
Administration of Nitrous Oxide Analgesia
Use of this form of conscious sedation represents an effective alternative pain management strategy for dental procedures.
This course was published in the January 2018 issue and expires January 2021. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
OBJECTIVES
- Explain the role of nitrous oxide (N2O) in dentistry and its effects on patients.
- List possible contraindications for the administration of conscious sedation during dental treatment.
- Discuss important clinical considerations when administering N2O sedation.
The administration of N2O-O2 is primarily indicated for patients who exhibit mild to moderate dental anxiety or fear. Minimal sedation is achieved when N2O is used in concentrations of less than 50%, but it can help relieve anxiety associated with the sights, sounds and smells of the operatory, as well as reduce the gag reflex.5 Administration of N2O-O2 can help patients with controlled conditions exacerbated by stress, such as asthma, angina or seizures.4 Some procedures can be completed using concentrations of less than 50%.6 In cases that require higher concentrations of N2O, dentists may elect to exceed the 50% minimal sedation level. Patients with severe dental anxiety, or who require more invasive procedures, may need deeper sedation. In these situations, providers may pair N2O with oral or intravenous sedatives.
CONTRAINDICATIONS
It is difficult to find consensus regarding absolute contraindications to N2O use. Each patient should be evaluated individually, weighing the risks and benefits to identify possible relative contraindications — which are the individual circumstance that increases a patient’s potential for an adverse reaction. For example, N2O is contraindicated for elective treatment or prophylaxis among pregnant patients, as it crosses the placental barrier, exposing the fetus. If a pregnant patient needed an emergency procedure, however, its use may be warranted after a medical consultation with her physician.4,7,8 Other relative contraindications include chronic obstructive pulmonary disease, cystic fibrosis, recent middle ear or eye surgery where a gas bubble was used, bowel obstruction, current or recovering addiction issues, psychological impairment, use of antidepressants or psychotropic drugs, or severe cardiovascular conditions.3,4,6,9 When used concurrently with a minimum of 30% oxygen, N2O possess no additional risks to patients, and can be used in most situations when minimal sedation is indicated.4,10
There are instances, however, in which N2O delivery simply will not be effective. For example, a patient must be able to breathe through his or her nose. Patients with active upper respiratory infection, rhinitis, or abnormal nasal anatomy that impairs nasal airflow may not be suitable candidates for conscious sedation. Patients who cannot tolerate the nasal hood or experience claustrophobia are also not good candidates for N2O-O2 sedation.

ADMINISTRATION
Administering N2O-O2 is quite simple (Table 1). Always begin with a thorough review of the patient’s health history and medications to determine the individual’s American Society of Anesthesiologists (ASA) physical status classification. Obtain vital signs that include blood pressure, pulse and respiratory rate. Additionally, the use of pulse oximetry — which includes monitoring and recording oxygen saturation of the blood at the beginning, during and upon completion of the procedure — is prudent when delivering inhalation sedation. According to the ASA’s “Practice Guidelines for Sedation and Analgesia by Non-Anesthesiologists,” the use of a pulse oximeter is required for any patient who will have more than 50% N2O delivered, which is considered more than minimal sedation.11
The gas mixture is delivered via a nasal hood that fits over the patient’s nose. Various hood sizes are available, and correct selection maximizes the benefits of the gaseous agent to the patient while minimizing leakage into the operatory. Begin conscious sedation procedures by turning on the oxygen to approximately 6 liters per minute. Assist the patient, but allow him or her to place the nasal hood over the nose, ensuring a comfortable and proper fit. Establish the patient’s tidal volume (typically, 6 to 8 liters/minute for adults, and 3 to 5 liters/minute for children) to identify the proper flow rate. If the patient is not able to breathe comfortably through the nose and feels the need to take a breath through the mouth, providers should increase the liters of oxygen being delivered.
Clinicians should begin inhalation sedation with the titration of N2O at a concentration level of 20%.4,6 Remember that sedation is a continuum, thus, it is not possible to know exactly how a patient will respond to a given concentration. Continue titration, using the “start low, go slow” mantra, by increasing N2O by 5% approximately every 1 to 2 minutes, making sure the established flow rate is maintained at all times. Once the proper level has been achieved and the patient is starting to show ideal signs and symptoms (Table 2, page 46) of conscious sedation, clinicians should maintain the desired concentration.4,12,13 Some experts suggest reducing the concentration by a few percent at this point to avoid delayed increases in intensity while establishing the baseline. Patients often refer to N2O as “laughing gas,” however, individuals who display uncontrollable laughter are likely receiving too high of a concentration of N2O gas.
Once the desired level of sedation is achieved, the procedure may begin. Patients should never be left unattended during administration. Upon completion of treatment, terminate the flow of N2O, maintain the established tidal volume, and increase the O2 concentration to 100% for no less than 3 to 5 minutes in order to remove the increased N2O present at the alveolar sites in the lungs.4,10 If patients are still experiencing signs and symptoms of sedation, additional O2 may be necessary. This helps avoid diffusion hypoxia, which may cause headache, nausea or grogginess due to the reduction of oxygen reaching the body from the lungs.
Administration documentation is required in the patient’s health record. The information should include vital signs, O2 saturation, established tidal volume, percent concentration of the gases, duration of N2O-O2 delivery and duration of O2 given upon completion of procedure. Documentation should also include how the patient responded to the N2O. Submit charges using the American Dental Association (ADA), current dental terminology code D9230.
MECHANISM OF ACTION
Because N2O is the least potent of the anesthetic gases, patients will not all respond in similar fashion. Approximately 5% of the population is considered to be hyporesponsive and will not feel the effects at all.3,4 Another 5% of the population is hyperresponsive and will feel the effects quickly.3 For inhalation anesthetics, the mechanism of action occurs through diffusion, which is dependent on a concentration gradient; meaning, gas will diffuse from areas of higher concentration to areas of lower concentration. As N2O is inhaled, there is a higher concentration of N2O in the lungs, so the N2O will diffuse into the blood. This process occurs rather quickly, as N2O has one of the lowest partition coefficients of any gas mixture.
Referred to as relative analgesia, N2O does not block or eliminate pain, but, instead, attenuates the body’s response to noxious stimuli by raising the pain threshold. Its role as an anesthetic is limited because when N2O is used as a single agent for minimal and moderate levels of conscious sedation, it is incapable of inducing general anesthesia.7,9 Though there has been great progress in the understanding of N2O over the past 30 years, its analgesic, anxiolytic and anesthetic mechanisms of action are still not fully understood.13
SAFETY
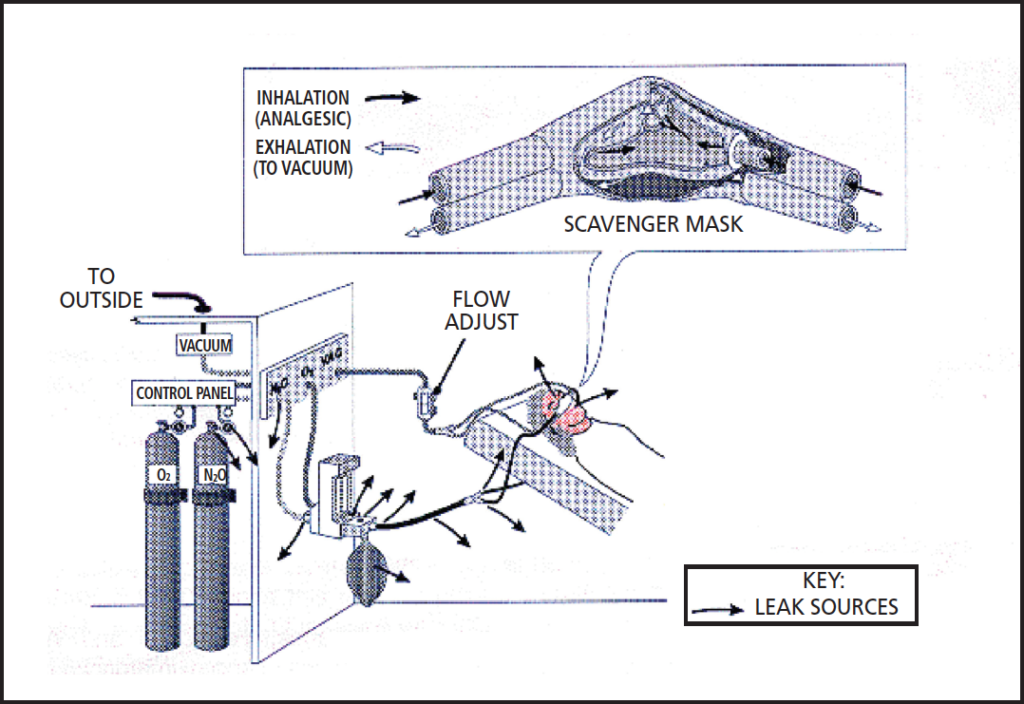
More defined regulations, equipment safety features, fail-safe mechanisms and monitoring protocols have contributed to the decrease in lethal doses and incidental occupational exposures resulting from N2O.14 In 1976, the ADA Council on Dental Materials, Instruments and Equipment adopted standards for inhalation units in the United States. By 1980, the council recommended that scavenging units be installed and monitoring protocols implemented.15 Today, operating a delivery system without a scavenging unit to decrease the amount of waste gas in the air fails to meet the minimum standard of care (Figure 1).10 The National Institute for Occupational Safety and Health recommends an exposure limit of 25 ppm in the dental operatory during N2O administration.16 For an 8-hour, time-weighted average, the American Conference of Governmental Industrial Hygienists recommends a threshold limit value of 50 ppm.17 While dosimeter badges are available to monitor a provider’s exposure of N2O, the accuracy of these badges is questionable, as trace amounts of N2O found in the ambient air can also be recorded.
The single most important safety step in delivery is the titration method. Incremental doses of N2O delivered over a short period allow for the minimum concentration to be used while still achieving the desired result. This increases patient safety, and also minimizes waste gas exposure to the clinician.4
As with any drug or agent, the potential for harm exists and risk increases with chronic, high-concentration exposure. Prolonged contact (more than six hours) and/or long term use of N2O has been shown to inactivate vitamin B12, causing a biochemical imbalance that may lead to bone marrow depression, megaloblastic anemia, pernicious anemia and neurological deficits, such as peripheral neuropathies.18–21 Additional studies have demonstrated that exposures, even at high levels, of six hours or less have not been shown to cause long-lasting physiological changes and are generally considered safe.3,4,12,22,23
CONCLUSION
Along with fluoride and local anesthesia, some consider the administration of N2O as one of the greatest achievements in oral health care.24 Dental professionals who wish to add to add N2O to their armamentariums must obtain the proper education and training to deliver this safe and effective agent. State dental practice acts also should be reviewed to ensure N2O administration falls within the scope of practice of the individual provider.
REFERENCES
- National Center for Biotechnology Information. Compound Summary for CID 948, Nitrous Oxide. Available at: pubchem.ncbi.nlm.nih.gov/compound/948. Accessed November 30, 2017.
- Wells H. A history of the discovery of the application of nitrous oxide gas, ether and other vapors to surgical operations. Available at: woodlibrarymuseum.org/museum/item/139/wells-h.-a-history-of-the-discovery-of-the-application-of-nitrous-oxide-gas,-ether,-and-other-vapors,-to-surgical-operations,-1847. Accessed November 30, 2017.
- Clark M, Brunick A. Handbook of Nitrous Oxide and Oxygen Sedation. 4th ed. St. Louis: Elsevier Mosby; 2015.
- Malamed S. Sedation: A Guide to Patient Management. 5th ed. St. Louis: Mosby Elsevier; 2010.
- Chidiac JJ, Chamseddine L, Bellos G. Gagging prevention using nitrous oxide or table salt: a comparative pilot study. Int J Prosthet. 2001;14:364.
- Darby M, Walsh M. Dental Hygiene Theory and Practice. 4th ed. St. Louis: Elsevier Saunders; 2015:768–785.
- Becker D, Rosenburg, M. Nitrous oxide and the inhalation anesthetics. Anesth Prog. 2008; 55:124–131.
- Crawford JS, Lewis M. Nitrous oxide in early pregnancy. Anaesthesia. 1986;41:900–905.
- Clark M. Back to the future: an update on nitrous oxide/oxygen sedation. Available at: dentalacademyofce.com/courses/2044/PDF/1103cei_nitrous.pdf. Accessed November 30, 2017.
- Shuman I. Nitrous oxide: use and safety. Available at: dentalacademyof ce.com/courses/3036%2FPDF%2F1604cei_Shuman_Nitrous%20-%20 updated%20web.pdf. Accessed November 30, 2017.
- American Society of Anesthesiologists Task Force on Sedation and Analgesia by Non-Anesthesiologists. Practice guidelines for sedation and analgesia by non-anesthesiologists. Anesthesiology. 2002;96:1004–1007.
- Dionne R, Phero JC, Becker DE. Management of Pain and Anxiety in the Dental Office. St. Louis: WB Saunders; 2002.
- Emmanouil DE, Quock RM. Advances in understanding the actions of nitrous oxide. Anesthesia Progress. 2007;54:9–18.
- Whitcher CE, Zimmerman DC, Tonn, EM, Piziali, RL. Control of occupational exposure to nitrous oxide in the dental operatory. J Am Dent Assoc. 1977;95:763–776.
- Nitrous oxide in the dental office. ADA Council on Scientific Affairs; ADA Council on Dental Practice. J Am Dent Assoc. 1997;128:364–365.
- National Institute for Occupational Safety and Health. Control of Nitrous Oxide in Dental Operatories. Available at: cdc.gov/niosh/docs/hazardcontrol/hc3.html. Accessed November 30, 2017.
- American Conference of Governmental Industrial Hygienists. TLVs, Threshold Limit Values for Chemical Substances in the Work Environment. Cincinnati; 1990:32.
- Lassen HC, Henriksen E, Neukirch F, Kristensen H. Treatment of tetanus: Severe bone-marrow depression after prolonged nitrous-oxide anesthesia. Lancet. 1956;267:527–530.
- Yagiela J. Health hazards and nitrous oxide: a time for reappraisal. Anesthesia Progress. 1991;38:1–11.
- Amess J, Burman JF, Rees GM, et al. Megaloblastic haemopoiesis in patients receiving nitrous oxide. Lancet. 1978;2:339–342.
- Nunn JF, Sharer NM, Gorchein A, et al. Megaloblastic haemopoiesis after multiple short-term exposures to nitrous oxide. Lancet. 1982;1:1379–1381.
- Weimann J. Toxicity of nitrous oxide. Best Pract Res Clin Anaesthesiol. 2003; 17:47–61.
- O’Sullivan H, Jennings F, Ward K, et al. Human bone marrow biochemical function and megaloblastic hematopoiesis after nitrous oxide anesthesia. Anesthesiology. 1981;55:645–649.
- Greene NM. A consideration of factors in the discovery of anesthesia and their effects on its development. Anesthesiology.1971;35:515–522.
The authors have no commercial conflicts of interest to disclose.
FEATURED IMAGE BY WAVEBREAKMEDIA/ISTOCK/GETTY IMAGES PLUS
From Decisions in Dentistry. January 2018;4(1):44-46,49.





