Antibiotic Stewardship and C. Difficile Infection
The need for robust stewardship practices gains prominence in protecting patients from unnecessary antibiotic use, improving outcomes, and curbing the rise of antibiotic-resistant bacteria, especially Clostridioides difficile
This course was published in the September 2023 issue and expires September 2026. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
AGD Subject Code: 010
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Define antibiotic stewardship.
- Discuss the risks of Clostridioides difficile infection.
- Identify treatments for C. difficile.
Antibiotic stewardship is the effort to measure antibiotic prescribing. As of January 2023, the Joint Commission, working with hospitals and dental schools, revised the antibiotic stewardship requirements.1 The goals of antibiotic stewardship are to protect patients from unnecessary use of antibiotics, improve clinical outcomes, and minimize any harm to patients using antibiotics.
In the United Kingdom and the United States, approximately 10% of the antibiotics prescribed are through dental practices, resulting in more than 25 million prescriptions a year.2 At the start of 2020, the dental community was included in the outpatient antibiotic stewardship program.3 Thompson et al4 report that less emphasis has been placed on antibiotic stewardship for dental office prescribing compared to medical office prescribing.
More than 72% of the dental community is composed of dentists in private practice who do not report to any accreditation body, making it difficult to track the number of antibiotic prescriptions written. With this in mind, dentists need to formulate definitive treatment plans and educate patients on how they provide optimal long-term outcomes, not just short-term symptomatic relief.4
Dentists play a crucial role in educating their patients about the risk-benefit ratio of using antibiotics for specific conditions. Antibiotics are commonly prescribed in dentistry to treat or prevent infections, but they must be used judiciously to minimize the risk of antibiotic resistance and other potential side effects.
A review by Stein et al5 delved into the antibiotic prescribing behaviors within dentistry. From the pool of 1,912 studies reviewed, 29 were centered on prophylactic and therapeutic antibiotics in dentistry. The use of antibiotics in dentistry is categorized into primary and secondary prophylaxis. Primary usage pertains to patients who are either healthy or medically compromised, believed to be susceptible to infection, and are undergoing invasive dental procedures.
Secondary usage involves patients with cardiac conditions who are considered at risk of developing infective endocarditis or possible infections in joint replacements, particularly within the initial 2 to 3 years post-replacement. The study also looked at the lifelong use of premedication for joint replacements.
The review’s analysis found that patients with medical vulnerabilities, such as rheumatic heart disease, coronary artery bypass graft, mitral valve prolapse, recent myocardial infarction, and total joint replacements, often receive prescriptions for antibiotic prophylaxis from dentists. However, dentists sometimes resort to prophylactic antibiotics for nonclinical reasons. This may stem from patient pressure or recommendations by cardiologists or physicians. Moreover, the unnecessary prescription of antibiotics has also been linked to a lack of awareness and the gradual adoption of new guidelines.
An alarming issue is the rise of antibiotic-resistant bacteria, given that patients contracting such infections face a heightened risk of severe clinical outcomes, potentially leading to death. The overuse of antibiotics has resulted in the emergence of resistant microorganisms, posing a substantial threat to public health. In response, the World Health Organization endorsed a global action plan in May 2015, with a particular emphasis on combating antibiotic resistance.
Adverse Effects of Antibiotics
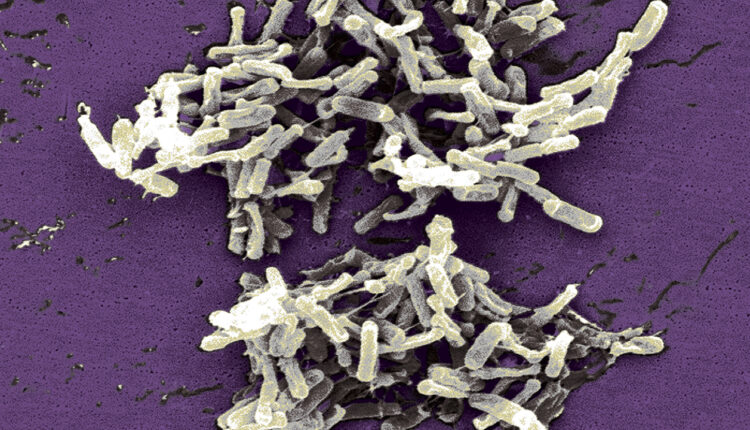
A significant concern surround the overprescription of antibiotics is the development of Clostridioides difficile (Figure 1). All oral health professionals must understand the importance of antibiotic stewardship and its relationship to C. difficile.
 While antibiotics are one of the most profound discoveries of the 20th century,6 many negatively affect gut bacteria. When gut bacteria are affected, the normal bacterial flora of the colon colonize with C. difficile and then release toxins that cause mucosal damage and inflammation.7,8 Table 1 lists symptoms of C. difficile infection.9
While antibiotics are one of the most profound discoveries of the 20th century,6 many negatively affect gut bacteria. When gut bacteria are affected, the normal bacterial flora of the colon colonize with C. difficile and then release toxins that cause mucosal damage and inflammation.7,8 Table 1 lists symptoms of C. difficile infection.9
C. difficile is normally found in gut bacteria, though at low levels. Once it gets out of balance in the presence of antibiotic treatment, difficile can become resistant to traditional antibiotics. C. difficile is responsible for 500,000 infections annually and approximately 29,000 deaths per year in the US.10
C. difficile is spore forming, and when the normal gut flora is upset, the greatest impact is on C. difficile cells. The C. difficile spores germinate, forming colonies within the intestine that disrupt the normal flora.
Clindamycin, a broad-spectrum antibiotic used to treat bacterial infections, is associated with increased risk for C. difficile infection.13 It is also the second most commonly prescribed antibiotic by dentists.11
Within the initial month of antibiotic treatment, patients face a seven to 10-fold heightened risk of developing a C. difficile infection.12 Research shows that the risk of diarrhea related to antibiotic use decreases 1 to 3 months after the drug has been discontinued.12
Certain individuals are more susceptible to antibiotic-related C. difficile infections. Patient populations at greater risk for developing C. difficile infection include:
- Age 65 and older
- Recently stayed at a healthcare facility
- Weakened immune system
- Past history of C. difficile infections
Medical history forms should be edited to include the questions, “Have you ever had a C. difficile infection?” and “Do you experience diarrhea related to antibiotic use?” Oral health professionals who have prescription capabilities within their scope of practice must participate in continuing education classes that discuss antibiotic stewardship training.
C. difficile infection is a potentially life-threatening condition that can cause severe diarrhea and colitis. Dentists must carefully consider the risks and benefits of prescribing clindamycin, especially in patients who are at increased risk for C. difficile infection.11 Alternative antibiotics with a lower risk of causing C. difficile infection, such as azithromycin, should be considered.
Cleaning and hand hygiene
C. difficile is highly contagious and may spread through direct contact (person to person) or indirect contact (healthcare community setting to person).13 An asymptomatic carrier can shed C. difficile spores for 5 months or more, and spores may survive on contaminated surfaces for months to years.13 Not all dental disinfectants are effective against C. difficile, therefore, staff must verify that disinfection products are effective at disrupting C. difficile spores.
Compliance with hand hygiene techniques is key to reducing transmission of C. difficile spores and other bacteria, as oral health professionals can carry the bacteria from room to room on their hands.14
Treatment Options
Fidaxomicin is an antibiotic specifically approved for the treatment of C. difficile infection. Vancomycin is also used to treat C. difficile. Fidaxomicin, however, has been shown to be more effective than vancomycin at reducing the recurrence of C. difficile infection. Recurrence is a significant concern, as it can lead to prolonged illness, increased healthcare costs, and potential complications.15
The decision to use fidaxomicin or vancomycin should be made in consultation with healthcare providers, while considering factors such as the patient’s specific condition, medical history, local resistance patterns, and resource availability.15
The availability and cost of fidaxomicin can vary across healthcare settings and regions. It is generally more expensive than vancomycin, which may impact its accessibility in some healthcare systems. In such cases, vancomycin remains a reasonable alternative for treatment of C. difficile infection.15
Bezlotoxumab is a monoclonal antibody that targets the toxin produced by C. difficile bacteria. It is used as an adjunctive therapy along with standard-of-care antibiotics to reduce the recurrence of C. difficile infection. The rationale behind using bezlotoxumab is that it may help prevent a relapse of C. difficile infection by neutralizing the toxins produced by the bacteria.15 However, the evidence supporting its effectiveness is limited, meaning significant uncertainty exists regarding bezlotoxumab’s benefits and risks.15
Fecal microbiota transplantation (FMT) is used to treat severe C. difficile cases. This approach can control C. difficile infection by adding healthy bacteria into the patient’s intestines. This procedure uses a slurry solution that originates from healthy, donated human stools, or feces. Healthy stool donors must be without any antibiotic exposure in the past 6 months, are not immunocompromised, are not at elevated risk for C. difficile infection or any other infectious disease, and do hot have any chronic gastrointestinal diseases. They are also screened for hepatitis A, B, and C; human immunodeficiency virus; syphilis; parasites including giardiasis or worms; and C. difficile infection.
Once the donor is tested, the stools are collected, mixed with saline or another solution, and strained. The slurry is delivered to the patient using one of the following methods.16,17–20
In the upper gut:
- Taken orally via capsule
- Nasogastric tube
In the mid gut:
- Gastroscopy in duodenum
- Mid-gut tubing
- Percutaneous endoscopic transgastric jejunostomy tube
In the lower gut:
- Colonoscopy
- Colonic transendoscopic enteral tube
- Stoma in ileocolonic
Antibiotic Prophylaxis
The use of antibiotic prophylaxis for dental procedures is controversial. Some studies suggest the risk of infective endocarditis (IE) from dental procedures is relatively low, and that the risks associated with taking antibiotics may outweigh the benefits in many cases.21
A study by Hussein et al21 found that the benefits of antibiotic prophylaxis for IE outweigh the risks, even though the evidence for its effectiveness may be lacking, especially in patients at high risk for IE. Some national and international accreditation bodies require antibiotic prophylaxis in dental procedures based on the principles of antibiotic stewardship.21
Antibiotic overuse and misuse are long-standing issues that contribute to the development of antimicrobial resistance (AMR) — a global public health threat.13 Little research exists on the prescription writing practices of the dental community. Recognizing the importance of antibiotic stewardship will help lower C. difficile infections and AMR.
All healthcare professionals should continue to prioritize cautious antibiotic prescribing to help reduce the risk of AMR. This means prescribing antibiotics only when they are truly necessary, based on evidence-based guidelines and appropriate clinical judgment, and using the narrowest spectrum antibiotic that is effective.
Conclusion
The dental community must understand antibiotic stewardship and its relationship to C. difficile. Antibiotics are not needed for all dental procedures and are not the answer to all nonsurgical dental procedures. Dentists play an important role in promoting patient education and awareness about AMR and the importance of responsible antibiotic use.22 This may help reduce patient demand for antibiotics when they are not needed and to encourage patients to complete the full course of antibiotics as prescribed.
References
- The Joint Commission. Antimicrobial Stewardship. Available at: jointcommission .org/resources/patient-safety-topics/infection-prevention-and-control/antibiotic-stewardship. Accessed August 4, 2023.
- Thompson W, Sandoe J, Pavitt S, Walsh T, Byrne-Davis L. Co-Developing an antibiotic stewardship tool for dentistry: Shared decision-making for adults with toothache or infection. J Antibiot. 2021;10:1345.
- Goff DA, Mangino JE, Trolli E, Scheetz R, Goff D. Private practice dentists improve antibiotic use after dental antibiotic stewardship education from infectious diseases experts. Open Forum Infect Dis. 2022;9:1-9.
- Thompson W, Douglas G, Pavitt S, Sandoe J, McEachan R, Tonkin-Crine S. Factors associated with prescribing of systemic antibacterial drugs to adult patients in urgent primary health care, especially dentistry. J Antimicrob Chemother. 2019;74:2139–2152.
- Stein K, Farmer J, Singhal S, et al. The use and misuse of antibiotics in dentistry: A scoping review. J Am Dent Assoc. 2018;149:869–884.
- Chung-Jong K. Current status of antibiotic stewardship and the role of biomarkers in antibiotic stewardship programs. J Infect Chemother. 2022;54:674-698.
- Kelly CP, Pothoulakis C, LaMont T. Clostridium difficile colitis. N Engl J Med. 1994;330:257–262.
- McCune VL, Struthers JK, Hawkey PM. Fecal transplantation for the treatment of Clostridium difficile infection: a review. Int J Antimicrob Agents. 2014;43:201–206.
- Mayo Clinic. Clostridioides Difficile Infection. Available at: mayoclinic.org/diseases-conditions/c-difficile/symptoms-causes/syc-20351691. Accessed August 4, 2023.
- Lessa FC, Mu Y, Bamberg WM, et al. Burden of Clostridium difficile infection in the United States. N Engl J Med. 2015;372:825–834.
- Mashrah H, Sharma P, Kadhium V, Dault J, Nalliah R. Original Research: Antibiotic prescribing trends among dentists. Decisions in Dentistry. 2021;7(5)26–31.
- Hensgens MP, Goorhuis A, Dekkers OM, Kuijper EJ. Time interval of increased risk for clostridium difficile infection after exposure to antibiotics. J Antimicrob Chemother. 2012;67:742–748.
- Jury LA, Sitzlar B, Kundrapu S, et al. Outpatient healthcare settings and transmission of clostridium difficile. PloS one. 2013;8:
- Banks M, Phillips AB. Evaluating the effect of automated hand hygiene technology on compliance and C. difficile rates in a long-term acute care hospital. Am J Infect Control. 2021;49:727–732.
- Johnson S, Lavergne V, Skinner AM, et al. Clinical practice guideline by the Infectious Diseases Society of America and Society for Healthcare Epidemiology of America: 2021 focused update guidelines on management of Clostridioides difficile infection in adults. Clin Infect Dis. 2021;73:e1029-e1044.
- United States Centers for Disease Control and Prevention. FAQs for Clinicians About C. diff. Available at: cdc.gov/cdiff/clinicians/faq.html. Accessed August 4, 2023.
- Wang Y, Zhang S, Borody TJ, Zhang F. Encyclopedia of fecal microbiota transplantation: a review of effectiveness in the treatment of 85 diseases. Chin Med J. 2022;135:1927–1939.
- The Fecal Transplant Foundation. What is FMT? Available at: thefecaltransplantfoundation.org/what-is-fecal-tran. plant. Accessed August 4, 2023
- Clindamycin: bacterial infections. In: Wynn, RL, Meiller TF, Crossley HL, eds. Drug Information Handbook for Dentistry. 28th ed. Hudson, Ohio: Wolters Kluwer: 1863.
- Ratner M. Microbial cocktails raise bar for C. diff. treatments. Nat Biotechnol. 2020;38:1366–1367.
- Hussein H, Montesinos-Guevara C, Abouelkheir M, Brown R, Hneiny L, Amer Y. Quality appraisal of antibiotic prophylaxis guidelines to prevent infective endocarditis following dental procedures: a systematic review. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2022:134:562–572.
- Healthline. Clindamycin for Tooth Infection: Purpose, Benefits, Side Effects. Available at: healthline.com/health/clindamycin-for-tooth-infection#benefits. Accessed August 4, 2023.
From Decisions in Dentistry. September 2023; 1(8):42-45.




