 MEDIOIMAGES/PHOTODISC/GETTY IMAGES PLUS
MEDIOIMAGES/PHOTODISC/GETTY IMAGES PLUS
Lubricating Adhering Discs for Relief of Oral Dryness
A literature review and report examining the use of oral adhering discs for patients experiencing xerostomia.
Oral dryness impacts oral health in a number of ways. It can cause throat, tongue, gingival and denture ridge pain,1 changes in taste, speech and swallowing,2 and sensory changes, such as tingling, burning or stickiness. When it occurs during the day, it can be managed with various over-the-counter (OTC) solutions, gels, sprays, rinses and similar products.3–10 By comparison, there are few management options for patients who experience nighttime oral dryness, when prescribed saliva-stimulating drugs may not be suitable. In these cases, an OTC lubricating oral adhering disc may be a suitable option (Figure 1).
This paper will review six published studies assessing the efficacy of two oral adhering disc products in the management of xerostomia occurring during the day or while sleeping. It will also report on a seventh study that tested a disc product for its potential effect on acid reflux, as well as oral dryness.
Five of the studies examined the efficacy of OraCoat XyliMelts in reducing xerostomia symptoms, while two studies evaluated OraMoist adhering discs. Of the latter, one was a comparative study with very different products, and the second was a double-masked, crossover design, short-term prospective study with two nearly identical adhering discs, but with different ingredients.
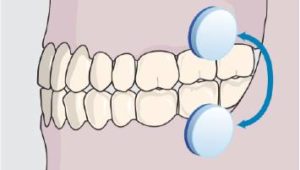
OraMoist Adhering Discs in Awake Subjects Compared to Mouthrinse — In 2010, Aframian et al11 compared the OraMoist lubricating and flavor-releasing adhering discs to a placebo in subjects with xerostomia. Twenty xerostomic patients were divided into two groups. In Group 1, the mucoadhesive tablet was applied to the hard palate, while in Group 2 a moisturizing mouthrinse was used as directed and served as a control. Sialometry measurements (consisting of the collection of whole saliva in a calibrated tube for 10 minutes), as well as a questionnaire assessing oral dryness (including four questions used in prior validated studies) were obtained before and 30 minutes after treatment.
Based on the answers to the four questions, a total xerostomic score was generated for statistical analysis. Application of the mucoadhesive discs reduced the sensation of oral dryness (P = 0.02) compared to the mouthrinse. Moreover, a mean 1.5-fold increase in unstimulated whole salivary flow was measured 30 minutes after disc placement in Group 1. Aframian et al11 concluded that use of a mucoadhesive tablet could have a beneficial role in reducing the sensation of dryness by stimulating salivary flow.
OraMoist Adhering Discs With Varying Ingredients in Awake Subjects — Kerr et al12 assessed the efficacy of OraMoist discs compared with a nearly identical disc, but without lubricating or sweetening agents. Twenty-seven subjects were identified as having limited salivary function and the sensation of xerostomia. This was determined through salivary testing (i.e., having an unstimulated whole salivary flow rate of ≤ 0.2 milliliter/minute) and a questionnaire (the New York University Bluestone Mouthfeel Questionnaire). The study used a randomized, double-masked crossover design. After a second visit involving a dental hygiene appointment, there was a one week no-treatment “washout” period prior to use of the disc products. Subjects using the OraMoist disc with lubricating agents and sweetener and those using the OraMoist discs without these components were asked to use three of the discs daily for one week, after which each subject returned for whole saliva measurements with a disc in their mouth at five and 60 minutes. This was followed by a second week of disc use and, at a final visit, another measurement of salivary flow with a disc in their mouth at five and 60 minutes. The aim of the study was to determine if disc ingredients affected efficacy.
All subjects reported the discs lasted at least an hour. Compared to baseline measures, both formulations reduced the perception of oral dryness 60 minutes after disc placement. Individuals in both groups also demonstrated a reduction in the sensation of xerostomia with discs after one and two weeks of use. In addition, a statistically significant improvement in salivary flow while having a disc in the mouth was measured in both groups after 60 minutes of use — both with initial use, and after one and two weeks of regular use. At the two-week time point, there was still no significant difference in xerostomia symptoms between the groups, even though the users of the discs with flavoring and lubricating agents demonstrated greater salivary flow than those using discs without sweetener or lubricant.
Kerr et al12 concluded that even though the study results did not demonstrate significant differences between the two formulations, the data show that mucoadhesive discs with and without flavoring ingredients could have a positive effect on the subjective experience of xerostomia. The discs were also shown to increase salivation with initial placement and with subsequent placement over a period of up to two weeks.
XyliMelts Adhering Discs Versus No Treatment, Randomized Crossover, Both Awake and While Sleeping — Ho et al13 studied the use of OraCoat XyliMelts oral adhering discs for xerostomia relief. For lubricant, the discs release cellulose gum, a hydrocolloid that forms a gel with saliva. For flavor, the discs release 550 mg of xylitol and mild mint. The study consisted of two arms. In one arm, the adhering discs were added to the subjects’ usual oral hygiene self-care. Each subject was instructed to use the discs as often as needed during the day to relieve xerostomia symptoms (mean usage 4.1 per day) and to place two discs intraorally while sleeping. In the second arm, subjects conducted their usual oral hygiene self-care without using discs. A 7-day washout period was used between the two study arms. During this period, subjects continued their usual oral hygiene self-care. Subjects were randomized with regard to the sequence of the two arms of the study. The production of saliva and salivary pH and buffering capacity were assessed, in addition to evaluation of other variables — including plaque index, gingival index and sulcus bleeding index.
It was found that salivary production more than doubled 10 and 40 minutes after disc placement (significance, p < 0.05). In addition, salivary buffering capacity improved with disc use in 80% of the subjects. A self-evaluation questionnaire demonstrated improvement in oral function, swallowing and xerostomia symptoms.
When discs were used while sleeping, the subjects reported an improvement in oral wetness upon waking. The flavor of the discs lasted approximately six hours. Ho et al13 concluded, “This research showed desirable effects on symptomatic management of xerostomia, demonstrating the test product provided effective local palliation and salivary stimulation, without the use of systemic sialagogue medications.”
XyliMelts Adhering Discs Versus Baseline of No Treatment While Sleeping — Burgess and Lee14 assessed the use of OraCoat XyliMelts adhering discs for managing dry mouth during sleep. The data demonstrated a significant reduction in the perception of xerostomia and oral discomfort upon waking.
The study was conducted as a non-blinded case series involving healthy subjects who reported xerostomia symptoms. Mean initial self-assessed oral wetness upon awakening (measured via the subject placing a mark on a visual 100-mm analog scale) was 22.2 (SD 15.3), and the mean self-assessed oral wetness score each morning after sleeping with two discs placed at bedtime increased to 67.8 (SD 15.0). The mean for initial discomfort upon waking was 65.2 (SD 21.8), and the mean discomfort level after sleeping with discs in place fell to 27.6 (SD 17.6). Comparison of pre/post means for oral wetness and discomfort was significant for both factors: wetness (p < 0.001) and discomfort (p < 0.017). Perceived morning oral wetness scores increased more than threefold. No adverse reactions were observed.
Independent Survey of Dentists — Based on a questionnaire about the effectiveness of OTC products in managing oral dryness, a 2016 article in Clinicians Report15 summarized responses from 1168 dentists regarding patients with xerostomia. Among the xerostomia remedies known to the dentists in the survey, and based on their assessment of patient experience, OraCoat XyliMelts discs were rated “very effective” by more dentists (9.2% of the 152 who were familiar with it) than any other xerostomia remedy, including prescription drugs, which came in second at 6%.
XyliMelts Single Subject Case Study — In a case study by Hoeg,16 an elderly female patient used two OraCoat XyliMelts discs nightly for 12 months. Disc use was associated with a reduction in perceived oral dryness and discomfort. A clinical examination revealed moist/wet intraoral tissue at the 12-month follow-up that was absent during the initial examination. In addition, the subject’s gingiva appeared healthy (characterized as having less gingival inflammation) at the 12-month follow-up. Periodontal pocket depths had not changed over the study period. At the initial examination (prior to disc use), several cervical caries lesions were present, but, at the one-year recall, no new lesions were noted.
XyliMelts Adhering Discs Versus Baseline of No Treatment While Sleeping — Van der Ven et al17 reported the results of a study involving the use of OraCoat XyliMelts to assess the possible effects of salivary stimulation during sleep on symptoms associated with nighttime gastroesophageal reflux disease (GERD), including reflux and heartburn.
This study was designed as a randomized, placebo-controlled and blinded trial involving oral adhering discs and an OTC water-based gel product for xerostomia. Prior to entering the study, each of the subjects reported GERD symptoms, as well as xerostomia while sleeping. Upon acceptance, subjects were asked to report if they perceived dry mouth while sleeping during 14 nights without intervention (baseline), and for an additional 14 nights during use of the discs or gel. A full explanation of the study methodology has previously been published.17
In respect to GERD, the discs, placed orally before sleep, were found to reduce the perception of reflux and morning hoarseness, as well as nighttime antacid use. They were also found to be more effective than the gel preparation in reducing heartburn (p < 0.001). Unpublished data on xerostomia demonstrate a reduction in the perception of dry mouth with both products tested. Compared to the baseline, disc use resulted in an average effect of 4.4 fewer nights of self-reported symptoms during two weeks of daily assessment (p < 0.001). The gel also demonstrated a significant reduction in the number of nights with dry mouth (p < 0.001). A comparative test between the gel and disc was not statistically significant.
DISCUSSION
The literature suggests that up to 40% of the general population experiences xerostomia with aging.18 Oral dryness can be caused by diseases such as diabetes, as well as autoimmune conditions, such as Sjόgren’s syndrome, or the use of prescription and nonprescription drugs19,20 and other medical problems, including seasonal allergies. In addition, mouth breathing while sleeping or use of a continuous positive airway pressure device for obstructive sleep apnea can produce symptoms of xerostomia.
As noted at the outset, oral dryness may be associated with oral burning or tingling, sore throat, and tongue, gum or denture ridge soreness.1 It can also result in taste dysfunction, speech problems, a sticky feeling in the mouth or lips, and difficulty swallowing.2 Xerostomia has been associated with an increased incidence of oral ulceration, infection (candidiasis), caries and periodontal disease. Other issues associated with oral dryness include halitosis, insomnia, irritability, depression, lip cracking, and speech and eating disorders.15 Research clearly shows that moderate to severe xerostomia can impact quality of life.21–23
For severe dry mouth associated with diseases such as Sjӧgren’s24 or occurring as a side effect of radiation of the salivary glands, medications — such as pilocarpine and cevimeline — are often prescribed.10 These drugs directly stimulate salivary function (when it is available).25 Although these medications can be effective in reducing xerostomia, their use may be associated with side effects on other tissues bearing muscarinic receptors. Both of these drugs can cause sweating, dizziness, urinary frequency, and, in some cases, hypersensitivity,26 and both are contraindicated in patients with uncontrolled asthma, narrow angle glaucoma, and certain cardiovascular and pulmonary diseases.27 As a result, perceived oral dryness associated with residual saliva production is not typically managed with these medications.
In individuals with xerostomia caused by medication use, substituting an alternative drug may reduce symptoms.24 That said, a change in medication(s) might not be possible or practical. The reviewed research suggests that when changing medications is not feasible, oral adhering discs could offer a useful strategy for increasing salivary production and oral lubrication.
CONCLUSION
The studies in this review indicate the mechanisms for the discs’ stimulating effect on salivary production are likely explained by the gradual release of flavor and the gel-forming hydrocolloid lubricant contained in each product. Further, the published research suggests adhering discs can be used safely while awake or sleeping.
KEY TAKEAWAYS
- When xerostomia symptoms occur during the day, they can be managed with various solutions, gels, sprays and rinses.3–10
- By comparison, there are few management options for patients who experience nighttime oral dryness, when prescribed saliva-stimulating drugs may not be suitable. In these cases, a lubricating oral adhering disc may be a viable option.
- The mechanisms for the discs’ stimulating effect on salivary production are likely explained by the gradual release of flavor and the gel-forming hydrocolloid lubricant contained in each product.
- Research shows that when discs are used while sleeping, subjects report an improvement in oral wetness upon waking.13
- In addition, published studies suggest oral adhering discs can be used safely while awake or sleeping.
REFERENCES
- Al-Dwairi Z, Lynch E: Xerostomia in complete denture wearers: prevalence, clinical findings and impact on oral functions. Gerodontology. 2014;31:49–55.
- Kaplan I, Zuk-Paz L, Wolff A. Association between salivary flow rates, oral symptoms and oral mucosal status. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2008;106:235–241.
- Dost F, Farah CS. Stimulating the discussion on saliva substitutes: a clinical perspective. Aust Dent J. 2013;58:11–17
- Furness S, Worthington HV, Bryan G, Birchenough S, McMillan R. Interventions for the management of dry mouth: topical therapies. Cochrane Database Syst Rev. 2011;12:CD008934.
- Gil‐Montoya JA, Guardia‐Lopez I, Gonzalez‐Moles MA. Evaluation of the clinical efficacy of a mouthwash and oral gel containing the antimicrobial proteins lactoperoxidase, lysozyme and lactoferrin in elderly patients with dry mouth — a pilot study. Gerodontology. 2008;25:3–9.
- Ship JA, McCutcheon JA, Spivakovsky S, Kerr AR. Safety and effectiveness of topical dry mouth products containing olive oil, betaine, and xylitol in reducing xerostomia for polypharmacy‐induced dry mouth. J Oral Rehabil. 2007;34:724–732.
- Oh DJ, Lee JY, Kim YK, Kho HS. Effects of carboxymethylcellulose (CMC) based artificial saliva in patients with xerostomia. Int J Oral Maxillofac Surg. 2008;37:1027–1031.
- Hahnel S, Behr M, Handel G, Bürgers R. Saliva substitutes for the treatment of radiation‐induced xerostomia — a review. Support Care Cancer. 2009;17:1331–1343.
- Aliko A, Alushi A, Tafaj A, Isufi R. Evaluation of the clinical efficacy of Biotène Oral Balance in patients with secondary Sjögren’s syndrome: a pilot study. Rheumatol Int. 2012;32:2877–2881.
- Mercadante V, Al Hamad A, Lodi G, Porter S, Fedele S. Interventions for the management of radiotherapy-induced xerostomia and hyposalivation: a systematic review and meta-analysis. Oral Oncol. 2017; 66:64–74.
- Aframian DJ, Mizrahi B, Granot I, Domb AJ. Evaluation of a mucoadhesive lipid-based bioerodable tablet compared with Biotène mouthwash for dry mouth relief-a pilot study. Quintessence Int. 2010;41:e36–e42.
- Kerr AR, Corby PM, Shah SS, Epler M, Fisch GS, Norman RG. Use of a mucoadhesive disk for relief of dry mouth: a randomized, double-masked, controlled crossover study. J Am Dent Assoc. 2010;141:1250–1256.
- Ho J, Firmalino MV, Anbarani AG, Takesh T, Epstein J, Wilder-Smith P. Effects of a novel disc formulation on dry mouth symptoms and enamel remineralization in patients with hyposalivation: an in vivo study. Dentistry (Sunnyvale). 2017:7–2.
- Burgess J, Lee P. XyliMelts time-release adhering discs for night-time oral dryness. Int J Dent Hyg. 2012;10:118–121.
- March 2016 Clinicians Report; additional data reported in Burgess J, OTC Management of Dry Mouth; Dentalproductsreport.com; July 2016.
- Hoeg C, Burgess J. Case study: Reducing caries and improving gingival status while sleeping. Available at: http://www.dentalproductsreport. com/dental/article/case-study-reducing-caries-and-improving-gingival-status-while-sleeping. Accessed May 30, 2020.
- Burgess JA, Van Der Ven P, Karcher M. Effect on acid reflux symptoms occurring during sleep of an adhering disk containing only food ingredients. J Gastrointest Dig Syst. 2017;7:4–7.
- Gupta A, Epstein JB, Sroussi H. Hyposalivation in elderly patients. J Can Dent Assoc. 2006;72:841–846.
- Ouanounou A. Xerostomia in the geriatric patient: causes, oral manifestations, and treatment. Compend Contin Educ Dent. 2016;37:306–311.
- Cassolato SF, Turnbull RS. Xerostomia: clinical aspects and treatment. Gerodontology. 2003;20:64–77.
- Gallardo JM. Xerostomia: etiology, diagnosis and treatment. Rev Med Inst Mex Seguro Soc. 2008; 46:109–116.
- Goldie MP. Xerostomia and quality of life. Int J Dent Hygiene.2007;5:60–61.
- Title of measure: The University of Michigan Xerostomia-Related Quality of Life Scale (XeQOLS). Available at: https://www.rtog.org/LinkClick.aspx? fileticket=ahsKgJEDLnA%3D&tabid=118. Assessed May 30, 2020.
- Leung KC, McMillan AS, Wong MC, Leung WK, Mok MY, Lau CS. The efficacy of cevimeline hydrochloride in the treatment of xerostomia in Sjogren’s syndrome in southern Chinese patients: a randomized double-blind, placebo-controlled crossover study. Clin Rheumatol. 2008;27:429–436.
- LinA, Kim HM, Terrell JE, Dawson LA, Ship JA, Eisbruch A. Quality of life after parotid-sparing IMRT for head and neck cancer: a prospective longitudinal study. Int J Radiat Oncol Biol Phys. 2003;57:61–70.
- Wolff A, Fox PC, Porter S, Konttinen YT. Established and novel approaches for the management of hyposalivation and xerostomia. Curr Pharm Des. 2012;18:5515–5521.
- LeVeque FG, Montgomery M, Potter D, et al. A multicenter, randomized, double-blind, placebo-controlled, dose-titration study of oral pilocarpine for treatment of radiation-induced xerostomia in head and neck cancer patients. J Clin Oncol. 1993;11:1124–1131.
The authors report having no commercial conflicts of interest to disclose.
From Decisions in Dentistry. June 2020;6(6):16–18,21.


