
Vaccination Recommendations for Oral Health Professionals
Dental teams should familiarize themselves with vaccination recommendations to reduce the risk of infectious diseases and better educate patients about the importance of immunizations.
This course was published in the September 2018 issue and expires September 2021. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
Educational Objectives
After reading this course, the participant should be able to:
- Identify vaccination recommendations for health care professionals.
- Discuss the dental team’s role in ensuring that staff members are appropriately vaccinated.
- Provide resources that will help ensure patients receive appropriate vaccinations to prevent disease and maintain optimal health.
HEPATITIS B VIRUS
Infection with the hepatitis B virus can lead to lifelong chronic infection and serious liver damage.1 Dental providers are in frequent contact with blood, saliva and other bodily fluids that increase their risk of contracting hepatitis B through percutaneous or mucosal exposure. The hepatitis B vaccination is the most effective measure of prevention.2–4 As part of standard precautions and the Occupational Safety and Health Administration’s Bloodborne Pathogens Standard, employers are required to make hepatitis B vaccination available to all employees who may come in contact with blood or other potentially infectious material.5
The hepatitis B vaccination is recommended for all health care professionals.3,5,6 For unvaccinated individuals, or if no proof of vaccination is available, a three-dose series (on a zero-, one- and six-month schedule) of hepatitis B vaccines is administered. Testing for antibodies against hepatitis B virus surface antigen (HBsAg) is recommended one to two months after the third dose to evaluate the response to the vaccination. The three-dose series produces a protective antibody response in more than 90% of healthy adults younger than 40. Completely vaccinated health care professionals with anti-HBsAg less than 10 mIU/ml should receive an additional dose of hepatitis B vaccine, followed by additional HBsAg testing one to two months later. This additional dose is referred to as a challenge dose or booster to provide rapid protective immunity against infection. The challenge dose may be used to determine the presence of vaccine-induced immunologic memory through the generation of an anamnestic response (renewed rapid production of an antibody on the second or subsequent encounter with the same antigen). Providers whose anti-HBsAg levels remain less than 10 mIU/ml should complete the second series (usually six doses total), and anti-HBsAg testing should be done one to two months after the final dose.
If no antibody response occurs after six doses of the vaccination, testing for HBsAg is strongly recommended. If HBsAg negative, health care professionals will be counseled about precautions to prevent hepatitis B virus infection, and the need to obtain hepatitis B immune globulin (HBIG) prophylaxis for any known or probable parenteral exposure to HBsAg-positive blood. This measure can increase protection until a response to vaccination is obtained. While HBIG administered alone is the primary means of protection after a hepatitis B virus exposure, HBIG also provides passively acquired anti-HBs and temporary protection for three to six months. Passively acquired anti-HBs can be detected for four to six months after administration of HBIG. If HBsAg positive, health care professionals will be referred for postexposure evaluation and follow-up, and counseled about the need for work restrictions to prevent transmission of hepatitis B to others.
For vaccinated health care professionals with documented anti-HBs ≥ 10 mIU/ml, there is no need for hepatitis B prophylaxis, regardless of the source patient hepatitis B surface antigen status. If dental team members decline the hepatitis B vaccination, a signed copy of a hepatitis B vaccination declination form should be filed in their personnel records.
In November 2017, the U.S. Food and Drug Administration (FDA) approved the HepB-CpG vaccine for the prevention of hepatitis B in individuals age 18 and older. It offers a two-dose schedule (zero and one month).7
MEASLES, MUMPS and RUBELLA
As one of the most transmissible vaccine-preventable diseases, measles does not require personal contact for transmission. The measles virus causes symptoms that can include fever, cough, runny nose and red, watery eyes, commonly followed by a rash that covers the entire body. It can lead to ear infections, diarrhea and pneumonia; however, it rarely causes brain damage or death.8
If vaccination recommendations are followed, herd immunity, in which unimmunized individuals are protected because a large enough percentage of individuals has been vaccinated, can be achieved by 92% to 95% vaccination coverage.9 Suboptimal vaccination rates can lead to regular outbreaks. In recent years, measles outbreaks have occurred in New York, New Jersey, Europe, Africa, Hong Kong, Taiwan and Japan. In 2015, a large multistate outbreak was linked to Disneyland in Anaheim, California, and started from a traveler who became infected overseas and then visited the park. The U.S. Centers for Disease Control and Prevention (CDC) confirmed this outbreak was the identical virus strain genotype that caused the large measles outbreak in the Philippines in 2014. The majority of those infected were unvaccinated at the time.10,11
Mumps is a viral disease whose main target is the parotid salivary gland. It can cause complications, such as encephalitis, meningitis, orchitis (inflammation of the testicles), oophoritis (inflammation of the ovaries), deafness and pancreatitis, but death is rare.2,8 The mumps virus causes fever, headache, muscle aches, tiredness, loss of appetite, and swollen and tender salivary glands under the ears on one or both sides. The herd immunity threshold for mumps is about 90%. Though the disease is no longer common in the developed world, mumps outbreaks still occur in the United States, especially in areas of close contact, such as schools, colleges and camps.10
Rubella (also known as German measles) is a mild infection in children, but can be serious in pregnant women due to the risk of congenital infection. The rubella virus causes fever, sore throat, rash, headache and eye irritation. In up to half of adolescent and adult women, rubella causes arthritis. If a woman gets rubella while pregnant, she could have a miscarriage or the baby could be born with serious birth defects.8 To prevent outbreaks, at least 85% of the population should be immune to rubella.
Measles, mumps, and rubella (MMR) are highly contagious viral infections. Compared with the general population, health care professionals have a greater risk of acquiring measles due to its highly contagious nature and ability to persist in aerosol suspension for at least an hour. Transmission from patients to unprotected dental team members can occur via infected individuals who seek care before developing clinically recognizable disease. Additionally, susceptible clinicians may expose colleagues and patients to increased risk.6
The MMR vaccine contains weakened versions of live MMR viruses. It works by triggering the immune system to produce antibodies against MMR. This vaccination recommendation applies to all U.S. clinicians. The World Health Organization has not provided any specific recommendations or evidence of measles immunity in health care professionals. Currently accepted proof of immunity includes documented administration of two MMR vaccine doses separated by at least 28 days, laboratory evidence of immunity, and laboratory confirmation of disease.6,12 In the United States, one or two doses of MMR vaccine are recommended for all unvaccinated health care professionals without laboratory-confirmed disease or immunity against MMR during nosocomial outbreaks.3
The Advisory Committee on Immunization Practices (ACIP) recommends that all health care professionals born in or after 1957 have adequate presumptive evidence of immunity to MMR. Further, it recommends that health care facilities should consider vaccination of all unvaccinated health care personnel who were born before 1957 and who lack laboratory evidence of MMR immunity or laboratory confirmation of disease. In October 2017, ACIP recommended that at-risk individuals previously vaccinated with two doses of a mumps-virus-containing vaccine should receive a third dose of a mumps-virus-containing vaccine to raise protection levels.2
PERTUSSIS, TETANUS AND DIPHTHERIA
Pertussis, also known as whooping cough, is a highly transmissible respiratory disease that causes severe coughing spells which, in turn, can lead to difficulty breathing, vomiting, disturbed sleep, weight loss, incontinence and rib fractures.2 Health care professionals are at increased risk for pertussis if in contact with infected patients, especially if the clinician has waning protection from either childhood pertussis vaccination or prior pertussis infection. Infected practitioners can serve as sources of infection for susceptible contacts, including dental teammates, patients and family members.
Caused by the bacterium Clostridium tetani, tetanus enters the body through cuts, scratches or wounds. Contamination with tetanus spores in unimmunized individuals can evoke illness, with muscle spasms and sometimes death. It causes painful muscle tightening and stiffness, usually all over the body. Tetanus, or lockjaw, prevents an individual from opening his or her mouth, swallowing, and sometimes from breathing due to tightening of muscles in the head and neck region.13 In order to maintain immunity, periodic boosters in adulthood are required. Reported tetanus cases continues to be low in the United States.13
Diphtheria usually produces respiratory symptoms, although it can also affect other organs and even cause death. A thick coating is formed in the back of the throat, leading to breathing problems, heart failure, paralysis or death. Cases are rarely seen in the United States.13
Generally, vaccination recommendations offer the best protection against pertussis infection. In 2006, ACIP recommended that health care professionals ages 19 to 64 receive a single dose of the tetanus, diphtheria and acellular pertussis vaccine (Tdap) to reduce the risk of pertussis transmission. In 2010, ACIP officials indicated that all health care professionals, regardless of age, should receive a single dose of Tdap as soon as possible if they had not previously received the vaccine.14 Another vaccine called Td protects against tetanus and diphtheria, but not pertussis. A Td booster should be given every 10 years. Tdap may be given as one of these boosters if the individual has never received the vaccine before; Tdap may also be given after a severe cut or burn to prevent tetanus infection. Currently, ACIP recommends that adults receive an initial dose of the Tdap vaccine, with a Td booster every 10 years.10,15 Protective antibody titers have been defined and validated as a correlation of protection against tetanus and diphtheria.
VARICELLA
Varicella (or chickenpox) is caused by varicella zoster virus (VZV), which is a herpes virus. It is a highly contagious disease that is spread by contact with respiratory droplets and/or vesicular fluid. It is usually self-limited, but may cause severe complications, such as lower respiratory tract infection, skin and soft tissue infection, or even death.16,17 The virus is responsible for chickenpox in primary infections, which occur mostly in children, teenagers and young adults. Infection during childhood induces long-lasting immunity. For herpes zoster, the VZV reactivates after a latent period in sensory nerve ganglia and occurs generally in older people.2 In 1996, two doses of varicella vaccine administered four to eight weeks apart were recommended for susceptible adults, including health care professionals. However, seroconversion to vaccination may not ideally correlate with protection, as vaccine-induced antibody titers can decline or disappear over time.18
Vaccination has greatly reduced varicella incidence, resulting in declines in varicella exposures in health care settings. Nevertheless, VZV exposures still occur, particularly from herpes zoster cases. Once infected, clinicians may transmit infection to coworkers and patients. Therefore, it is important to vaccinate health care professionals against varicella. Documentation of VZV immunoglobulin G status of all providers and the vaccination of susceptible clinicians is suggested for effective prevention of nosocomial varicella.10 Until 2017, ACIP recommended that individuals age 60 and older receive a herpes zoster or shingles vaccination.15 In October 2017, the FDA approved a new recombinant, adjuvenated vaccine for the prevention of herpes zoster in adults age 50 and older. It is a two-dose schedule (two to six months apart).16
INFLUENZA
Influenza is an infectious disease caused by a virus that can affect humans, birds, pigs and chickens.2 The virus mutates quickly, and its seasonal strains cause outbreaks (mostly during winter). The influenza virus infects the nose, throat and sometimes the lungs, causing fever and musculoskeletal pain. It can cause mild to severe illness, and can prove fatal.19 Most experts believe that flu viruses are spread mainly by tiny droplets created when infected individuals cough, sneeze or converse. These droplets can land in the mouth or nose of those who are nearby. Individuals can also be infected through contact with surfaces or inanimate objects that have the active virus on them, and then touching their mouths, nose or possibly eyes.
Seasonal flu vaccines are designed to protect against infection and illness. How well the vaccine works or its ability to prevent flu illness can range widely. The vaccine’s effectiveness can also vary, depending on who is being vaccinated.19 Research assessing the benefits and risks of the flu vaccine in providers and patients supports the vaccine’s efficacy; thus, ACIP recommends that all health care professionals and those in training receive an annual vaccination.3,4,10,20 Unimmunized practitioners can act as an infection source, potentially leading to nosocomial influenza outbreaks.21 Vaccination of health care professionals has been associated with reduced work absenteeism and fewer deaths among nursing home patients and elderly hospitalized patients. The ACIP also recommends that adults receive an annual seasonal influenza vaccination.15
ORAL HEALTH PROFESSIONALS’ ROLE
Oral health professionals play a key role in the immunization process at two levels. As members of the extended health care team, dental personnel can develop and implement office protocols regarding appropriate vaccinations. Once an office policy is implemented, team members can assist with ongoing monitoring and documentation.22 Dentists, dental hygienists and dental assistants are also integral to patient education. Because oral health professionals may practice in a variety of settings, such as public health clinics, schools, correctional facilities, nursing homes and private practice settings, they are able to educate patients about the importance of immunizations. Incorporating questions on the health history that reveal patients’ knowledge regarding their vaccination status can open a dialogue between patients and providers. General questions can be added to the health history form regarding vaccination status, including past history and/or exposure.
Clinicians can help reduce disease by emphasizing the importance of immunizations to staff members and patients. As part of the interprofessional team, oral health professionals are responsible for identifying a patient’s risk factors based on age, health, pregnancy status, type of work environment, and travel outside of the United States.20 Each member of the dental team should be knowledgeable about the most current vaccination recommendations, as outlined by the CDC — including policies and documentation of vaccinations. Table 1 lists helpful resources for health care professionals and the public regarding immunization schedules.
CONCLUSION
Vaccines are continuously being developed, monitored, evaluated and revised through recommendations provided by ACIP. Health care professionals are advised to be familiar with these ongoing changes to protect providers and patients from exposure to infectious diseases, and so they can educate patients about the vital role immunizations play in disease prevention. As health care continues to move toward an interprofessional approach, oral health professionals have the potential to be instrumental in improving public health outcomes.
VACCINATION BACKGROUND AND HISTORY
accination is an essential component of public health programs and ranks among the most cost effective medical interventions.23 According to the U.S. Centers for Disease Control and Prevention (CDC), immunization is the means by which a person is protected against a disease.20,24 The terms immunization, vaccination and inoculation are often used interchangeably.20,24 A vaccine is a weakened form or passive form of the disease that is given for inoculation.23,24 It is generally administered by injection, but it can also be administered by mouth or sprayed into the nose.20 Immunization can occur in two ways: natural immunization or vaccination. Natural immunization is when the body develops antibodies to the specific disease after exposure, and the immune system subsequently is able to respond to the disease and prevent infection.24 Vaccination is exposing a person to a disease, such as polio, by introducing a small amount of the disease into the body.20,23,24 The body reacts to these foreign molecules as though it has contracted the disease and creates antibodies to fight the polio vaccine. If the individual is subsequently exposed to polio, the immune response — once established — will be triggered and keep the body polio free.20,23,24
One of the most significant public health interventions, vaccination is clearly effective in reducing the risk of diseases that previously attacked large proportions of the population, caused many deaths and left many with disabilities.20,23,24 Infectious disease was the most serious threat to human life at the beginning of the 20th century.23,24 Outbreaks of diseases — such as diphtheria and pertussis — were common, and mortality rates were significant: 160 of every 1000 children born at the turn of the century died of an infectious disease before the age of 5.23 The first known vaccinations for smallpox are believed to have been used as far back as 1000 AD in China. Two methods of vaccinating included making a powder from smallpox scabs which was blown into a person’s nose, and another was to scratch the matter from an open sore onto another person’s skin.23 The modern-day smallpox vaccine was developed by Dr. Edward Jenner in Berkley, England, who performed a vaccination in 1796 by taking exudate from a cowpox lesion on a milkmaid’s hand and inoculating an 8-year-old boy, who was unaffected by this and subsequent exposures — thus laying the foundation for modern vaccinology.24–26 Since then, smallpox is considered eradicated, with the last U.S. case reported in 1948 and the last international case in 1977 in Somalia.24,26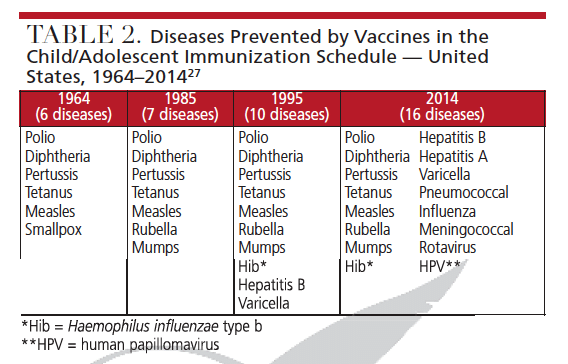
Similar to all other pharmaceutical products, vaccines are not entirely risk-free; while most known side effects are minor, some have been associated with rare, but serious, adverse effects.23 The U.S. government maintains ongoing surveillance programs to monitor vaccine safety.20,23 In 1964, the Advisory Committee on Immunization Practices (ACIP) was appointed by the Surgeon General, head of the U.S. Public Health Service (USPHS), to provide expert advice to the CDC and Secretary of the U.S. Department of Health and Human Services on the use of vaccines. Prior to this, USPHS relied on committees and convened intermittently to address various biologic-related health issues.27 Since the inception of ACIP, the number of recommendations included in the child/adolescent immunization schedule has increased from vaccines targeting six diseases to those for the prevention of 16 diseases (Table 2), while the recommended immunization schedule for adults now includes 15 vaccine-preventable diseases.27
In 1993, enactment of the Vaccines for Children program was added to the ACIP’s role, which provides free vaccination for children age 0 through 18 years who are uninsured, underinsured, Medicaid eligible or American Indian/ Alaska Native. Eligible children receive vaccines at federally qualified health centers or rural health clinics.27 The links in Table 3 offer further information pertaining to the immunization schedules and a “Journey of a Vaccine and Monitoring” portion that provides a clear and comprehensive understanding of the importance of child immunizations. The ACIP has evolved over the years and uses a systematic economic analysis in the development
of vaccines, in addition to evidence-based format for presentation of recommendations. Both immunization schedules are updated and approved by ACIP and other professional societies and cited on each schedule.
Healthcare workers (HCWs) who are directly or indirectly involved in patient care can be exposed to infectious agents that can be transmitted to and from HCWs and patients. This provider group includes dental personnel, physicians, nurses, nursing assistants, therapists, technicians, emergency medical service personnel, pharmacists, laboratory personnel, autopsy personnel, students and trainees.14 HCWs are recommended to have vaccinations for protection against exposure to infectious agents and to minimize risks of cross infection.
REFERENCES
- Cohen C, Caballero J, Martin M, et al. Eradication of hepatitis B: A nationwide community coalition approach to improving vaccinations, screening, and linkage to care. J Community Health. 2013;38:799–804.
- Haviari S, Benet T, Saadatian-Elahi M, et al. Vaccination of healthcare workers: a review. Hum Vaccin Immunother. 2015;11:2522–2537.
- Schillie S, Vellozzi C, Reingold A, et al. Prevention of hepatitis B virus infection in the United States: recommendations of the Advisory Committee on immunization practices. MMWR Morb Mortal Wkly Rep. 2018;67(RR-1):1–31.
- Abara WE, Qaseem A, Schillie S, et al. Hepatitis B vaccination, screening, and linkage to care: Best practice advice from the American College of Physicians and the Centers for Disease Control and Prevention. Ann Int Med. 2017;167:794–804.
- Guidelines for infection control in dental health-care settings — 2003. MMWR Morb Mortal Wkly Rep. 2003;52(RR17);1–61.
- New York State Department of Health. Health Advisory: Recommendations for Vaccinations of Health Care Personnel Available at: health.ny.gov/prevention/immunization/toolkits/docs/health_advisory.pdf. Accessed July 25, 2018.
- Schillie S, Harris A, Link-Gelles R, et al. Recommendations of the Advisory Committee on Immunization Practices for the use of a hepatitis B vaccine with a novel adjuvant. MMWR Morb Mortal Wkly Rep. 2018;67:455–458.
- U.S. Centers for Disease Control and Prevention. Measles, Mumps, and Rubella. Available at: cdc.gov/vaccines/hcp/vis/vis-statements/mmr.html. Accessed July 25, 2018.
- Brever NT, Moss JL. Herd immunity and the herd severity effect. Lancer Infec Dis. 2011;15:868–869.
- U.S. Centers for Disease Control and Prevention. Mumps Cases and Outbreaks. Available at: cdc.gov/mumps/outbreaks.html. Accessed July 25, 2018.
- Zipprich J, Winter K, Hacker J, et al. Measles outbreak — California, December 2017–February 2015. MMWR Morb Mortal Wkly Rep. 2015;64:153–154.
- U.S. Centers for Disease Control and Prevention. Routine Measles, Mumps, and Rubella Vaccination. Available at: cdc.gov/vaccines/vpd/ mmr/hcp/recommendations.html Accessed July 25, 2018.
- U.S. Centers for Disease Control and Prevention. Tdap (Tetanus, Diphtheria, Pertussis. Available at: cdc.gov/vaccines/hcp/vis/vis-statements/tdap.html. Accessed July 25, 2018.
- Lu PJ, Graitor SB, O’Halloran A, et al. Tetanus, diphtheria and acellular pertussis (tdap) vaccination among healthcare personnel — United States, 2011. Vaccine. 2014;32:572–578.
- La EM, Trantham L, Kurosky SK, et al. An analysis of factors associated with influenza, pneumococcal, Tdap, and herpes zoster vaccine uptake in the U.S. adult population and corresponding inter-state variability. Hum Vaccin Immunother. 2018;14:430–441.
- Dooling KL, Guo A, Patel M, et al. Recommendations of the Advisory Committee on Immunization Practices for use of herpes zoster vaccines. MMWR Morb Mortal Wkly Rep. 2018;67:103–108.
- Wu MF, Yang YW, Lin WY, et al. Varicella zoster virus infection among healthcare workers in Taiwan: seroprevalence and predictive value of history of varicella infection. J Hosp Infect. 2012;80:162–167.
- Behrman A, Lopez AS, Chaves SS, et al. Varicella immunity in vaccinated healthcare workers. J Clin Viro. 2013;57:109–114.
- U.S. Centers for Disease Control and Prevention. Vaccine Effectiveness — How Well Does the Flu Vaccine Work? Available at: cdc.gov/flu/about/qa/vaccineeffect.htm. Accessed July 25, 2018.
- U.S. Centers for Disease Control and Prevention. Immunization: the Basics. Available at: https://www.cdc.gov/vaccines/vac-gen/imz-basics.htm. Accessed July 25, 2018.
- Hagemeister MH, Stock NK, Lydwig T, et al. Self-reported influenza vaccination rates and attitudes towards vaccination among health care workers: Results of a survey in a German university hospital. Public Health. 2018;154:102–109.
- Giblin L. Exposure control: barriers for patient and clinician. In: Wilkins EM, Wyche CJ, Boyd LD, eds. Clinical Practice of the Dental Hygienist. 12th ed. Philadelphia: Lippincott, Williams & Wilkins; 2016.
- Ellenberg SS, Chen, RT. The complicated task of monitoring vaccine safety. Public Health Rep. 1997;112:10–20.
- Goldbas A. The law and immunization in the United States. Int J Childbirth Educ. 2016;31:41–43.
- Stern AM, Markel H. The history of vaccines and immunization: Familiar patterns, new challenges. Health Aff. 2005;24:611–621.
- U.S. Centers for Disease Control and Prevention. The History of Smallpox. Available at: https://www.cdc.gov/smallpox/history/history.html. Accessed July 25, 2018.
- Smith JC, Hinman AR, Pickering LK. History and evolution of the advisory committee on immunization practices — United States, 1964–2014. MMWR Morb Mortal Wkly Rep. 2014;63:955–958.
Featured Image by SASIISTOCK/ISTOCK/GETTY IMAGES PLUS
From Decisions in Dentistry. September 2018;4(9):36–39.




