
Nonsurgical Treatment of Chronic Periodontitis
This seven-step protocol is designed to mitigate risk factors and address existing periodontal inflammation.
Chronic periodontitis results from the continuous and progressive interaction between a subgingival pathogenic microbial biofilm and the host immune system. Such interactions lead to inflammation and resorption of the supporting bone of the teeth. Depending on disease severity and the architecture of soft tissue destruction and bone loss, treatment may be nonsurgical or surgical. Nonsurgical therapy is considered the gold standard for the initial treatment of inflammatory periodontal disease. Indeed, nonsurgical treatment involving meticulous scaling and root planing (SRP) has been extensively documented and has been shown to be a highly predictable and successful therapy.1
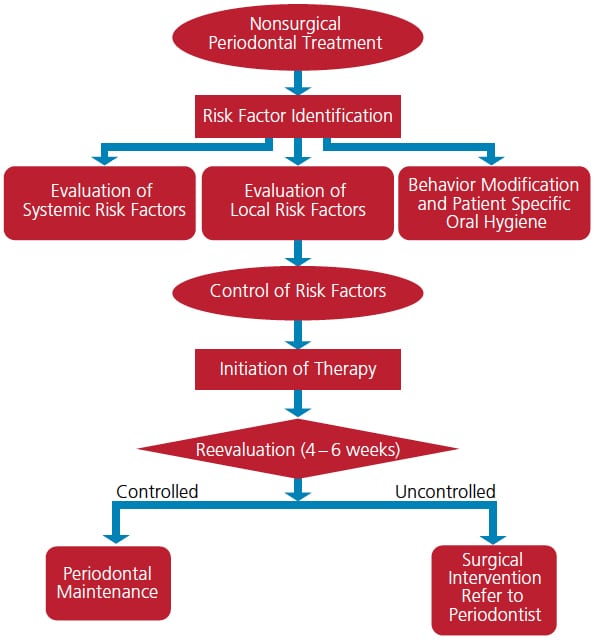
The main goals of nonsurgical periodontal therapy (NSPT) are to remove and control supra- and subgingival microbial biofilms, eliminate and control inflammation, and eliminate and control systemic and local risk factors that contribute to disease development and progression. Depending on the severity and degree of bony involvement, effective NSPT may minimize or eliminate the need for surgical intervention. Hence, the seven-step protocol described here (Figure 1, page 13) will enable clinicians to identify and modify local and systemic risk factors, facilitate decisions about the use of systemic or locally delivered antibiotics, and prescribe appropriate oral hygiene measures. It must be emphasized that meticulous SRP is inherent to any successful nonsurgical protocol. Furthermore, not all periodontal disease can be successfully managed with a nonsurgical approach; this is particularly true of advanced disease with inherent osseous defects and irregular bony architecture.
The seven-step protocol for NSPT includes:
- Eliminating or controlling systemic risk factors
- Eliminating or controlling local risk factors
- Behavior modification with respect to oral hygiene
- Meticulous SRP
- Systemic antibiotics, locally delivered antimicrobials, and/or antiseptic mouthrinses
- Reevaluation of therapy and decisions regarding future treatment
- Establishing an appropriate periodontal maintenance interval
STEP 1. CONTROLLING SYSTEMIC RISK FACTORS
Several risk factors have well established associations with both periodontal and systemic diseases, such as diabetes, smoking, stress, immunodeficiency, medications, obesity, hormones and nutrition. For optimal treatment results, systemic risk factors must be modified or eliminated. Some are under the patient’s control (including smoking, stress, obesity and nutrition), and may, therefore, require lifestyle changes to achieve significant modification. (Obviously, this list is not all-inclusive, but instead represents the more common risk factors.) Other more subtle systemic and environmental issues may also need to be addressed to achieve optimal results.
Diabetes: A bidirectional relationship between diabetes and periodontitis was proposed by Löe2 and has been confirmed repeatedly by other investigators.3 According to a 2011 report from the U.S. Centers for Disease Control (CDC), nearly 8.3% of the U.S. population has diabetes. For a patient with diabetes or a patient suspected of prediabetes, it is imperative to know the individual’s glycosylated hemoglobin level (HbA1c) prior to initiating periodontal treatment. Patients with diabetes and HbA1c values less than 7% are considered well controlled and can be treated in similar fashion to the general population; however, even in these patients, the possibility of delayed healing and infection should be discussed prior to treatment. Patients with diabetes that is poorly controlled should be referred to a medical provider for better metabolic control prior to initiating periodontal therapy. Mutual management of both chronic diseases is vital to attaining stable periodontal health, as well as optimal metabolic control.
 Smoking: As documented in the literature, is estimated that smokers have a twofold to fourfold greater risk of developing periodontitis.4 In clinical studies, patients are generally stratified based on the number of cigarettes smoked per day; for example, light (< 10 cigarettes/day), moderate (10 to 19 cigarettes/day), or heavy smokers (≥ 20 cigarettes/day). The effect of smoking on periodontal health is dose dependent; thus, the risk of developing periodontitis is positively correlated to the number of cigarettes smoked. Prior to initiating nonsurgical periodontal therapy, patients who smoke should be educated about the effects of smoking and encouraged to quit — or at least become “light smokers.” Studies have reported that smokers exhibit less reduction in subgingival microbial loads and probing depths (PD) during NSPT than nonsmoking patients.5,6
Smoking: As documented in the literature, is estimated that smokers have a twofold to fourfold greater risk of developing periodontitis.4 In clinical studies, patients are generally stratified based on the number of cigarettes smoked per day; for example, light (< 10 cigarettes/day), moderate (10 to 19 cigarettes/day), or heavy smokers (≥ 20 cigarettes/day). The effect of smoking on periodontal health is dose dependent; thus, the risk of developing periodontitis is positively correlated to the number of cigarettes smoked. Prior to initiating nonsurgical periodontal therapy, patients who smoke should be educated about the effects of smoking and encouraged to quit — or at least become “light smokers.” Studies have reported that smokers exhibit less reduction in subgingival microbial loads and probing depths (PD) during NSPT than nonsmoking patients.5,6
Medications: Roughly 6% to 15% of patients taking a calcium channel blocker, 25% to 30% receiving immunosuppressants, and 50% being treated with antiepileptic drugs will exhibit some degree of drug-induced gingival enlargement.7 Clinical symptoms of gingival overgrowth are generally seen one to three months after initiating drug therapy. For these patients, the medical provider should be consulted to explore the possibility of changing to a different drug category to minimize or eliminate gingival enlargement.8 In addition, thorough oral hygiene instruction and appropriate periodontal maintenance (PM) intervals need to be established for these patients.9
Obesity: The World Health Organization defines obesity as having a body mass index (BMI) ≥ 30 kg/m2, which is further defined based on body fat of > 30% in men and > 25% in women.10 Obesity and BMI are positively correlated to an increased incidence of periodontal disease. Further, it appears that obesity can adversely impact periodontal treatment outcomes.11 Gorman et al12 reported that as BMI increases by one unit, the rate of alveolar bone loss increases by 5%. Consequently, periodontal patients should be encouraged to adapt a healthy lifestyle to maintain an ideal BMI of < 25kg/m2. Calculation and recording of BMI needs to be incorporated in the comprehensive initial evaluation. This can be easily done using the CDC’s BMI calculator (cdc.gov/healthyweight/assessing/bmi/adult_bmi/english_bmi_calculator/bmi_calculator.html).
Nutrition: The role of nutrition is well documented in management of chronic systemic illnesses and should be extended to include periodontitis. A diet plan to prevent periodontal disease and aid healing following periodontal therapy should include high fiber, low carbohydrate,13 high protein,13 Omega-3 fatty acids, micronutrients (such as vitamins D, C and B12), and antioxidants (e.g., vitamin A, folic acid, and beta-carotene) to optimize periodontal health. Chapple et al14 demonstrated a statistically significant relation between the prevalence of periodontal disease and low serum vitamin C levels. Levine et al15 reported that 200 mg of vitamin C per day could help maintain periodontal health. Compared to a control group, in a study of patients in a PM program, subjects taking ≥ 1000 mg of calcium and ≥ 400 IU of vitamin D had more shallow PDs and less gingival inflammation.16
Merchant et al17 noted that higher intake of whole grains is associated with a 23% decreased risk for developing periodontal disease. Whole grains have a high fiber content that, in turn, may help decrease inflammation and improve host insulin sensitivity. Carbohydrates have the opposite effect in that high intake of carbohydrates is associated with increased gingival inflammation.18 According to Al-Zahrani et al,19 individuals with a normal BMI, habit of good exercise, and a healthy diet are 40% less likely to develop periodontitis than at-risk groups.
STEP 2. ELIMINATING LOCAL RISK FACTORS
A variety of tooth-related local risk factors can predispose a patient to developing an inflammatory periodontal lesion. These can be grouped into three general categories: anatomical, tooth position and iatrogenic. While local risk factors are not considered to be a direct cause of periodontitis, they contribute to biofilm development, increased biofilm pathogenicity and calculus retention. Local risk factors can be modified and/or eliminated — and, in fact, periodontal therapy should not be considered complete until all local risk factors have been addressed.
Anatomical risk factors may include cervical enamel projections and enamel pearls, furcations, root grooves and flutings, untreated caries, frenulum attachments and gingival recession. Risk factors involving tooth position are likely to include malposition, crowding, tipping and migration of teeth. Abnormal occlusal forces resulting from traumatic occlusal relationships and bruxism and clenching habits also fall into this category. Lastly, iatrogenic risk factors might involve an ill-fitting removable prostheses, temporary and permanent restorations with rough and inadequate contours, overhangs, open margins and open contacts.20–22
STEP 3. BEHAVIOR MODIFICATION
Effective plaque control is an absolute requirement in nonsurgical and surgical periodontal therapy. Assessment, motivation and reinforcement of oral hygiene should be performed at each appointment. A recent systematic review and meta-analysis evaluating the association between oral hygiene and periodontitis showed that the risk of periodontitis increased by twofold to fivefold in patients with poor oral hygiene.23 Self-care instructions need to be personalized and should be recommended based on the severity of the patient’s periodontal status.
As noted in Table 1, for healthy patients or those with gingivitis, effective brushing, tongue cleaning, flossing and use of an antiseptic mouthrinse should be sufficient. Interestingly, given the prodigious bacterial population of the dorsal tongue surface, daily cleaning of the tongue has received little attention in the dental literature. Matsui et al24 recommended the practice of toothbrushing and tongue cleaning to effectively decrease the oral microbial load. Tongue scraping reportedly produces a statistically significant reduction in halitosis, as compared to mechanical brushing of the teeth.25 Patients with plaque-induced gingivitis should be counseled that good oral hygiene can restore gingival health without significant periodontal treatment. If plaque is not controlled, however, the inflammatory lesion may progress to a more severe stage of periodontal disease. Adjunctive use of a chlorhexidine oral rinse can be beneficial, as research has shown it contributes to an approximate 33% reduction in plaque and 26% reduction in gingivitis.26
Periodontitis is characterized by gingival inflammation and loss of alveolar bone. In moderate to severe stages, gingival recession, loss of interproximal papilla, tooth mobility and furcation involvement may also be clinical features. Referring to Table 1, for patients with moderate to severe disease, toothbrushing and flossing alone are inadequate to control biofilm. When compared to flossing, interdental brushes have been shown to decrease gingival inflammation by 52% and to be more effective in controlling interproximal plaque accumulation in patients with periodontitis.27 In patients with moderate or severe periodontitis, flossing is less effective due to loss of papillary guidance. Oral irrigators are reported to be effective in reducing gingival inflammation,28 yet it should be noted that the adjunctive benefits of using an oral irrigator remain controversial, as the peer-reviewed research offers conflicting results.
STEP 4. SCALING AND ROOT PLANING
As previously noted, meticulous SRP is the most critical component of initial periodontal therapy. The primary goals of SRP are to significantly reduce or eliminate subgingival microbial loads, remove subgingival calculus, remove soft and diseased cementum, and smooth roughened root surfaces.1,29 A systematic review by Cobb1 reported that average improvement in PD following SRP was 1.29 mm in pockets that had an initial PD of 4 to 6 mm, and 2.16 mm in pockets of ≥ 7 mm. Gains in clinical attachment levels (CAL) averaged 0.55 mm and 1.19 mm for initial PDs of 4 to 6 mm and ≥ 7 mm, respectively. In most forms of periodontitis, SRP is highly effective in controlling the signs of periodontal inflammation. However, surgical intervention may be required to definitively treat the bony defects often associated with the moderate and severe stages of periodontitis.
STEP 5. SYSTEMIC AND LOCALLY DELIVERED ANTIMICROBIALS
The adjunctive use of systemic antibiotics with SRP is known to yield a statistical difference, but arguably not a clinically significant difference. In a consensus report from the 6th European Workshop on Periodontology, Sanz and Teughels30 suggested the following guidelines regarding the use of systemic antibiotics in the treatment of periodontal disease:
- Systemic antibiotics are not necessary for most patients with periodontitis
- Systemic antibiotics may benefit specific patient groups (e.g., those with aggressive periodontitis or necrotizing ulcerative gingivitis) or defined conditions (e.g., severe and/or progressing periodontitis)
- If used as part of a treatment protocol for periodontitis, systemic antibiotics should be used in conjunction with SRP
- Indirect evidence suggests that for optimal clinical results, systemic antibiotics should be used at the time of SRP, and all treatment should be completed within seven days
- The use of systemic antibiotics should be restricted
This cautionary note regarding restricted use of systemic antibiotics is supported by a 2015 American Dental Association statement of clinical guidelines and results noting the increasing emergence of microbial antibiotic resistance.31
Reporting on the potential benefits of topical antiseptic agents (such as 10% povidone iodine) as an adjunct to mechanical debridement, Slots32 noted that it is important to maintain a tissue contact time of five to seven minutes to obtain the optimal antimicrobial effect. Reported benefits from subgingival iodine application include significantly greater reductions in PD and putative periodontal pathogen counts, as well as gains in CAL.33,34
Studies that have evaluated the use of locally delivered antimicrobials — such as chlorhexidine chips, doxycycline hyclate gel and minocycline spheres — report modest clinical benefits. Whether the benefit is clinically significant depends chiefly on expert opinion and not statistical evidence.31,35 That noted, locally delivered antimicrobials have some use in medically compromised patients, such as individuals with brittle or marginally controlled diabetes, those undergoing kidney dialysis or active intravenous bisphosphonate therapy, and patients with mental disabilities that impair effective oral hygiene.
STEP 6. REEVALUATION
Clinicians are advised that reevaluation is not the end-point of active periodontal therapy — it is part of a decision matrix. Periodontal status should be thoroughly reassessed at four to six weeks following the nonsurgical phase of treatment, as the greater part of healing is completed by six weeks, but collagen maturation might continue for up to nine months.35 Post-therapy reevaluation is a critical step in determining the status of gingival inflammation, oral hygiene and healing. Ideal end points for nonsurgical therapy include a decreasing plaque index ≤ 10%, bleeding on probing (BOP) ≤ 20%, PDs ≤ 4 mm, mean gain and stability of CAL, maintenance of alveolar bone height, and preservation of form and function of dentition.36
Reevaluation provides an opportunity to determine if the patient’s periodontal health is stable enough for assignment to a PM program, or whether further therapy is required. Further treatment may involve reinforcement of oral hygiene techniques, nonsurgical retreatment of teeth with no intrabony defects, or surgical intervention.
STEP 7. PERIODONTAL MAINTENANCE
Achieving long-term periodontal stability requires an effective PM program and patient compliance. Maintenance generally includes a reevaluation to determine the patient’s periodontal status, an evaluation of systemic and local risk factors, supragingival scaling and polishing, SRP of residual PDs — particularly those that exhibit BOP. The interval between PM appointments should be customized for each patient, based on local and systemic risk factors, severity and extent of periodontal disease, rate of disease progression, and patient compliance. The standard of care for patients who have received definitive treatment for chronic or aggressive periodontitis is a three-month PM interval; however, stable patients can be appointed for longer intervals.
CONCLUSION
A basic NSPT protocol should consist of an evaluation of local and systemic risk factors, modification or elimination of risk factors (to the extent possible), patient-specific oral hygiene techniques, mechanical debridement and SRP (with or without antimicrobial therapy). Following definitive therapy, reevaluation of treatment response, determination of a patient-specific PM interval, and gaining patient compliance are necessary to maintain periodontal health. Even with optimal care, however, up to 10% of patients may not exhibit the expected response to nonsurgical therapy, and may thus require more aggressive treatment.
KEY TAKEAWAYS
- Nonsurgical treatment involving meticulous scaling and root planing has been shown to be a highly predictable and successful periodontal therapy.1
- The main goals of nonsurgical periodontal treatment are to remove and control supra- and subgingival microbial biofilms, eliminate and control inflammation, and eliminate and control systemic and local risk factors.
- Self-care instructions need to be personalized and should be recommended based on the severity of the patient’s periodontal status.
- Periodontal status should be thoroughly reassessed at four to six weeks following the nonsurgical phase of treatment.
- Reevaluation provides an opportunity to determine if the patient’s periodontal health is stable enough for assignment to a periodontal maintenance program, or whether further therapy is required.
Acknowledgement
Bharathi Devi Myneni, BDS, thanks Chief Executive Officer Eugene C. Skourtes, DMD, and Kristen Simmons, RDH, MBA, of Willamette Dental Group for their support in the development of this manuscript.
References
- Cobb CM. Nonsurgical periodontal therapy: mechanical. Annals of Periodontol. 1996;1:443–490.
- Löe H. Periodontal disease. The sixth complication of diabetes mellitus. Diabetes Care. 1993;16: 329–334.
- Kumar M, Mishra L, Mohanty R, Nayak R. Diabetes and gum disease: the diabolic duo. Diabetes Metabo Syndr. 2014;8:255–258.
- Tomar SL, Asma S. Smoking-attributable periodontitis in the United States: findings from NHANES III. National Health and Nutrition Examination Survey. J Periodontol. 2000;71:743–751.
- Van der Velden U, Varoufaki A, Hutter JW, et al. Effect of smoking and periodontal treatment on the subgingival microflora. J Clin Periodontol. 2003;30:603–610.
- Garcia RI. Smokers have less reductions in probing depth than non-smokers following nonsurgical periodontal therapy. Evid Based Dent. 2005;6:37–38.
- Dongari-Bagtzoglou A, Research Science and Therapy Committee, American Academy of Periodontology. Drug-associated gingival enlargement. J Periodontol. 2004;75:1424–1431.
- Hernández G, Arriba L, Frías MC, et al. Conversion from cyclosporin a to tacrolimus as a nonsurgical alternative to reduce gingival enlargement: a preliminary case series. J Periodontol. 2003;74:1816–1823.
- Hall EE. Prevention and treatment considerations in patients with drug-induced gingival enlargement. Curr Opin Periodontol. 1997;4:59–63.
- Hein C, Batista EL Jr. Risk assessment for obesity and periodontal disease. Decisions in Dentistry. 2017;3(6):42–46.
- Suvan J, Petrie A, Moles DR, et al. Body mass index as a predictive factor of periodontal therapy outcomes. J Dent Res. 2014; 93:49–54.
- Gorman A, Kaye EK, Apovian C, Fung TT, Nunn M, Garcia RI. Overweight and obesity predict time to periodontal disease progression in men. J Clin Periodontol. 2012;39:107–114.
- Hujoel PP, Lingström P. Nutrition, dental caries and periodontal disease: a narrative review. J Clin Periodontol. 2017;44(Suppl 18):S79–S84.
- Chapple IL, Milward MR, Dietrich T. The prevalence of inflammatory periodontitis is negatively associated with serum antioxidant concentrations. J Nutr. 2007;137:657–664.
- Levine M, Wang Y, Katz A, et al. Ideal vitamin C intake. Biofactors. 2001;15:71–74.
- Miley DD, Garcia MN, Hildebolt CF, et al. Cross-sectional study of vitamin D and calcium supplementation effects on chronic periodontitis. J Periodontol. 2009;80:1433–1439.
- Merchant AT, Pitiphat W, Franz M, Joshipura KJ. Whole-grain and fiber intakes and periodontitis risk in men. Am J Clin Nutr. 2006;83:1395–1400.
- Woelber JP, Bremer K, Vach K, et al. An oral health optimized diet can reduce gingival and periodontal inflammation in humans–a randomized controlled pilot study. BMC Oral Health. 2016;16:109.
- Al-Zahrani MS, Borawski EA, Bissada NF. Periodontitis and three health-enhancing behaviors: maintaining normal weight, engaging in recommended level of exercise, and consuming a high-quality diet. J Periodontol. 2005;76:1362–1366.
- Schätzle M, Lang NP, Ånerud A, Boysen H, Bürgin W, Löe H. The influence of margins of restoration on the periodontal tissues over 26 years. J Clin Periodontol. 2001;28:57–64.
- Matthews DC, Tabesh M. Detection of localized tooth-related factors that predispose to periodontal infections. Periodontol 2000. 2004;34:136–150.
- Zlatarić DK, Celebić A, Valentić-Peruzović M. The effect of removable partial dentures on periodontal health of abutment and non-abutment teeth. J Periodontol. 2002;73:137–144.
- Lertpimonchai A, Rattanasiri S, Arj-Ong Vallibhakara S, Attia J, Thakkinstian A. The association between oral hygiene and periodontitis: a systematic review and meta-analysis. Int Dent J. 2017;67:332–343.
- Matsui M, Chosa N, Shimoyama Y, Minami K, Kimura S, Kishi M. Effects of tongue cleaning on bacterial flora in tongue coating and dental plaque: a crossover study. BMC Oral Health. 2014;14:4.
- Outhouse TL, Al-Alawi R, Fedorowicz Z, Keenan JV. Tongue scraping for treating halitosis. Cochrane Database Syst Rev. 2006:CD005519.
- Herrera D. Chlorhexidine mouthwash reduces plaque and gingivitis. Evid Based Dent. 2013;14:17–18.
- Poklepovic T, Worthington HV, Johnson TM, et al. Interdental brushing for the prevention and control of periodontal diseases and dental caries in adults. Cochrane Database Syst Rev. 2013:CD009857.
- Barnes CM, Russell CM, Reinhardt RA, Payne JB, Lyle DM. Comparison of irrigation to floss as an adjunct to tooth brushing: effect on bleeding, gingivitis, and supragingival plaque. J Clin Dent. 2005;16:71–77.
- Apatzidou DA. Modern approaches to nonsurgical biofilm management. Front Oral Biol. 2012;15:99–116.
- Sanz M, Teughels W, Group A of European Workshop on Periodontology. Innovations in nonsurgical periodontal therapy: consensus report of the sixth european workshop on periodontology. J Clin Periodontol. 2008;35(8 Suppl):3–7.
- Smiley CJ, Tracy SL, Abt E, et al. Evidence-based clinical practice guideline on the nonsurgical treatment of chronic periodontitis by means of scaling and root planing with or without adjuncts. J Am Dent Assoc. 2015;146:525–535.
- Slots J. Low-cost periodontal therapy. Periodontol 2000. 2012;60:110–137.
- Krück C, Eick S, Knöfler GU, Purschwitz RE, Jentsch HF. Clinical and microbiologic results 12 months after scaling and root planing with different irrigation solutions in patients with moderate chronic periodontitis: a pilot randomized trial. J Periodontol. 2012;83:312–320.
- Sahrmann P, Imfeld T, RonayV, Attin T, Schmidlin PR. Povidone-iodine gel and solution as adjunct to ultrasonic debridement in nonsurgical periodontitis treatment: an RCT pilot study. Quintessence Int. 2014;45:281–290.
- Greenstein G. Nonsurgical periodontal therapy in 2000: a literature review. J Am Dent Assoc. 2000; 131:1580–1592.
- Consensus report. Nonsurgical pocket therapy: mechanical, pharmacotherapeutics, and dental occlusion. Ann Periodontol.1996;1:581–588.
Featured image by ZLIKOVEC/ISTOCK/GETTY IMAGES PLUS
From Decisions in Dentistry. May 2018;4(5):11-14.



[…] oral cavity. These cells have been found to protect against oral thrush, but also have a link to periodontitis, which causes bone and tooth loss. Findings from the study, “A Dysbiotic Microbiome Trigger TH17 […]