Update on Maximum Local Anesthesia Dosages
Adhering to U.S. Food and Drug Administration guidelines will support patient safety and comfort during dental treatment.
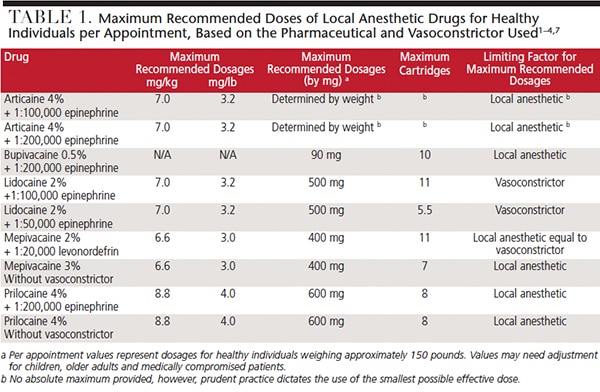
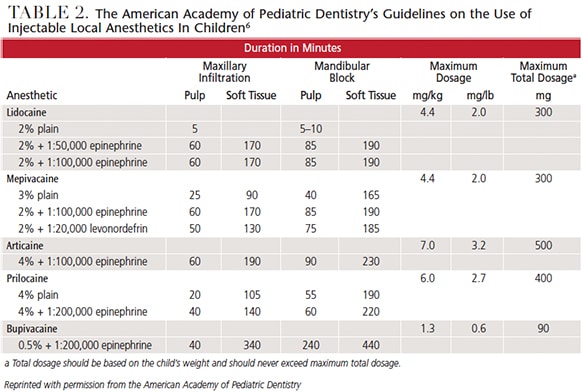
The U.S. Food and Drug Administration (FDA) Center for Drug Evaluation and Research, Office of Pharmaceutical Science, Informatics and Computational Safety Analysis created and regularly updates maximum recommended dosages (MRDs) for local anesthetic drugs — although some clinicians may not know of the guideline’s existence.1 And among those who do, some use the MRDs published in older editions of popular textbooks on local anesthesia when calculating drug dosages for patients, which may differ significantly from the values provided in the latest FDA-approved recommendations (Table 1). Oral health professionals — particularly those in the Western U.S. — have been guided for a number of years by lower MRD values than those approved by the FDA.2–4 There are benefits to following lower MRDs — such as enhanced patient safety — and as long as administered doses remain within the FDA recommendations, there is no breach of the guidelines. That said, maintaining two sets of guidelines can be confusing for dental teams5 — which serves as a convincing argument for eliminating non-FDA-established values.5 At this time, however, some clinicians and professional groups have not adopted the higher values. The American Academy of Pediatric Dentistry (AAPD), for example, continues to list the lower values in its monograph (Table 2).5,6 Like the AAPD, other organizations, clinicians and clinics who use lower MRDs add an additional layer of safety to their protocols, without compromising treatment or comfort.
The impact of the differences in the recommendations can be illustrated using 2% lidocaine, 1:100,000 epinephrine as an example. Lower MRD values for lidocaine are 2 mg/lb maximum, with an absolute maximum of 300 mg per appointment.3 When calculating the MRD using these lower values, a healthy 150-pound individual may receive eight cartridges (14.4 ml; 288 mg) of 2% lidocaine, 1:100,000 epinephrine.3,4 The established FDA recommendations are 3.2 mg/lb, with an absolute maximum of 500 mg per appointment.2 When using these values for the same hypothetical patient, the result is significantly different: a maximum of 13 cartridges of the local anesthetic drug (23.4 ml; 468 mg) may be administered.2 However, the actual maximum number of cartridges that may be safely administered to the patient in this example is not 13; rather, the MRD is limited by the epinephrine content to 11 cartridges (19.8 ml; 396 mg).4 When using the lower values, the drug that limits the maximum number of cartridges is the local anesthetic drug (in the first example of eight cartridges: 8 < 11). When using the FDA values, the limiting drug is epinephrine (using the second example of 11 cartridges; 11 < 13).
The use of articaine requires important considerations and modifications. The mg/lb MRD for articaine is the same in both sets of recommendations. Through at least 2013, product information leaflets for 4% articaine, 1:100,000 epinephrine list an absolute MRD of 500 mg, or approximately seven cartridges (12.5 ml; 500 mg).7 The current FDA recommendation lists a 3.2 mg/lb maximum, with no absolute maximum established; however, based on the epinephrine content, there is a maximum limit of 11 cartridges of 4% articaine,1:100,000 epinephrine.7
OVERDOSE POTENTIAL
Many dentists and dental hygienists have grown accustomed to calculations based on the previously published absolute maximum dose of 500 mg of articaine per appointment. When comparing calculations of the maximum number of cartridges in the absence of a published articaine limit, there is an increased potential for overdose. For example, 11 cartridges of 4% articaine, 1:100,000 epinephrine — considered a safe maximum dose guideline of epinephrine for healthy patients — contain almost 800 mg of articaine (11 x 72 mg per cartridge). Calculating the maximum dose for a 250-pound individual yields similar results (250 lbs x 3.2 mg/lb = 800 mg, or about 11 cartridges). Both results, in most conceivable clinical situations, may appear to conflict with FDA advice to limit doses to the smallest amount required to provide the needed anesthesia, and exceed pre-2013 recommendations by a considerable margin (500 mg absolute maximum versus 800 mg). In the absence of a published absolute maximum dose, many clinicians continue to observe a 500 mg absolute maximum for articaine.
In addition to concerns over central nervous system (CNS) and cardiovascular system (CVS) toxicity with increasing doses, articaine, benzocaine, prilocaine, tetracaine and lidocaine have been identified as increasing the risk of methemoglobinemia.8,9 Whether acquired or congenital, this condition’s risk rises with increasing doses.2,4,9 Although typical doses of articaine in dentistry have not resulted in methemoglobinemia,4 800 mg exceeds typical doses by a wide margin. Restricting doses to those that provide adequate anesthesia for appropriate treatment seems prudent; as such, sound clinical judgment should guide decisions in this area.
The second formulation of articaine, 4% with 1:200,000 epinephrine, deserves special mention regarding the potential for toxicity considering that many product information leaflets describe this formulation as preferred for most routine procedures. Based on current FDA guidelines, in which an absolute maximum dose for articaine is no longer provided, a 1:200,000 epinephrine dilution would allow up to 22 cartridges per appointment in a healthy patient (based on its epinephrine content). While within accepted guidelines for healthy patients, when considering epinephrine dosing, this amounts to 1584 mg of articaine (22 x 72 mg/cartridge), an excessive dose with even greater concerns of CNS toxicity, CVS toxicity and methemoglobin levels. Dentists and dental hygienists who use the smallest amounts needed to provide profound local anesthesia avoid approaching such extreme doses.
In addition to insert updates for articaine, bupivacaine product information leaflets also establish no mg/lb maximum. The current recommendation for 0.5% bupivacaine, 1:200,000 epinephrine is an absolute maximum of 90 mg. While MRD per pound or kilogram information is no longer available in the U.S., the recommendations for bupivacaine are 0.9 mg/lb and 2.0 mg/kg in Canada.2
CLOSING THOUGHTS
Although the FDA has provided MRD guidelines for local anesthetic drugs (Table 1), patient response to drug dosage is not always predictable. Two equal doses of the same anesthetic agent can produce markedly different responses, depending on the patient; for example, individuals who are less susceptible to an anesthetic may require higher-than-average doses in order to provide effective anesthesia. Consequently, the possibility of overdose is greater for these individuals. Others who do not fit easily into dose recommendations may experience effective anesthesia with less-than-typical doses. If typical doses are administered to these individuals, extensive and prolonged periods of anesthesia can result.
In addition to observing MRDs, the administration of local anesthetic drugs continues to be guided by several important factors, including the area to be anesthetized, vascularity of the tissues, technique and individual tolerances. Perhaps the most important advice to follow comes from a similarly worded statement that may be found in all local anesthetic product inserts: “The lowest dosage needed to provide effective anesthesia should be administered.”8
KEY TAKEAWAYS
- The U.S. Food and Drug Administration (FDA) regularly updates maximum recommended dosages (MRDs) for local anesthetic drugs.
- Although the FDA has provided MRD guidelines, patient response to drug dosage is not always predictable.
- In some cases, oral health professionals are guided by lower MRD values than those approved by the FDA.2–4
- There are benefits to following lower MRDs — such as enhanced patient safety — and as long as administered doses remain within FDA recommendations, there is no breach of the guidelines.
- In addition to concerns over central nervous system and cardiovascular system toxicity with increasing doses, articaine, benzocaine, prilocaine, tetracaine and lidocaine have been identified as increasing the risk of methemoglobinemia.8,9
- In addition to observing MRDs, the administration of local anesthetics continues to be guided by several factors, including the area to be anesthetized, vascularity of the tissues, technique and individual tolerances.
- Restricting doses to those that provide adequate anesthesia for treatment is prudent; sound clinical
REFERENCES
- U.S. National Library of Medicine. DailyMed. Available at: dailymed.nlm.nih.gov/dailymed/drugList.cfm?startsWith=All. Accessed October 28, 2017.
- Malamed SF. Handbook of Local Anesthesia. 6th ed. St. Louis, Mo: Elsevier Mosby; 2013.
- Malamed SF. Handbook of Local Anesthesia. 5th ed. St. Louis, Mo: Elsevier Mosby; 2004.
- Bassett K, DiMarco A, Naughton D. Local Anesthesia for Dental Professionals. Upper Saddle River, NJ: Pearson; 2009.
- Malamed SF. What’s new in local anesthesia. Dimensions of Dental Hygiene. 2013:11(7):21–22.
- American Academy of Pediatric Dentistry. Guideline on Use of Local Anesthesia for Pediatric Dental Patients 2015. Available at: aapd.org/media/policies_guidelines/g_localanesthesia.pdf. Accessed October 28, 2017.
- Drugs.com. Articaine and Epinephrine Injection. Available at: drugs.com/pro/articaine-and-epinephrine-injection.html. Accessed October 28, 2017.
- U.S. National Library of Medicine. DailyMed: 4% articaine, 1:100,000 epinephrine product insert. Available at: dailymed.nlm.nih.gov/dailymed/archives/fdaDrug Info.cfm?archiveid=28573. Accessed October 28, 2017.
- Durieux ME, Gin TG, Joanne MD. Methemoglobinemia related to local anesthetics: A summary of 242 episodes. Anesth Analg. 108;3:837–845.
FEATURED IMAGE BY MAMINAVA_KATERINA/ISTOCK/GETTY IMAGES PLUS
The authors have no commercial conflicts of interest to disclose.
From Decisions in Dentistry. December 2017;3(12):26—29.