mediated disruption of microbial homeostasis. Unlearning learned concepts. Periodontol 2000. 2013;62:203–217.
- Bartold PM, Van Dyke TE. Host modulation: Controlling the inflammation to control the infection. Periodontol 2000. 2017;75:317–329.
- U.S. Department of Health and Human Services, Office of Disease Prevention and Health Promotion. National Action Plan to Improve Health Literacy. Available at: https://health.gov/communication/hlactionplan/pdf/Health_Literacy_Action_Plan.pdf. Accessed April 23, 2019.
- Loechner J. Fitness self trackers and wearables merge. Available at: https://www.mediapost.com/publications/article/242885/fitness-self-trackers and-wearables-merge.html. Accessed April 23, 2019.
- Tiffany B, Blasi P, Catz SL, McClure JB. Mobile apps for oral health promotion: Content review and heuristic usability analysis. JMIR Mhealth Uhealth. 2018;6:e11432.
Featured image by ISAYILDIZ/ ISTOCK/ GETTY IMAGES PlUS
From Decisions in Dentistry. June 2019;5(6):25–30.