
Use of the Dental Videoscope in Endodontics
More closely associated with periodontal procedures, this imaging modality holds promise for surgical root canal therapy.
In clinical dentistry, the practitioner is routinely faced with the decision as to whether a tooth is salvageable with a reasonable investment of time and expense for the patient. One of the most common endodontic presentations is a tooth with irreversible pulpal pathosis or pulpal necrosis. This will frequently present with the patient in pain and seeking relief. The immediate treatment options are extraction or endodontic therapy. With the advent and growing popularity of dental implants, extraction is too often the choice at this initial acute stage. There is ample evidence that retention of restorable natural teeth may be the more favorable and sustainable option, long term.1 Thus, a tooth with irreversible pulpal pathosis must be carefully evaluated for potential endodontic therapy and restorability. Clinical considerations include the probable long-term outcome, which should be explained to the patient.
Technology that provides magnification and illumination will greatly improve visualization during endodontic treatment. While the surgical operating microscope is commonly used in these procedures and is considered the standard of care, other imaging modalities have been used for endodontic treatment; Bahcall et al,2 for example, documented the use of the endoscope for endodontic surgery. This article will explore use of the dental videoscope to enhance visualization, as this technology uses a micro video camera and monitor to provide a real-time, color view of the operating site.
When the decision is made to retain a tooth, endodontic therapy should be initiated expeditiously to relieve pain. Nonsurgical root canal therapy is a highly successful means for the treatment and prevention of apical periodontitis, with a success rate of approximately 92% to 98% when there is no evidence of radiographic apical bone loss, and 74% to 86% when such pathosis is present.3 In cases in which previous endodontic treatment has not been satisfactory, nonsurgical retreatment may be the best treatment for the nonhealing periapical inflammatory lesion. There are also times where a surgical approach would be more prudent. Glickman and Hartwell4 outlined three main reasons clinicians may opt to treat a case surgically:
- Failure of nonsurgical retreatment (treatment has been rendered at least two times)
- Failure of nonsurgical initial treatment, and retreatment is not possible, practical or likely to yield a better result. This could include anatomic considerations, such as extreme apical curvatures, iatrogenic considerations that resulted from operator difficulty in nonsurgical treatment (such as transportation of the anatomical foramen, ledging or broken (separated) instruments in the apical third), or practical reasons, such as the presence of a large post with a well-adapted, intact crown
- When a biopsy is necessary
The success rates of surgical root canal therapy using a modern technique is highly predictable, with a reported success rate of 94%.5
PRESURGICAL PLANNING
Surgical root canal therapy is a complex and detailed procedure. The starting point should be meticulous presurgical planning. The American Association of Endodontists recommends cone beam computed tomography as the imaging modality of choice for presurgical planning.6 Special attention should be given to the anatomical relationship of the periapical region to areas of anatomic vulnerability, such as the mandibular nerve, mental foramen, maxillary sinus, and any other areas where apical surgery might incur high risk to the patient. If there are any significant anatomical factors that may negatively influence long-term treatment success, the advisability of extraction should again be evaluated.
Once the decision is made to perform surgical endodontic treatment, clinicians must prepare for anesthesia, patient preparation and site assessment. Traditional local anesthetic techniques can be used, such as mandibular blocks or local infiltration. In addition, clinicians may consider using an anesthetic solution containing 1:50,000 epinephrine directly over the projected surgical site for increased hemostasis.7 A full-thickness flap is then completed using a rectangular or triangular design. When adequate attached gingiva is present, a submarginal incision (i.e., a full-thickness incision that follows the contour of the gingival margin and remains in the attached gingiva) may also be employed, as was used in the case report described in this paper. This was done to prevent recession and “black triangle” formation between esthetic anterior crowns. After adequate reflection, an osteotomy is made with a large, surgical-length round bur with copious saline spray to gain access to the periapical area or surgical site. In some instances, a radiograph will need to be exposed with a radiopaque marker on the alveolar bone at the location projected to be the root end.
Once the apex of the root has been determined, an approximate 3 mm section of root is removed. This resection should be as perpendicular as possible to the long axis of tooth. A slight bevel toward the facial may be required for adequate visualization. The 3 mm resection is recommended, as most lateral and accessory canals occur in the apical 3 mm.8 Any granulomatous or cystic tissue is removed with a curette, either before or after resection of the root, and sent to a pathologist for diagnosis.
Methylene blue is often used at this point to assess the root for fractures or other irregularities, and to ensure a full resection has been completed. If there is vertical root fracture, this can be readily identified as a crack extending from the canal space to the surface of the root. The methylene blue will stain the periodontal ligament and should be evident on all sides of the resected root surface. A microsurgical ultrasonic insert or tip is then used to prepare a 3-mm-deep preparation in the resected root end, removing the gutta-percha (or other filling material) placed during previous nonsurgical endodontic therapy. The root end preparation should be in the long axis of the original canal space and will be the width of the microsurgical tip, typically from 0.5 to 1 mm in diameter. The root end preparation is dried with trimmed paper points before a filling material is placed. A tricalcium silicate-based material is recommended. The excess root end filling material is gently removed with a saline hydrated microbrush. Prior to flap reapproximation, the site is rinsed with saline, the osteotomy is refreshed by curettage of the bony walls to induce bleeding, and, after several minutes of pressure on the lip or cheek (with the flap resting in place), the site is closed using the operator’s suturing technique of choice. Interrupted 6-0 absorbable sutures were used in the illustrated case.
ENHANCED VISUALIZATION
Since the advent of the surgical microscope, the success of apical surgery has greatly improved.5 Extreme precision is required in root end surgery, and without sufficient visualization the clinician lacks the proper instruments to perform the procedure at this precise level. In root end resections, the operator must resect the entire root tip, leaving no unresected root extensions on the palatal/lingual surface. This can be difficult to visualize, even with the surgical operating microscope. Visualization within the canal system when starting the root end preparation would benefit from enhanced imaging technology. Determining whether or not all of the gutta-percha has been removed from the internal surface of the walls can also be problematic. In these instances, use of the dental videoscope can provide visualization that helps minimize or eliminate uncertainty.
Developed for use in videoscope-assisted minimally invasive periodontal surgery, the dental videoscope features a micro video camera that is small enough to be inserted into the surgical area.9 A close-up image of the surgical site is displayed on a monitor. Although studies have investigated use of the videoscope in periodontal surgery,10,11 to date, its use for endodontic surgery has not been described. The need for high magnification with bright illumination and the necessity of visualizing narrow bony defects is similar in both periodontal and endodontic surgery. The following case report illustrates endodontic surgery performed with the assistance of the dental videoscope.
CASE REPORT
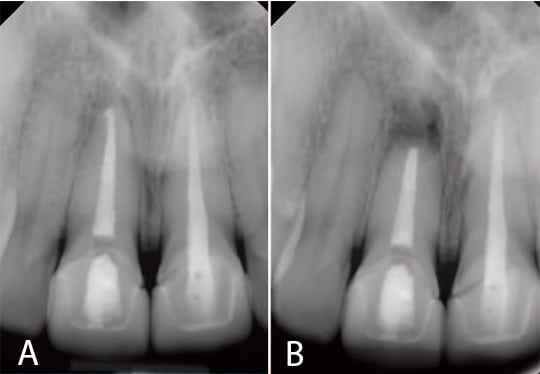
A 30-year-old female presented with a 4 mm by 4 mm localized buccal swelling in the apical region of tooth #8 that could be felt during palpation. The patient had a traumatic injury six years earlier, which, at the time, required root canal therapy. Nonsurgical retreatment was required and completed six years after the initial injury (radiographs of the completed retreatment can be seen in Figures 1A and 1B). The patient returned two months later with a chief complaint of pain when she touched the root apex area of #8. Upon examination, the localized swelling was still present and the decision was made to perform apical surgery.
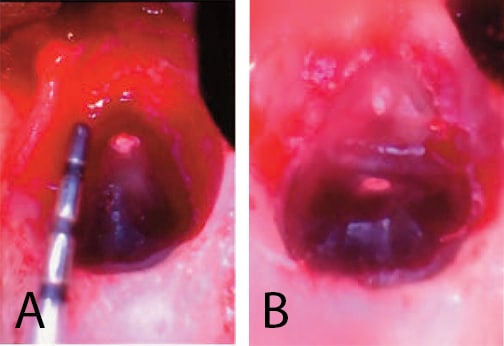
A submarginal incision was elected secondary to esthetic concerns of #8 and #9. This type of incision diminishes the occurrence for loss of gingival height. The incision was scalloped to mirror the gingival contour and confined to the attached gingiva. A vertical release was placed distal to #7 so the incision and closure would be on intact bone, remote from the osteotomy. The incision was extended to the mesial of #10 so there could be adequate visualization during reflection. The flap was reflected and a bony fenestration of the facial cortical plate was seen. The granulation tissue was removed with a curette and sent for biopsy, and the root end was exposed. A #6 round bur was used to enlarge the osteotomy. A periodontal probe was used to measure the appropriate amount of resection, as shown in Figure 2A. A Lindemann burr was used to make the resection, but, as shown in Figure 2B, the resection was only partially through the root.
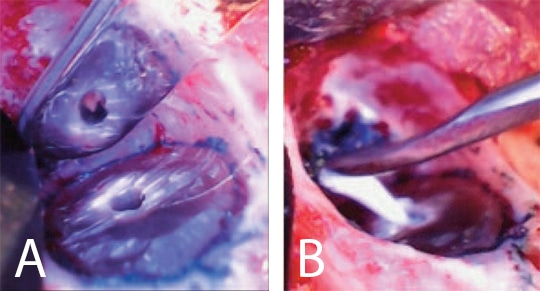
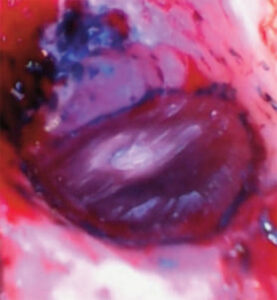
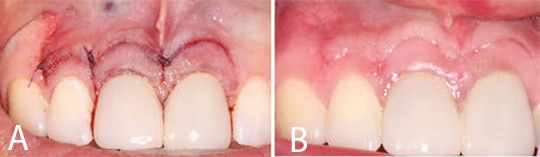
After the resection was completed, a root-end-preparation was made with a microsurgical ultrasonic insert/tip, and methylene blue was used to stain the periodontal ligament to aid in the detection of possible cracks (Figure 3A). The root end preparation was dried with trimmed paper points and an endodontic sealer was injected into the root end preparation. A root end filling material was carefully rolled into a small cone and placed in the prepared space (Figure 3B). The surgical site was rinsed with sterile saline and the root end surface finished with a microbrush (Figure 4). The flap was closed using three interrupted 6-0 absorbable sutures and cyanoacrylate tissue adhesive (Figure 5A). The patient was released and scheduled to return in five days for suture removal. At the follow-up, the patient reported mild discomfort, and soft tissue healing appeared to be progressing well (Figure 5B).
Conclusion
Apical surgery is a difficult but rewarding procedure that allows patients to retain their natural dentition. With proper technique, results are typically predictable and success rates are high. However, adequate visualization is vital to clinical proficiency. In this regard, the dental videoscope may prove helpful by providing magnification and illumination of the surgical site. By reducing uncertainty, this technology shows potential to help improve outcomes in apical surgery, although research is needed to validate use of this enhanced imaging modality in periapical surgery.
KEY TAKEAWAYS
- One of the most common endodontic presentations is a tooth with irreversible pulpal pathosis or pulpal necrosis. While the immediate treatment options are extraction or endodontic therapy, there is ample evidence that retention of restorable natural teeth may be the more favorable and sustainable long-term option.
- Consequently, a tooth with irreversible pulpal pathosis must be carefully evaluated for potential endodontic therapy and restorability; this includes assessing the probable long-term outcome.
- Although nonsurgical root canal therapy is a highly successful means for treating and preventing apical periodontitis, there are also times when a surgical approach is more prudent.4
- Technology that provides magnification and illumination will greatly improve visualization during endodontic treatment.
- Since the advent of the surgical operating microscope, surgical root canal therapy using a modern technique is highly predictable, with a reported success rate of 94%.5
- Extreme precision is required in root end surgery, and without sufficient visualization the clinician lacks the proper instruments to perform the procedure at this precise level.
- Designed for enhanced visualization, the dental videoscope uses a micro video camera that is small enough to be inserted into the surgical area;9 this technology has potential to help improve outcomes in endodontic surgery.
- In apical surgery, for example, determining whether or not all of the gutta-percha has been removed from the internal surface of canal walls can be problematic. In these instances, use of the dental videoscope can provide visualization that helps minimize or eliminate uncertainty.
REFERENCES
- Setzer FC, Kim S. Comparison of long-term survival of implants and endodontically treated teeth. J Dent Res. 2014;93:19–26
- Bahcall JK, DiFiore PM, Poulakidas TK. An endoscopic technique for endodontic surgery. J Endod. 1999;25:132–135.
- Friedman S, Mor C. The success of endodontic therapy: healing and functionality. J Calif Dent Assoc. 2004;32:493–503.
- Glickman GN, Hartwell GR. Endodontic surgery. In: Ingle JI, Bakland LK, Baumgartner JC. Ingle’s Endodontics 6. 6th ed. Hamilton, Ontario: BC Decker Inc; 2008:1233–1294.
- Setzer FC, Shah SB, Kohli MR, Karabucak B, Kim S. Outcome of endodontic surgery: a meta-analysis of the literature–part 1: comparison of traditional root-end surgery and endodontic microsurgery. J Endod. 2010;36:1757–1765.
- American Association of Endodontists, American Academy of Oral and Maxillofacial Radiology. Position statement: Use of Cone Beam Computed Tomography in Endodontics 2015 Update. Available at: aae.org/uploadedfiles/clinical_resources/guidelines_and_position_statements/cbctstatement_2015update.pdf. Accessed March 14, 2017.
- Buckley JA, Ciancio SG, McMullen JA. Efficacy of epinephrine concentration in local anesthesia during periodontal surgery. J Periodontol. 1984;55:653–657.
- De Deus QD. Frequency, location, and direction of the lateral, secondary, and accessory canals. J Endod. 1975;1:361–366.
- Harrel SK, Wilson TG Jr, Rivera-Hidalgo F. A videoscope for use in minimally invasive periodontal surgery. J Clin Periodontol. 2013;40:868–874.
- Harrel SK, Abraham CM, Rivera-Hidalgo F, Shulman JD, Nunn ME. Videoscope-assisted minimally invasive periodontal surgery (V-MIS). J Clin Periodontol. 2014;41:900–907.
- Harrel SK, Abraham CM, Rivera-Hidalgo F, Shulman JD, Nunn ME. Videoscope–assisted minimally invasive periodontal surgery: one year outcome and patient. morbidity. Int J Periodontics Restorative Dent. 2016;36:363–371.
Cole W. Barnett, DDS, has no commercial conflicts of interest to disclose.
Stephen K. Harrel, DDS, holds the patent on the dental videoscope.
Featured photo by zlikovec / istock / Getty Images Plus
From Decisions in Dentistry. April 2017;3(4):16,18,21–22.


