 OZINA/ISTOCK/GETTY IMAGES PLUS
OZINA/ISTOCK/GETTY IMAGES PLUS
Applications for Platelet Rich Fibrin in Dentistry
Useful in various dental procedures, platelet rich fibrin promotes wound healing, guides epithelial cell migration, and can be used as an adjunct for bone and soft tissue regeneration.
This course was published in the April 2021 issue and expires April 2024. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Explain the use of platelet rich fibrin in dentistry, and the generational evolution of autologous platelet concentrates.
- Discuss the processes by which platelet rich fibrin promotes wound healing.
- List clinical applications for various autologous platelet concentrates.
The phenomenon of wound healing has intrigued clinicians and researchers for decades. Various bioactive surgical additives have been developed and explored for inflammation regulation and enhanced wound healing.1 Platelet contains a high concentration of essential growth factors that stimulate cell migration and proliferation, which support wound healing.2 Due to this favorable property, autologous platelet concentrates were introduced as a therapeutic modality for expedited healing. The autologous platelet principle revolves around accumulating platelets from the patient’s whole blood in a fibrin clot, and utilizing it for hard and soft tissue healing.1,3 Platelet rich fibrin is an autologous leucocyte platelet rich fibrin matrix containing cytokines, platelets and numerous growth factors.4 It is a biodegradable scaffold that promotes wound healing, guides epithelial cell migration, and is used as an adjunct for bone and soft tissue regeneration.4
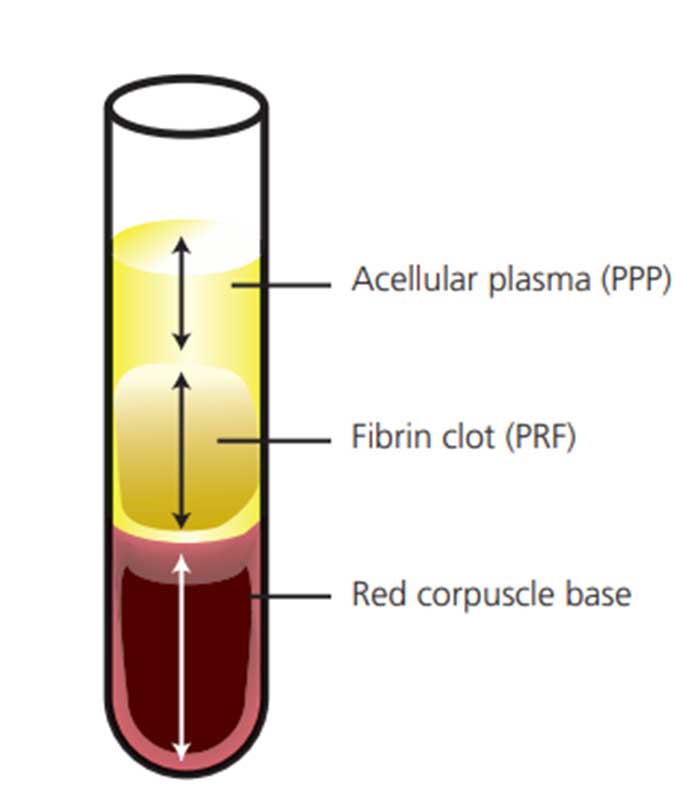
Platelet rich fibrin protocol requires single centrifugation without the addition of an anticoagulant; 10 ml of venous blood is taken and added to sterile glass tubes and immediately centrifuged at 3000 rpm for 10 minutes. The fibrin clot is formed in the middle layer, in which most of the platelets and leucocytes are concentrated. The topmost layer consists of cellular plasma, and the base is formed by the red blood cells (Figure 1).5
AUTOLOGOUS PLATELET CONCENTRATE GENERATIONS
The protocol of platelet rich fibrin formation has evolved over time. The first generation of platelet concentrate from autologous blood used for tissue regeneration was platelet rich plasma (PRP). This process involves adding an anticoagulant to the venous blood and two centrifugations (soft and hard spins) to obtain the concentrated PRP.1 However, the clinical protocol was cumbersome, and the addition of anticoagulants and bovine serum poses a risk of cross contamination.6
To combat these limitations, a second-generation platelet concentrate, platelet rich fibrin, was developed.2,6 The second generation, called leukocyte-PRF (L-PRF) or Choukroun’s PRF, was made using 10 ml of autologous blood, centrifuged once (3000 rpm for 10 minutes) without added anticoagulants or bovine serum.3,6 The fibrin aspect allows for better scaffolding that mirrors a natural blood clot. Choukroun’s PRF is well studied and involves autologous blood that contains platelets, lymphocytes, monocytes, stem cells, granulocytes and growth factors.6 More processing techniques were introduced for newer preparations. These techniques use lower centrifugation forces (1500 rpm for 10 minutes), and the formulations are called advanced platelet rich fibrin, which demonstrated an increase in platelets and neutrophilic granules.7 Recently, an injectable platelet rich fibrin was developed, with one centrifugation cycle for 700 rpm for three minutes. It contains a larger quantity of leucocytes and plasma proteins due to the low centrifugation speed.7
WHAT MAKES IT UNIQUE?
Platelet rich fibrin is created from autogenous blood and is unique to the patient. It can connect elements in tissue for a better clinical outcome. For example, mixing bone graft with platelet rich fibrin can help the grafted tissues heal more efficiently. The added matrix components, such as fibrin, glycosaminoglycans, fibronectin and others, aid this process.8 Growth factors released from the platelet rich fibrin clot in vitro after five to 10 minutes of the initial clotting process, and continue to be released for three to five hours.7–9 The architecture of the clot functions as a scaffold for tissue regeneration.7 It can carry mesenchymal cells and allow them to differentiate and proliferate. He et al9 studied the release of TGF-β, platelet-derived growth factor-AB, and alkaline phosphatase from L-PRF, and found that all three were released at a slow, constant rate. Platelet-derived growth factor, which is naturally found in platelets and other cells, is actively involved in wound healing and has shown improved bone regeneration potential.10 Ghanaati et al6 noted that T-lymphocytes, B-lymphocytes, stem cells and monocytes were in the first 25% to 30% proximal part of the platelet rich fibrin clot. The investigators also found that using different settings for centrifugation could alter the clot’s specific cell types. These other centrifugation protocols created either standard platelet rich fibrin or advanced platelet rich fibrin, based on rpm and centrifugation time.6
APPLICATIONS
Platelet rich fibrin is commonly used in oral and maxillofacial surgery to aid healing. It is taken as an autologous blood sample, centrifuged, and placed into the surgical site as a clot. The architecture of the fibrin in trapping cytokines and growth factors, along with the leukocyte content, play an essential role in its ability to accelerate healing. The most common oral and maxillofacial surgery preparations are L-PRF. These autologous fibrin clots and platelet concentrates mirror the effects of proper bleeding from a surgical site — that is, they allow the site to form a good fibrin clot, stimulate neoangiogenesis, and provide the architecture for leukocytes to protect the area from infection and clean debris. This circumnavigates issues with lack of clotting or controlled bleeding from the surgical site.
Platelet rich fibrin has been used in periodontal surgery as a barrier between tissues to prevent healing by a long junctional epithelium. It has also been used to avoid damage of gingival tissues during surgery with bone grafting, and accelerate soft tissue healing. In guided tissue regeneration (GTR), L-PRF can be used as a filling material and membrane. The platelet rich fibrin acts as a blood clot and barrier membrane, where the blood clot must first be stabilized, and a barrier must be created. The L-PRF membrane is compatible with the host tissue and does not interfere with the natural healing process.
More robust than a natural blood clot, L-PRF can be used as a filling material when regenerating hard tissue in intrabony defects.8 The increased strength of the platelet rich fibrin compared to PRP gel makes it easier to manipulate into the desired sites. The release of growth factor allows for quicker remodeling of soft tissues when L-PRF is used as a protective membrane. Since the L-PRF membrane boosts the periosteum’s healing properties, it augments the periosteum in protecting intrabony defects as they heal.8 In a randomized controlled trial, the adjunctive use of platelet rich fibrin with open flap debridement showed significant improvement compared to the control group (open flap debridement only) for bone fill, reduction in probing depths, and soft tissue healing.11
Platelet rich fibrin can also be used as an adjunct to palatal wound healing after harvesting a palatal graft or along with root coverage procedures to aid accelerated soft tissue healing and achieve root coverage.4,12 It is also widely utilized in implantology; for example, as an adjunct in socket grafting procedures — with or without the use of bone grafts — to maintain alveolar ridge dimensions and enhance healing.13,14 Better clinical and histological results were seen in groups treated with platelet rich fibrin in comparison to non-grafted control sites; similar improvements were seen in the preservation of alveolar ridge dimensions and patient comfort.13,14
Due to its favorable healing properties, platelet rich fibrin is also utilized as a grafting material in sinus augmentation procedures, in combination with bone grafts, or as a membrane to cover the graft material, or for the repair of a perforated Schneiderian membrane.15–17
In endodontics, platelet rich fibrin has been used as a successful scaffold for pulp revascularization procedures of the immature necrotic tooth by upregulating cellular proliferation, differentiation and angiogenesis.18
ADVANTAGES AND DISADVANTAGES
As noted, the primary advantage of platelet rich fibrin is its ability to promote wound healing. There are four phases to healing: hemostasis, inflammation, proliferation and maturation. Each phase encompasses various cell types that must interact with a three-dimensional (3D) extracellular matrix and growth factors to obtain successful results. The platelet rich fibrin element is host cells’ presence (i.e., platelets, leukocytes and red blood cells); in particular, leukocytes play a vital role in the formation of new blood vessels and tissue. The second component that promotes healing is the 3D fibrin matrix. Unlike PRP, this matrix is obtainable due to acquiring platelet rich fibrin without anticoagulants (which delay wound healing). The last component is its ability to serve as a reservoir of natural growth factors released for 10 to 14 days.7
The major disadvantage of platelet rich fibrin is the requirement of an autologous blood source that contains various immune cells and highly antigenic plasmatic molecules in the fibrin matrix. These cells’ presence makes platelet rich fibrin donor-specific, therefore limiting its use as an allogenic graft tissue.19 Additionally, the required use of autologous blood restricts the amount of platelet rich fibrin available, as a low quantity is produced; thus, it can only be used in limited amounts.19 The protocol for platelet rich fibrin preparation is also not standardized, which could also pose potential limitations.5
CASE PRESENTATIONS
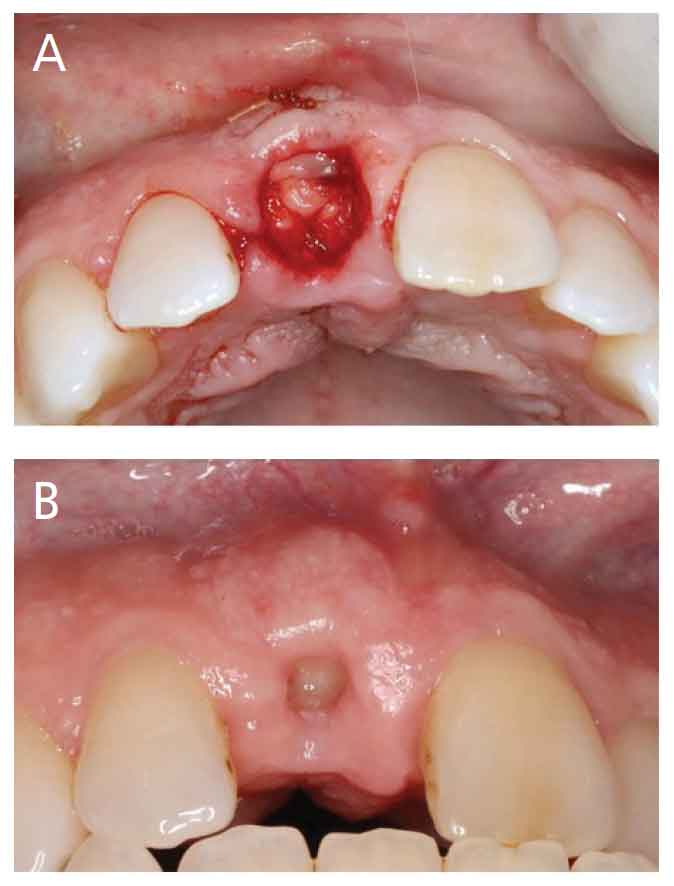
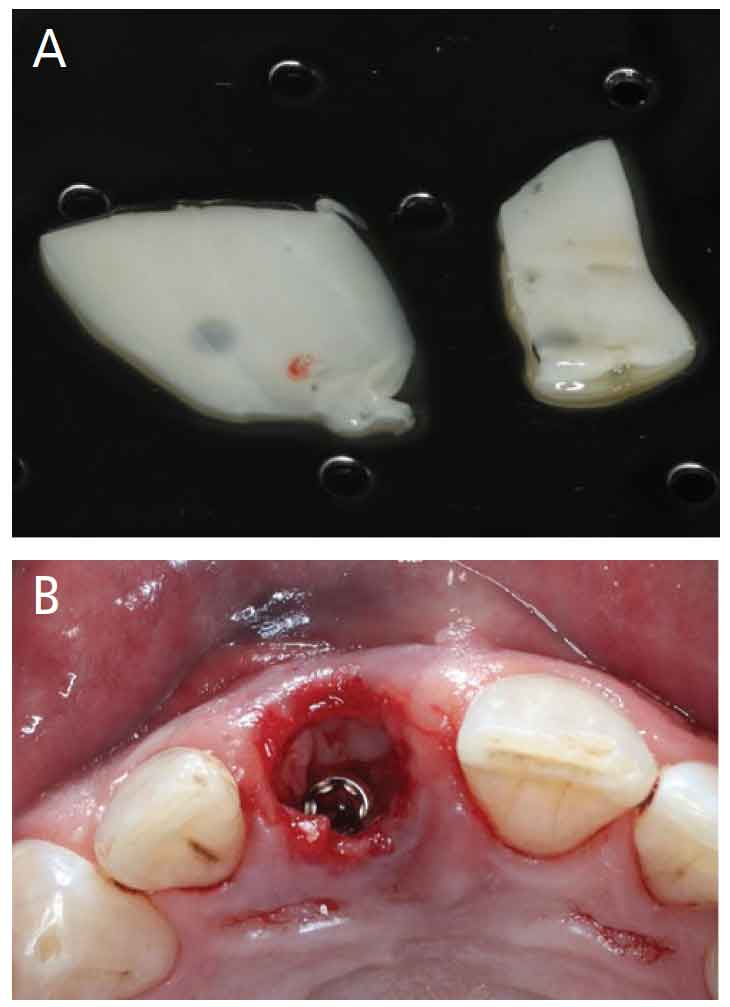
Case 1: A 54-year-old male presented with internal root resorption of tooth #8. His medical history was unremarkable other than hypertension controlled with medication. The buccal bone was less than 1 mm thick, per the cone beam computed tomography scans, which was confirmed clinically during extraction. A staged approach was chosen for implant placement. Socket preservation was performed using a 50/50 mixture of freeze-dried bone allograft (FDBA) and demineralized FDBA (DFDBA) allograft particulate that was hydrated using the platelet rich fibrin exudate obtained after compression in the lower chamber. The platelet rich fibrin was flattened and folded lengthwise in half. The platelet rich fibrin membrane was placed over the graft material and secured using an internal cross mattress 4-0 chromic gut suture (Figure 2A). Figure 2B shows the patient at his 10-day postoperative visit. The patient stated his healing was uneventful, and he only had the over-the-counter pain medication given to him at the time of surgery.
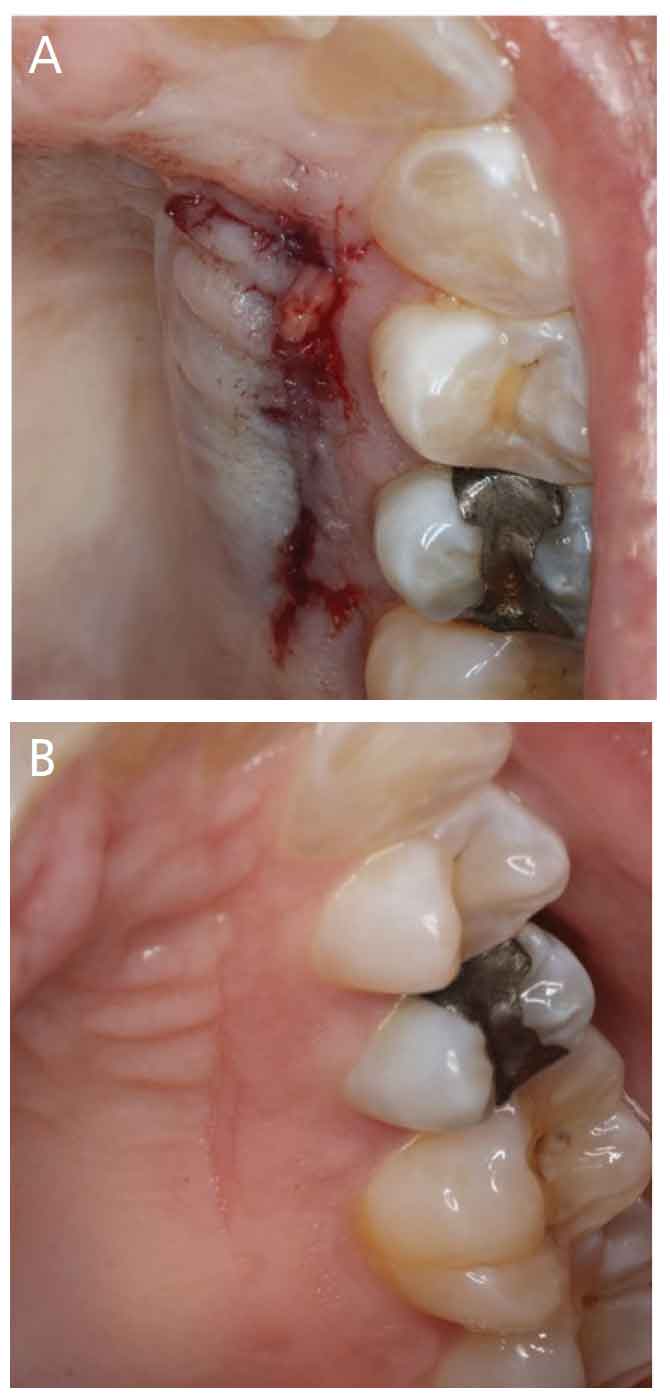
Case 2: This patient is a 27-year-old healthy male who presented with a 4 mm mucogingival recession defect on tooth #22. A subepithelial connective tissue graft was performed to increase the amount of keratinized tissue and achieve root coverage. A single-incision technique was used to harvest the connective tissue from the left palate. A flattened platelet rich fibrin membrane was inserted in the donor site, and pressure was applied to achieve hemostasis. The single incision was secured using cyanoacrylate. Figure 3A shows the closed donor site.
In some cases, hemostasis is not achieved right away. The cyanoacrylate seal allows the area to tamponade, and the platelet rich fibrin helps stabilize the blood clot. Figure 3B shows the palate at the two-week postoperative visit. The patient reported no pain or issues during healing. Platelet rich fibrin used in palatal donor sites can be a practice builder, especially when patients have experiences with prior grafts from the roof of the mouth or have heard stories of pain from palatal harvests.
Case 3: A 57-year-old male presented with a medical history of hypertension (moderately controlled with medication), and tooth #9 fractured at the gingival margin. The cone beam computed tomography scans showed 1 mm of facial bone, which was confirmed intraoperatively. Extraction and an immediate implant with immediate provisional were planned. Tooth #9 was extracted with minimal trauma, and the site was prepped for a 4×12-mm implant. The implant access hole was positioned toward the anticipated future cingulum of tooth #9, which created a jump gap between the implant and buccal bone of more than 2 mm. This area was grafted using a 50/50 mixture of FDBA and DFDBA particulate allograft hydrated in the platelet rich fibrin exudate. A trimmed platelet rich fibrin membrane (Figure 4A) was placed in the jump gap over the bone particulate seen in Figure 4B. A well-contoured provisional was placed, with no need for sutures to stabile the membrane. Healing proved uneventful.
DISCUSSION AND CONCLUSION
Relatively inexpensive and simple to use, L-PRF can lead to healthy fibrin membrane production. It can be used in patients who are typically at risk for compromised healing, such as smokers, patients using anticoagulants, and those with uncontrolled diabetes. Using L-PRF membrane is not as technique sensitive as other modalities, and can have good clinical outcomes. It can also be used during surgeries where a flap has been made; this approach can reduce the risk of necrosis, promote soft tissue healing and angiogenesis, and can help heal a damaged flap. However, L-PRF cannot be used as a cell-proof barrier in GTR techniques because it allows cell migration. This ability to enable cell migration improves neoangiogenesis and bone healing through interactions with the gingival flap’s periosteum. In fact, L-PRF membranes stimulate the periosteum to help with regeneration — unlike GTR membranes, which block the periosteum by acting as a barrier to soft tissue invagination.8 Clinicians should understand that many of these studies were done using in vitro cell models. Clinical evidence of benefits from platelet rich fibrin from randomized clinical trials is sparse, with neutral outcomes. However, many case reports and case series have shown favorable results with the use of platelet rich fibrin. Platelet concentration technology can be expensive, time-consuming, and fail to provide enough benefits to be worth the investment.8 The protocol for platelet rich fibrin preparation has not been standardized, which can account for the variation in findings when examining cytokine content.5
In vitro and in vivo evidence confirm that platelet rich fibrin is a potential healing biomaterial with various applications in dentistry. It has demonstrated promising results, with better healing outcomes and decreased postoperative discomfort for patients. Platelet rich fibrin can be utilized for many periodontal, implant, oral surgical and endodontic procedures, maximizing its benefits as an autologous source, and enhancing healing and regeneration potential. Ultimately, this approach offers a less time consuming and less expensive alternative to commercially available materials.
REFERENCES
- Dohan DM, Choukroun J, Diss A, et al. Platelet-rich fibrin (PRF): a second-generation platelet concentrate. Part I: technological concepts and evolution. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006;101:e37–e44.
- Ehrenfest DM, Rasmusson L, Albrektsson T. Classification of platelet concentrates: from pure platelet-rich plasma (P-PRP) to leucocyte-and platelet-rich fibrin (L-PRF). Trends Biotechnol. 2009;27:158–167.
- Dohan DM, Choukroun J, Diss A, et al. Platelet-rich fibrin (PRF): a second-generation platelet concentrate. Part II: platelet-related biologic features. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006;101:e45–e50.
- Borie E, Oliví DG, Orsi IA, et al. Platelet-rich fibrin application in dentistry: a literature review. Int J Clin Exp Med. 2015;8:7922–7929.
- Gabling VL, Açil Y, Springer IN, Hubert N, Wiltfang J. Platelet-rich plasma and platelet-rich fibrin in human cell culture. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2009;108:48–55.
- Ghanaati S, Booms P, Orlowska A, et al. Advanced platelet-rich fibrin: a new concept for cell-based tissue engineering by means of inflammatory cells. J Oral Implantol. 2014;40:679–689.
- Miron RJ, Choukroun J. Platelet Rich Fibrin in Regenerative Dentistry: Biological Background and Clinical Indications. Hoboken, New Jersey: John Wiley & Sons; 2017.
- Del Corso M, Vervelle A, Simonpieri A, et al. Current knowledge and perspectives for the use of platelet-rich plasma (PRP) and platelet-rich fibrin (PRF) in oral and maxillofacial surgery part 1: Periodontal and dentoalveolar surgery. Curr Pharm Biotechnol. 2012;13:1207–1230.
- He L, Lin Y, Hu X, Zhang Y, Wu H. A comparative study of platelet-rich fibrin (PRF) and platelet-rich plasma (PRP) on the effect of proliferation and differentiation of rat osteoblasts in vitro. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2009;108:707–713.
- Anitua E. Plasma rich in growth factors: preliminary results of use in the preparation of future sites for implants. Int J Oral Maxillofac Implants. 1999;14:529–535.
- Patel GK, Gaekwad SS, Gujjari SK, Kumar V. Platelet–rich fibrin in regeneration of intrabony defects: a randomized controlled trial. J Periodontol. 2017;88:1192–1199.
- Jankovic S, Aleksic Z, Klokkevold P, et al. Use of platelet-rich fibrin membrane following treatment of gingival recession: a randomized clinical trial. Int J Periodontics Restorative Dent. 2012;32:e41–e50.
- Hauser F, Gaydarov N, Badoud I, Vazquez L, Bernard JP, Ammann P. Clinical and histological evaluation of postextraction platelet-rich fibrin socket filling: a prospective randomized controlled study. Implant Dent. 2013;22:295–303.
- Temmerman A, Vandessel J, Castro A, et al. The use of leucocyte and platelet‐rich fibrin in socket management and ridge preservation: a split‐mouth, randomized, controlled clinical trial. J Clin Periodontol. 2016;43:990–999.
- Choukroun J, Diss A, Simonpieri A, et al. Platelet-rich fibrin (PRF): a second-generation platelet concentrate. Part V: histologic evaluations of PRF effects on bone allograft maturation in sinus lift. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006;101:299–303.
- Ali S, Bakry SA, Abd-Elhakam H. Platelet-rich fibrin in maxillary sinus augmentation: A systematic review. J Oral Implantol. 2015;41:746–753.
- Tajima N, Ohba S, Sawase T, Asahina I. Evaluation of sinus floor augmentation with simultaneous implant placement using platelet-rich fibrin as sole grafting material. Int J Oral Maxillofac Implants. 2013;28:77–83.
- Keswani D, Pandey RK. Revascularization of an immature tooth with a necrotic pulp using platelet‐rich fibrin: a case report. Int Endod J. 2013;46:1096–1104.
- Choukroun J, Diss A, Simonpieri A, et al. Platelet-rich fibrin (PRF): a second-generation platelet concentrate. Part IV: clinical effects on tissue healing. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006;101:e56–e60.
From Decisions in Dentistry. April 2021;7(4):26–28,31.




