Transmucosal Drug Delivery in the Oral Cavity
Offering advantages over conventional methods, transmucosal drug delivery presents an alternative to the traditional enteral, intravenous or intramuscular routes.
This course was published in the December 2018 issue and expires December 2021. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Describe oral transmucosal drug delivery, and the specific tissues that are the primary focus of this transmission route.
- List the advantages and limitations of this method of drug administration.
- Explain various forms of oral transmucosal delivery, and clinical applications for this approach.
Transmucosal delivery offers several advantages over conventional oral drug delivery. These include avoiding harsh acids and enzymes encountered during digestion;1 the agent also circumvents the first-pass metabolism by the liver, and pre-systemic elimination that occurs in the gastrointestinal tract.2 Additionally, transmucosal administration presents an alternative when enteral delivery is not available or impossible (e.g., in patients with nausea, vomiting or intestinal failure, and some pediatric patients).3 It is also less invasive than traditional intravenous or intramuscular routes.3 This method does not require technical equipment (e.g., pumps, catheters and fluids) or the expertise that intravenous delivery requires. It is thus more cost effective. This is beneficial in dentistry because most general practitioners do not have the technical skills or equipment for starting IV lines and administering drugs in this fashion.3
Another consideration with transmucosal delivery is that certain agents have an onset of action similar to intravenous drugs.3 One limitation of this method is the variability in absorption, as the oral mucosa is an effective barrier to certain drugs breaching into the environment. Consequently, only high-potency drugs are good candidates for this delivery method. Certain disease states, such as mucositis or blisters, may affect the integrity of the oral mucosa, and when conditions like this are present, transmucosal drug administration may not be appropriate.3 This paper will explore key properties of the oral cavity, various methods of delivery, and prospects for dental applications.
MUCOSA OF THE ORAL CAVITY
As noted, the oral cavity consists of various structures (Figure 1), such as the oral mucosa or lining within the oral cavity, which includes sublingual, buccal, labial, palatal and gingival tissues.3 Compared to the skin or gastrointestinal tract, the surface area of the oral mucosa is small, yet it is highly vascularized — and this allows agents direct access to systemic circulation via venous and capillary systems.3 The sublingual and buccal gingiva are the primary focus for transmucosal drug delivery because they are the most permeable.3 Intraoral mucosa possess different permeability based on cellular thickness and the quantity of keratinized epithelium.4
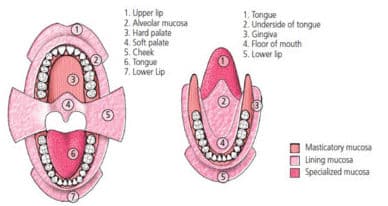
Sublingual Mucosa: The structure of the sublingual mucosa is similar to the buccal mucosa. Like the buccal mucosa, the sublingual mucosa is nonkeratinized. One distinguishing feature is the epithelial thickness: The sublingual mucosa is 100 to 200 µm, compared to the buccal’s 500 to 600 µm.1 The thinness of the sublingual mucosa makes it relatively more permeable,1 and in terms of blood flow, the sublingual mucosa receives less than the buccal mucosa.
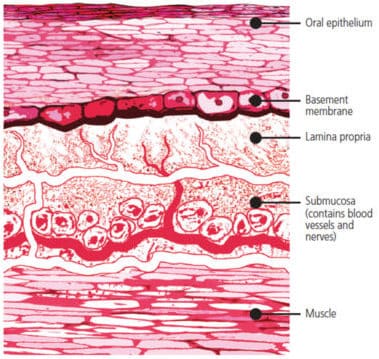
Buccal Mucosa: This mucosa demarcates the inside lining of the cheek, and divides the area between the gingiva and upper and lower lips (Figure 2).1 It protects the underlying tissues from chemical and mechanical damage,1 and also prevents foreign substances from entering. It consists of a multilayer structure,1 with outer epithelium and basement membrane supported by connective tissue consisting of lamina propria and submucosa.1 The epithelium is classified as nonkeratinized stratified squamous,4 and buccal epithelial cells are replaced every five to seven days.1
Gingival and Palatal Tissues: The tissues that make up the gingiva and palate are keratinized, and have a thickness of 250 and 200 µm, respectively.1 They are less permeable than the buccal and sublingual tissues. At this point, it should be noted that saliva plays a crucial role in the success of transmucosal drug delivery. Saliva is a water-rich environment with a mucus component. The major composition of mucus is mucin, which is mainly composed of glycoprotein. Salivary flow helps determine the amount of time a drug is released into the oral cavity, thus, “saliva washout” can occur, depending on the flow rate.1 Little research has been done to see if this concept affects the efficiency of oral transmucosal drug delivery.2
PATCHES AND OTHER DELIVERY VEHICLES
In dentistry, transmucosal drug delivery is mostly utilized in the patch form. In fact, adhesive patches have been developed to overcome the disadvantages of other forms of delivery. Advantages of the patch include rapid onset, sustained release, and a rapid decrease in the drug when the patch is released. The tissues in the oral cavity are immobile, and this makes it an ideal location for this delivery system.2 In the oral cavity, patches can be used to administer drugs systemically (by means of the veins and capillaries) or locally.2
Lozenges are an example of solid-form delivery, and many drugs are available for this route of administration, including nitroglycerin, fentanyl and prochlorperazine.3 Although taste can present a barrier to compliance,3 this is a simple form for patients to use. The lozenge dissolves and coats the oral cavity and top one-third of the esophagus. While the dissolving rate depends on how hard the patient sucks,3 on average, it takes 30 minutes for the lozenge to dissolve, and this short time frame limits how much drug can be delivered.
Chewing gum is one of the more recent and convenient forms of transmucosal drug delivery. Gum offers an advantage over lozenges, as there is a potential for a more controlled release of the agent.3 As a result, there is a possibility of release over a longer time span.
EVALUATION OF TRANSMUCOSAL DRUG DELIVERY
In the late 1960s, researchers developed a method for measuring oral absorption known as the “swirl and spit” test,1 during which subjects are given a known volume of a specific drug concentration and asked to swirl it for an allotted time. Next, the subject expectorates the drug and rinses with a specific volume of a buffer solution. The expelled drug solution and rinse are analyzed for drug content.1 The difference between the initial and final drug concentration in the solution should be the amount of drug that was absorbed.
This testing methodology poses limitations, however. Saliva is constantly being produced, and this can cause drug dilution, as well an unintentional swallowing. Another issue is the lack of control over where absorption takes place within the oral cavity; in essence, the absorption happens all over. In addition, disappearance from the oral cavity does not necessarily correlate with systemic absorption — an important consideration when evaluating this method.1
The oral mucosa of pigs has been shown to be similar histologically to human oral mucosa.1 Although the porcine floor of mouth has been shown to be more permeable than human tissues, the buccal mucosa is similar in both.1 Drugs that have been studied range from pain medications and β-blockers to hormones and sedatives;1 also worth noting is that most porcine studies have been in vitro.
Nielsen and Rassing5 conducted in vitro studies with cadaver tissues or biopsied human tissues to evaluate buccal absorption of β-blockers and testosterone. The authors concluded the cell culture model TR146 they utilized is appropriate for the buccal mucosa to demonstrate permeability using drugs with different lipophilicity.
The cell lineage known as TR146 was created from buccal mucosa of a squamous cell carcinoma and was originally developed as an in vitro model for buccal mucosa.1 The barrier function of this cell lineage was noted to be less than that of porcine or human tissues.1 Some similarities between TR146 and porcine mucosa have been reported during in vitro research utilizing nicotine.6 More recently, metoprolol has shown similarities in porcine buccal mucosa and TR146, as noted by teams conducting in vitro and in vivo porcine studies.1 These discoveries suggest cell culture models may be an innovative approach in the development of transmucosal drug delivery.1,5
CURRENT USAGE
Buccal and sublingual drug formulations have been created and are being used in some dental applications. Midazolam, a popular drug used for sedation, can be administered via an oral mucosal solution. A 2002 analysis demonstrated high bioavailability (74.5%) and consistent plasma concentration following the buccal administration of 5 mg midazolam.3 In 2011, investigators observed that within five to 10 minutes of sublingual administration of 10 mg midazolam, electroencephalogram changes were seen, making this formulation effective for use in treating seizures,2 as well as for anxiolysis during dental care. Clinical studies have shown buccal midazolam is superior or equally effective in seizure relief as rectal diazepam.7
Triazolam, a benzodiazepine used in dentistry for sedation, has also been evaluated. This medication can be given sublingually or orally. Compared to oral administration, transmucosal sublingual administration has been shown to have an earlier and higher peak concentration. In addition, a 2011 study showed that 0.25 mg triazolam administered sublingually was more effective than oral administration in reducing anxiety and pain.2
As noted, transmucosal drug delivery can either be local or systemic; in the examples above, drugs were administered buccally and sublingually with a systemic effect. An important drug in dentistry, lidocaine is an example of an agent that can be used locally, and a novel patch device is available for transmucosal delivery2 to provide mucosal and gingival anesthesia.
A study by Perry et al8 compared the lidocaine patch to a placebo for pain control during scaling and root planing. In this study, the patches were applied following the manufacturer’s instructions, with the largest transfer of anesthesia to mucosal tissues taking place within 15 minutes. According to the manufacturer, the working time is approximately 45 minutes.8 Following treatment, the subjects’ pain perception was assessed using a visual analogue pain scale. The authors concluded that lidocaine patches provide sufficient anesthesia for quadrant scaling and root planing.8
Anxiety over injection pain is common among dental patients, and Carr and Horton9 note that lidocaine patch delivery is effective in relieving pain and discomfort caused by needle sticks. Hersch et al10 reported similar findings in an earlier study, which suggests this approach may be beneficial for this patient population.
Subsequent research by Hersch et al11 tested benzocaine gel delivery in two concentrations (10% and 20%) to evaluate efficacy and tolerability in relieving acute toothaches. The team reported that while both concentrations provided temporary relief, the 20% formulation was most effective.
CONCLUSIONS
Giannola et al12 proposed that transmucosal drug delivery should be considered a slow intravenous infusion, with the drug allowed to enter systemic circulation through the capillaries. This allows drugs to have higher bioavailability.1 The transmucosal route is noninvasive and does not require specialized equipment or technical skills. The oral cavity can be utilized for transmucosal delivery, and the challenges of limited surface area and salivary action can be overcome with efficient agents and appropriate administration.1
Transmucosal drug delivery may allow dental teams to offer minimal sedation and more comfortable local injections. It also requires less equipment than some delivery methods, making it a cost-effective alternative. Yet, while there are advantages to this modality, there are also restrictions to developing drugs for transmucosal delivery. Economic motivations drive drug development, and if the only benefit companies perceive is higher bioavailability, it may not be enough incentive to incur the expense of drug creation and approval. Developers need to see a clinical void.
Toward this goal, drug companies, researchers and oral health professionals may wish to consider transmucosal delivery as a viable approach, and look for innovative ways to make dental therapies as safe and comfortable as possible. For example, iontophoresis is a noninvasive delivery system that utilizes electric current to assist in the release of medicaments.13 Long used in medicine for transdermal drug delivery, investigators have also examined if this technique could be used in the buccal mucosa for deep anesthesia with lidocaine. The researchers reported this approach “can serve as a needle-free strategy to speed the onset and prolong the duration of buccal anesthesia.”13
Compared to conventional oral drug delivery, the transmucosal route avoids harsh acids and enzymes encountered during digestion.1 It also circumvents the first-pass metabolism by the liver, and pre-systemic elimination that occurs in the gastrointestinal tract.2 While it should be noted that current research is limited and professional consensus on best practice has not yet been achieved, oral transmucosal delivery presents an alternative when enteral delivery is not available; it is also less invasive than traditional intravenous or intramuscular administration.3 In addition to use in general dentistry, this approach holds promise for many specialties, including pediatric and periodontal practice.
REFERENCES
- Sattar M, Sayed OM, Lane ME. Oral transmucosal drug delivery — current status and future prospects. Int J Pharm. 2014;471:498–505.
- Patel VF, Liu F, Brown MB. Advances in oral transmucosal drug delivery. J Control Release. 2011;153:106–116.
- Zhang H, Zhang J, Streisand JB. Oral mucosal drug delivery — clinical pharmacokinetics and therapeutic applications. Clin Pharmacokinet. 2002;41:661–680.
- Satheesh Madhav NV, Semwal R, Semwal DK, Semwal RB. Recent trends in oral transmucosal drug delivery systems: an emphasis on the soft palatal route. Expert Opin Drug Deliv. 2012;9:629–647.
- Nielsen HM, Rassing MR. TR146 cells grown on filters as a model of human buccal epithelium: IV. Permeability of water, mannitol, testosterone and β-adrenoceptor antagonists. Comparison to human, monkey and porcine buccal mucosa. Int J Pharm. 2000;194:155–167.
- Nielsen HM, Rassing MR. Nicotine permeability across the buccal TR146 cell culture model and porcine buccal mucosa in vitro: effect of pH and concentration. Eur J Pharm Sci. 2002;16:151–157.
- Lam JK, Xu Y, Worsley A, Wong IC. Oral transmucosal drug delivery for pediatric use. Advanced Drug Deliv Rev. 2014;73:50–62.
- Perry DA, Gansky SA, Loomer PM. Effectiveness of a transmucosal lidocaine delivery system for local anesthesia during scaling and root planing. J Clin Periodontol. 2005;32:590–594.
- Carr MP, Horton JE. Evaluation of a transoral delivery system for topical anesthesia. J Am Dent Assoc. 2001;132:1714–1719.
- Hersch EV, Houpt MI, Cooper SA, Feldman RS, Wolff MS, Levin LM. Analgesic efficacy and safety of an intraoral lidocaine patch. J Am Dent Assoc. 1996;127:1626–1634.
- Hersch EV, Ciancio SG, Kuperstein AS, et al. An evaluation of 10 percent and 20 percent benzocaine gel in patient with acute toothaches. J Am Dent Assoc. 2013;144:517–526.
- Giannola LI, Sutera FM, De Caro V. Physical methods to promote drug delivery on mucosal tissues of the oral cavity. Expert Opin Drug Deliv. 2013;10:1449–1462.
- Cubayachi C, Couto RO, de Gaitani CM, Padrazzi V, Freitas O, Lopez RF. Needle-free buccal anesthesia using iontophoresis and amino amide salts combined in a mucoadhesive formulation. Colloidd Surf B Biointerfaces. 2015;136:1193–1201.
Featured image by SKYNESHER/E+/GETTY IMAGES PLUS
From Decisions in Dentistry. December 2018;4(12):36—39.




