
Socket Augmentation Using Polytetrafluoroethylene Membranes
Utilizing these materials for socket augmentation can help maintain alveolar ridge width, increase keratinized gingiva and minimize bone remodeling.
Significant alveolar ridge deformation is expected after performing a tooth extraction.1,2 Several classifications systems have been developed to aid clinicians in evidence-based management when treating residual alveolar ridge deformities.3–7 With the advent of implant therapy, socket augmentation procedures (SAPs) are commonly indicated to minimize physiologic alveolar bone remodeling following a tooth extraction and to prepare an edentulous ridge for the future implant.
Within the literature, myriad SAP studies have advocated the use of various bone grafting materials (e.g., autografts, allografts, xenografts and alloplasts), cell-occlusive barrier membranes (absorbable and nonresorbable), and biologic agents (enamel matrix derivatives, platelet-derived growth factor, bone morphogenetic protein-2 and others). These biomaterials are meant to serve as space maintainers, to provide epithelial exclusion and wound stabilization, and to promote new bone formation. A clear understanding of the healing pattern of an extraction socket and post-extraction socket morphology will help clinicians choose the ideal biomaterial for a specific case.
A careful clinical and radiographic assessment of bone morphology, including a tentative treatment plan, should be obtained before the extraction. When indicated, minimally traumatic and less invasive approaches are recommended during extractions in order to preserve as much periodontal structure as possible. Numerous variables (such as buccal plate thickness, extracted tooth locations, tissue biotypes and existing infection) appear to play a crucial role in decision-making for socket remodeling — and thus determine the timing and technique for implant placement.8–10
Elian et al developed a simple socket classification based on the soft tissue relation to the cementoenamel junction (CEJ) and presence (or absence) of the buccal plate.11 Type I sockets represented sites in which soft and hard tissues are at normal levels in regard to the CEJ, and remain intact following the extraction. Sockets with buccal dehiscences — with or without soft tissue recession — are described as type II and III sockets, respectively. Subsequently, a subclassification of Type II sockets was proposed that takes into consideration the extent of the missing buccal plate in an attempt to prevent soft tissue collapse by immediate provisionalization at the time of extraction.12
Highly predictable outcomes are expected following the treatment of Type I sockets — and, in some situations featuring thick facial bony plates, socket augmentation may not be necessary. Conversely, more challenging clinical scenarios can arise from type II and III sockets due to the possibility of significant bone resorption, which has the potential to compromise implant esthetics. In addition to SAPs, additional soft tissue grafting and advanced bone grafting procedures — such as guided bone regeneration (GBR) — could be indicated to further enhance implant esthetics, especially when treating type II and III sockets.
INDICATIONS FOR CELL-OCCLUSIVE BARRIER MEMBRANES
Since the introduction of guided tissue/bone regeneration,13,14 absorbable (e.g., collagen, polylactic/polyglycolic acid, human amnion/chorion tissue, and human dermal/pericardium tissue) and nonresorbable membranes (e.g., expanded polytetrafluoroethylene or e-PTFE, high-density polytetrafluoroethylene or d-PTFE, millipore filters and titanium mesh) were developed to assist periodontal regeneration in the treatment of intrabony defects,15–17 root coverage procedures18–20 and guide bone regeneration.14 Based on the principle proposed by Melcher,21 distinct cellular phenotypes may be responsible for various healing patterns within an extraction socket. Due to the increased mitotic activity and faster migration of epithelial cells into denuded or deepithelized surfaces,22,23 exclusion of these nonosteogenic progenitors cells is often desired to allow cells with osteogenic properties to colonize the extraction socket, and, thus, maximize the potential for healing. In this manner, barrier membranes — such as e- or d-PTFE cell-occlusive devices — can greatly enhance bone formation during SAPs.
Limited to case series, earlier studies evaluated the use of e-PTFE membranes (with or without a combination of bone grafting materials) for SAPs and implant therapy.24–27 Doblin et al confirmed the formation of viable and denser new bone through histologic evidence when e-PTFE membrane was combined with demineralized, freeze-dried bone allograft after three, six and nine months.25 Lekovic et al measured the dimensional changes of untreated and treated sockets by the addition of an e-PTFE membrane.24 Favorable results were reported in the treated sockets — however, cases with early membrane exposure had comparable dimensions to their untreated-socket counterparts. Consequently, early exposure can lead to bacterial colonization due to the membrane porous topography, and, thus, hinder the effectiveness of regenerative procedures.27–30
The use of absorbable membranes has been advocated to eliminate surgical reentry to retrieve nonresorbable membranes. Lekovic et al tested SAPs using glycolide and lactide polymers-based membranes in an attempt to find an alternative to the negative outcomes of e-PTFE membranes. Small dimensional changes were observed when compared to control sockets. In addition, similar outcomes were observed with the use of bioabsorbable membranes that eliminate the need for a second surgery and minimize risk for infection.31
Despite technical advances designed to increase the longevity and resistance to enzymatic degradation by cross-linking mechanisms, absorbable membranes still require primary wound closure to be effective. The use of absorbable or nonresorbable e-PTFE membranes presents surgical complexities, however, including a reduction of keratinized gingiva and tissue thickness, and the resulting disruption of the soft tissue architecture.32,33
d-PTFE MEMBRANES FOR SOCKET AUGMENTATION
In order to address these limitations, the use of d-PTFE membranes was proposed as an alternative to absorbable and e-PTFE membranes. The high density and submicron (0.2µm) porosity of d-PTFE reduces the risk for bacterial contamination of the undermining tissues and biomaterials.32,34–39 Table 1 lists the components, features and commercial names of relevant d-PTFE membranes.
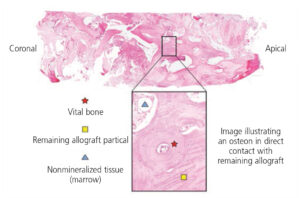
Based on animal models and human case reports, Bartee et al provided the first clinical and histologic evidence of d-PTFE membranes utility in socket augmentation.38,39 This research noted new, dense lamellar bone formation within the grafted materials (e.g., demineralized freeze-dried bone allografts, hydroxyapatite and calcium phosphate), with well-defined vascularization. In addition, no inflammatory infiltrate was reported, and the remaining graft materials were in a state of dissolution, resorption and/or remodeling.39,40
 Hoffmann et al provided predictable outcomes in SAPs in conjunction with d-PTFE membranes in a private-practice-based, retrospective, nonrandomized study from a total of 276 sockets after 12 months.32 The regeneration and healing patterns of newly formed tissues from their core samples were comparable to fully healed nongrafted sockets from other studies.41 No adverse tissue reactions or inflammatory changes were noted during the study, regardless of known detrimental variables, such as smoking.
Hoffmann et al provided predictable outcomes in SAPs in conjunction with d-PTFE membranes in a private-practice-based, retrospective, nonrandomized study from a total of 276 sockets after 12 months.32 The regeneration and healing patterns of newly formed tissues from their core samples were comparable to fully healed nongrafted sockets from other studies.41 No adverse tissue reactions or inflammatory changes were noted during the study, regardless of known detrimental variables, such as smoking.
Generally, regenerative procedures require primary closure to avoid exposure of grafting materials, but this can lead poor treatment outcomes. Although socket augmentation is often performed with a minimally invasive approach, primary closure is not always achieved. Compared to absorbable membranes or e-PTFE materials, the surface characteristics of d-PTFE membranes provide an advantage that contributes to more predictable outcomes.32
A dual-layer socket augmentation approach using d-PTFE membranes over those derived from collagen has been proposed to enhance bone formation from levels seen with single-layer membranes.34,42 In an animal study using the dual-layer technique, four months after the procedure, significant increases in alveolar bone volume, vertical height and ridge width were reported.42 In a pilot randomized control trial in humans, however, SAP performed with single- or dual-layer modalities did not show either approach being superior in terms of clinical and volumetric results.43

Key Takeaways
- An evaluation of the socket morphology following tooth extraction will guide the decision-making process for socket augmentation procedures (SAPs).
- By minimizing the physiologic alveolar bone remodeling after tooth extraction, SAPs reduce the potential need for additional grafting procedures for implant placement.
- The selection of bone grafting materials can play a crucial role in the healing patterns of an extraction socket and turnover rates, and thus impact long-term alveolar ridge stability.
- During socket augmentation, barrier membranes help stabilize regenerative biomaterials and enhance wound healing, especially in intact sockets with insufficient bone width for implant placement.
- The advantages of using d-PTFE membrane include: it’s a biocompatible material, there’s no need for primary closure, it increases the zone of keratinized gingiva, alveolar ridge remodeling is minimized, and it is easily placed and removed.
Due to its less than ideal tissue adhesive properties, d-PTFE membranes should be removed during postoperative appointments to allow complete epithelial healing.44 For this reason, clinicians should avoid placing these membranes in close proximity to teeth due to the possibility of attachment loss.
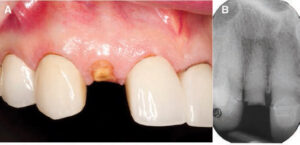
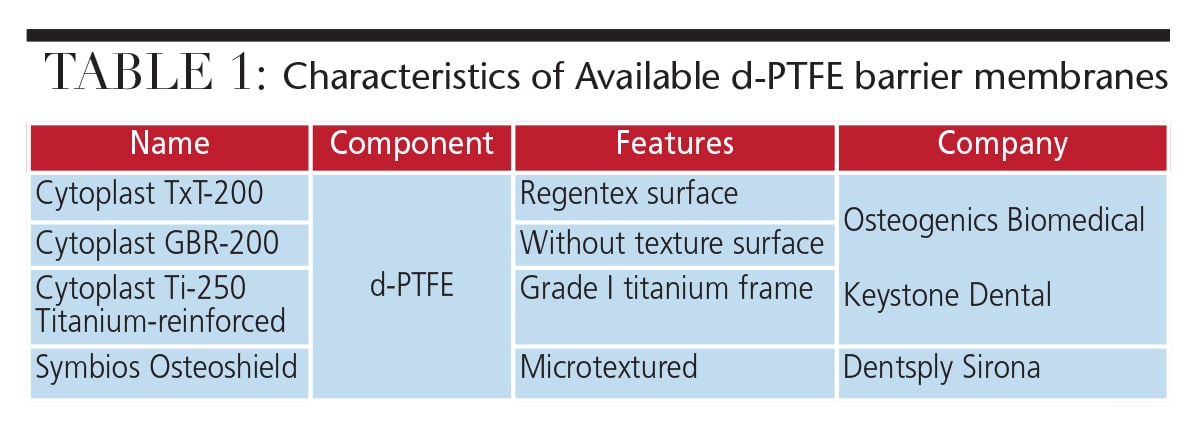
to extraction of #7 (B).
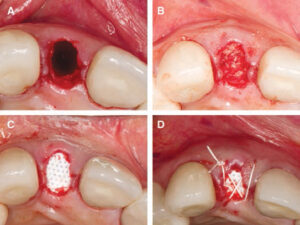
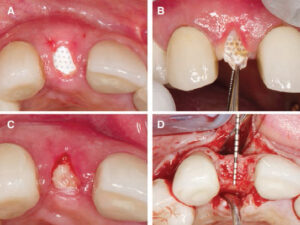
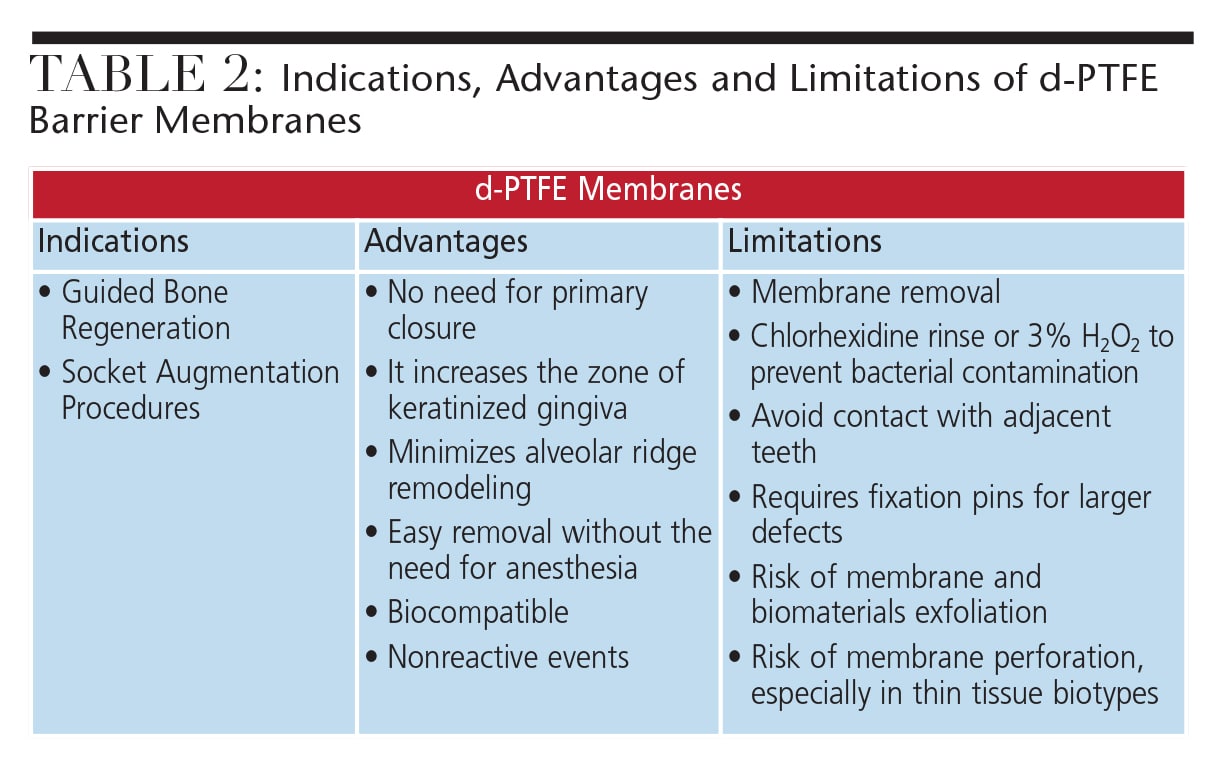
Ultimately, the cost-effectiveness of absorbable and nonresorbable membranes should be considered in light of the Arbab et al study that suggests there is no difference in outcomes between these materials.43 It is also important to note that socket morphology remains a key predictor to determine the selection of ideal materials and overall effectiveness of an SAP, and that socket augmentation might not totally eliminate the need for a GBR procedure.45,46 Hence, the patient should be advised that GBR may be needed after an SAP procedure. As a result, it is important to properly assess the surrounding bone architecture and tissue characteristics so as to select a barrier membrane that will maximize chances for a successful outcome. A summary of the indications, advantages and limitations of d-PTFE membranes is presented in Table 2. Additionally, descriptive techniques are presented in Table 3 and illustrated in Figures 1 through 6.
CONCLUSION
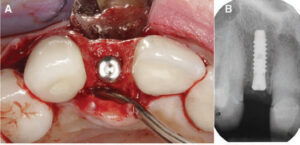
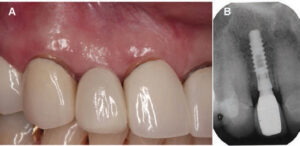
Before attempting socket augmentation, it is important that clinicians understand the fundamental nature of appropriate dental biomaterials and the biologic principles of soft tissue management. Based on the latest evidence-based review,34 the use of bone grafting materials will minimize the need for bone remodeling following an extraction.
Additionally, barrier membranes may potentially enhance outcomes by stabilizing regenerative biomaterials and promoting wound healing, especially in Type II and Type III sockets. Among the available materials, d-PTFE membranes offer many advantages. They eliminate the need for primary closure, increase the zone of keratinized gingiva, and minimize alveolar ridge remodeling. They are also easy to place and remove. Ultimately — and depending on the patient and case — d-PTFE appears to be a suitable choice for socket augmentation therapy.
References
- Schropp L, Wenzel A, Kostopoulos L, Karring T. Bone healing and soft tissue contour changes following single-tooth extraction: a clinical and radiographic 12-month prospective study. Int J Periodontics Restorative Dent. 2003;23:313–323.
- Pietrokovski J, Massler M. Alveolar ridge resorption following tooth extraction. J Prosthet Dent. 1967;17:21–27.
- Allen EP, Gainza CS, Farthing GG, Newbold DA. Improved technique for localized ridge augmentation. A report of 21 cases. J Perio. 1985;56:195–199.
- Misch CE, Judy KW. Classification of partially edentulous arches for implant dentistry. Int J Oral Implantol. 1987;4:7–13.
- Wang HL, Al-Shammari K. HVC ridge deficiency classification: a therapeutically oriented classification. Int J Periodontics Restorative Dent. 2002;22:335–343.
- Seibert JS. Reconstruction of deformed, partially edentulous ridges, using full thickness onlay grafts. Part II. Prosthetic/periodontal interrelationships. Compend Contin Educ Dent. 1983;4:549–562.
- Lekholm U, Zarb GA. Patient selection and preparation. In: Brånemark PI, Zarb GA, Albrektsson T, eds. Tissue-Integrated Prostheses: Osseointegration in Clinical Dentistry. Chicago: Quintessence; 1985:199–209.
- Garber DA, Belser UC. Restoration-driven implant placement with restoration-generated site development. Compend Contin Educ Dent. 1995;16:796,798–802,804.
- Meltzer AM. Nonresorbable-membrane-assisted bone regeneration: stabilization and the avoidance of micromovement. Dental Impl Update. 1995;6:45–48.
- Salama H, Salama M. The role of orthodontic extrusive remodeling in the enhancement of soft and hard tissue profiles prior to implant placement: a systematic approach to the management of extraction site defects. Int J Periodontics Restorative Dent. 1993;13:312–333.
- Elian N, Cho SC, Froum S, Smith RB, Tarnow DP. A simplified socket classification and repair technique. Prac Proced Aesthet Dent. 2007;19:99–104.
- Chu SJ, Sarnachiaro GO, Hochman MN, Tarnow DP. Subclassification and clinical management of extraction sockets with labial dentoalveolar dehiscence defects. Compend Contin Educ Dent. 2015;36:516,518–520,522.
- Nyman S, Lindhe J, Karring T, Rylander H. New attachment following surgical treatment of human periodontal disease. J Clin Periodontol. 1982;9:290–296.
- Dahlin C, Linde A, Gottlow J, Nyman S. Healing of bone defects by guided tissue regeneration. Plast Reconstr Surg. 1988;81:672–676.
- Becker W, Becker BE, Prichard JF, Caffesse R, Rosenberg E, Gian-Grasso J. Root isolation for new attachment procedures. A surgical and suturing method: three case reports. J Periodontal. 1987;58:819–826.
- Blumenthal N, Steinberg J. The use of collagen membrane barriers in conjunction with combined demineralized bone-collagen gel implants in human infrabony defects. J Periodontal. 1990;61:319–327.
- Gottlow J, Nyman S, Lindhe J, Karring T, Wennstrom J. New attachment formation in the human periodontium by guided tissue regeneration. Case reports. J Clin Periodontal. 1986;13:604–616.
- Cortellini P, Pini Prato GP, DeSanctis M, Baldi C, Clauser C. Guided tissue regeneration procedure in the treatment of a bone dehiscence associated with a gingival recession: a case report. Int J Periodontics Restorative Dent. 1991;11:460–467.
- Pini Prato G, Tinti C, Vincenzi G, Magnani C, Cortellini P, Clauser C. Guided tissue regeneration versus mucogingival surgery in the treatment of human buccal gingival recession. J Periodontol. 1992;63:919–928.
- Tinti C, Vincenzi G, Cortellini P, Pini Prato G, Clauser C. Guided tissue regeneration in the treatment of human facial recession. A 12-case report. J Periodontol. 1992;63:554–560.
- Melcher AH. On the repair potential of periodontal tissues. J Periodontol. 1976;47:256-260.
- Engler WO, Ramfjord SP, Hiniker JJ. Healing following simple gingivectomy. A tritiated thymidine radioautographic study. I. Epithelialization. J Periodontol. 1966;37:298–308.
- Listgarten M. Ultrastructure of the dento-gingival junction after gingivectomy. J Periodontal Res. 1972;7:151–160.
- Lekovic V, Kenney EB, Weinlaender M, et al. A bone regenerative approach to alveolar ridge maintenance following tooth extraction. Report of 10 cases. J Periodontol. 1997;68:563–570.
- Doblin JM, Salkin LM, Mellado JR, Freedman AL, Stein MD. A histologic evaluation of localized ridge augmentation utilizing DFDBA in combination with e-PTFE membranes and stainless steel bone pins in humans. Int J Periodontics Restorative Dent. 1996;16:120–129.
- Brugnami F, Then PR, Moroi H, Leone CW. Histologic evaluation of human extraction sockets treated with demineralized freeze-dried bone allograft (DFDBA) and cell occlusive membrane. J Periodontol. 1996;67:821–825.
- Chen ST, Dahlin C. Connective tissue grafting for primary closure of extraction sockets treated with an osteopromotive membrane technique: surgical technique and clinical results. Int J Periodontics Restorative Dent. 1996;16:348–355.
- Simion M, Baldoni M, Rossi P, Zaffe D. A comparative study of the effectiveness of e-PTFE membranes with and without early exposure during the healing period. Int J Periodontics Restorative Dent. 1994;14:166–180.
- Wikesjo UM, Lim WH, Thomson RC, Hardwick WR. Periodontal repair in dogs: gingival tissue occlusion, a critical requirement for GTR? J Clin Periodontol. 2003;30:655–664.
- Simion M, Trisi P, Maglione M, Piattelli A. A preliminary report on a method for studying the permeability of expanded polytetrafluoroethylene membrane to bacteria in vitro: a scanning electron microscopic and histological study. J Periodontol. 1994;65:755–761.
- Lekovic V, Camargo PM, Klokkevold PR, et al. Preservation of alveolar bone in extraction sockets using bioabsorbable membranes. J Periodontol. 1998;69:1044–1049.
- Hoffmann O, Bartee BK, Beaumont C, Kasaj A, Deli G, Zafiropoulos GG. Alveolar bone preservation in extraction sockets using nonresorbable dPTFE membranes: a retrospective non-randomized study. J Periodontol. 2008;79:1355–1369.
- Iasella JM, Greenwell H, Miller RL, et al. Ridge preservation with freeze-dried bone allograft and a collagen membrane compared to extraction alone for implant site development: a clinical and histologic study in humans. J Periodontol. 2003;74:990–999.
- Avila-Ortiz G, Elangovan S, Kramer KW, Blanchette D, Dawson DV. Effect of alveolar ridge preservation after tooth extraction: a systematic review and meta-analysis. J Dent Res. 2014;93:950–958.
- Barboza EP, Stutz B, Ferreira VF, Carvalho W. Guided bone regeneration using nonexpanded polytetra-fluoroethylene membranes in preparation for dental implant placements — a report of 420 cases. Implant Dent. 2010;19:2–7.
- Fotek PD, Neiva RF, Wang HL. Comparison of dermal matrix and polytetrafluoroethylene membrane for socket bone augmentation: a clinical and histologic study. J Periodontol. 2009;80:776–785.
- Barber HD, Lignelli J, Smith BM, Bartee BK. Using a dense PTFE membrane without primary closure to achieve bone and tissue regeneration. J Oral Maxillofac Surg. 2007;65:748–752.
- Bartee BK. The use of high-density polytetra-fluoroethylene membrane to treat osseous defects: clinical reports. Implant Dent. 1995;4:21–26.
- Bartee BK, Carr JA. Evaluation of a high-density polytetrafluoroethylene (n-PTFE) membrane as a barrier material to facilitate guided bone regeneration in the rat mandible. J Oral Implantol. 1995;21:88–95.
- Bartee BK. Evaluation of a new polytetrafluoroethylene guided tissue regeneration membrane in healing extraction sites. Compend Contin Educ Dent. 1998;19:1256–1258,1260,1262–1254.
- Irinakis T. Rationale for socket preservation after extraction of a single-rooted tooth when planning for future implant placement. J Can Dent Assoc. 2006;72:917–922.
- Al-Hezaimi K, Rudek I, Al-Hamdan KS, Javed F, Nooh N, Wang HL. Efficacy of using a dual layer of membrane (dPTFE placed over collagen) for ridge preservation in fresh extraction sites: a micro-computed tomographic study in dogs. Clin Oral Implants Res. 2013;24:1152–1157.
- Arbab H, Greenwell H, Hill M, et al. Ridge preservation comparing a nonresorbable PTFE membrane to a resorbable collagen membrane: A clinical and histologic study in humans. Implant Dent. 2016;25:128–134.
- Park SH, Brooks SL, Oh TJ, Wang HL. Effect of ridge morphology on guided bone regeneration outcome: conventional tomographic study. J Periodontol. 2009;80:1231–1236.
- Mardas N, Trullenque-Eriksson A, MacBeth N, Petrie A, Donos N. Does ridge preservation following tooth extraction improve implant treatment outcomes: a systematic review: Group 4: Therapeutic concepts & methods. Clin Oral Implants Res. 2015;26 (Suppl 11):180–201.
- Urban IA, Monje A, Wang HL. Vertical ridge augmentation and soft tissue reconstruction of the anterior atrophic maxillae: A case series. Int J Periodontics Restorative Dent. 2015;35:613–623.
From Decisions in Dentistry. September 2016;2(09):14–16,18,21.


