
Screening for Skin Cancer
Due to their clinical proximity to areas often subject to skin lesions, oral health professionals have an opportunity to identify malignant growths and refer accordingly.
This course was published in the January 2018 issue and expires January 2021. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
OBJECTIVES
- Describe various types of skin cancers, their prevalence, and risk factors.
- Explain nonsurgical and surgical treatment options.
- List ways that skin cancer screening can be incorporated into dental practice.
In the United States, basal cell carcinoma (BCC) is the most common type of nonmelanoma skin cancer (NMSC), followed by squamous cell carcinoma (SCC).2 In the U.S. alone, the American Cancer Society estimates more than 5 million BCC and SCC are diagnosed each year, with many individuals developing multiple lesions over time.2 Currently the fifth most common cancer in men and sixth most common in women,3 malignant melanoma (MM) is a reportable cancer in the U.S. National Program of Cancer Registries; therefore, there are more reliable statistics than with NMSC.4 An estimated 87,110 new cases of MM were diagnosed in the U.S. in 2017, of which an estimated 9730 proved fatal.3 Although BCC and SCC are more common, rates are difficult to track because practitioners are not required to report cases to national cancer registries.
Skin cancers vary in clinical appearance, but most often are first identified visually, thus providing an opportunity for screening in the dental setting. This paper will help oral health professionals develop the working knowledge and confidence to examine for — and identify — various forms of skin cancers.
In humans, small amounts of ultraviolet (UV) radiation are essential for production of vitamin D; however, excessive radiation is considered the main cause of skin cancers. Exposure to UV radiation, both natural and artificial, damages the deoxyribonucleic acid (DNA) inside normal skin cells that holds the code for the way these cells grow. About 90% of NMSC are associated with exposure to UV radiation from the sun.5 Shortwave UV-B rays penetrate the top layers of skin, while long-wave UV-A rays penetrate to the deeper layers; however, each can produce genetic mutations that may lead to skin cancer. Both the U.S. Department of Health and Human Services and World Health Organization have identified chronic exposure to UV radiation as a human carcinogen.5,6
Human skin consists of three basic layers — epidermis, dermis and hypodermis — and several kinds of cells.7 The outer epidermis layer contains flat cells (squamous cells), round cells (basal cells) and melanocytes, which are the skin cells responsible for producing melanin, the pigment that darkens the skin. Some individuals develop white spots on the skin surface, which are essentially areas of depigmentation caused by increased destruction or deactivation of melanocytes. The middle dermis layer contains collagen, elastic tissue and reticular fibers situated between the epidermis and bottom layer of subcutaneous tissues. It is within these layers that skin cancers differentiate themselves. Skin cancer is defined as the uncontrolled growth of abnormal skin cells that start to change as a result of cumulative UV exposure, either natural or artificial.
DIFFERENTIAL DIAGNOSIS
Skin cancers fall into two major categories: MM or NMSC. The nonmelanoma group is further classified into two main types: BCC and SCC. Unlike MM, NMSC forms from skin cells other than melanocytes. Tending to be less aggressive, BCC often resembles other skin conditions — such as psoriasis or eczema — and, consequently, may go undetected. More prevalent than SCC, BCC accounts for the majority of NMSC; furthermore, up to 50% of individuals who have been diagnosed with BCC will develop a new lesion within five years.1,2 Approximately 80% of NMSC fall into the BCC category.1,2 Although MM accounts for less than 1% of diagnosed cases, it results in the vast majority of skin cancer deaths.3 While the adjunctive use of a dermatoscope may aid diagnosis, ultimately, the only true determination of a lesion can be made through histological pathologic evaluation.
ACTINIC KERATOSIS
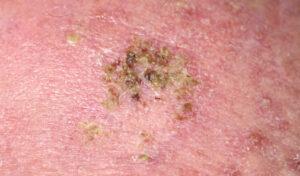
Actinic keratosis (AK) is the most common type of sun-induced precancerous skin lesion (Figure 1). Also known as solar keratosis, AK is the most frequent reason individuals seek treatment.1 Found in older adults after years of chronic and cumulative sun exposure, AK is now becoming more common in individuals in their 20s and 30s. These lesions may initially be subclinical, but clinical symptoms appear over time. The lesions can develop anywhere on the body that is exposed to UV rays, and are often found in more than one area. Visually, they may appear as dry or crusty areas on the scalp, face, ears, lips, backs of the hands and forearms. Although AKs are considered premalignant, they may progress to SCC if left untreated. As a precaution, AKs are routinely treated since it is difficult to determine which patches or lesions might turn in to cancer.
BASAL CELL CARCINOMA
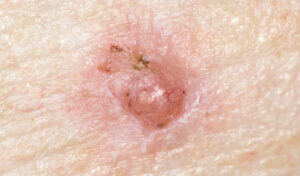
As noted, the majority of NMSC are BCC (Figure 2). These are abnormal, uncontrolled growths that begin in the basal cells that line the deepest layer of the epidermis, referred to as the basal cell layer. Slow growing and painless, BCC can vary greatly in appearance and may resemble unexplained skin problems. Often fragile, these lesions may bleed easily (such as after shaving or washing the face). Although the lesion can grow wide and deep, invading and gradually destroying surrounding soft and hard tissues, BCC is the most easily treated skin cancer and rarely metastasizes to other areas of the body. However, when BCC grows near the eyes, mouth or nose, the consequences of invasion can be serious and difficult to treat. Unfortunately, BCC has a high recurrence rate.
SQUAMOUS CELL CARCINOMA
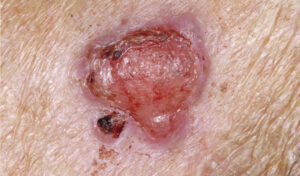
Consisting of abnormal cells arising in the squamous skin cells, located in the upper portion of the epidermis layer, SCC is the second most common type of skin cancer (Figure 3). It tends to be more aggressive than BCC, yet is relatively slow growing. Nevertheless, if left untreated, it can invade surrounding tissues and metastasize to other sites. It is most commonly seen in individuals age 50 and older, and lesions can occur anywhere on the body. Because SCC varies in appearance and can mimic other lesions, it is often mistaken for AK. Frequently, the skin in the surrounding areas reveals other signs of sun damage, such as wrinkles, age spots, loss of elasticity or freckles. Collectively, BCC and SCC make up the majority of NMSC.
MALIGNANT MELANOMA
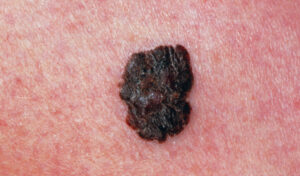
This is the least common, but most dangerous type of skin cancer (Figure 4). Originating in the pigment-producing melanocyte cells in the basal layer of the epidermis, it tends to invade locally and has a high probability of metastasis. The exact cause of MM is unclear, but genetics and prolonged exposure to the sun or tanning devices increases the risk. If MM is detected early, it is almost always curable. If it spreads and reaches vital internal organs, however, treatment is more difficult and the disease can prove fatal.
The treatment recommended depends on the stage of the lesion. There are five stages of MM (ranging from 0 to IV), escalating from early superficial lesions to those that have penetrated deeper into the tissues and metastasized. Of the estimated 160,000 Americans that were diagnosed with MM in 2017, approximately 87,000 were diagnosed with invasive (stage I, II, III or IV) MM, and another 74,000 were diagnosed with MM in situ (Stage 0).3 This skin cancer is linked with advanced age, yet is increasingly associated with younger individuals, especially females between 15 and 29 years of age who use artificial or indoor tanning devices. Though lesions vary in appearance, they often originate in or near a mole or dark spot, and may appear as mixed shades of tan, brown and black.
NONSURGICAL TREATMENT
Skin cancer treatment varies depending on the type, size, depth and location of the lesion. There are several nonsurgical modalities, including cryosurgery (freezing), topical chemotherapeutic creams or ointments, dermabrasion, chemical peeling, lasers, radiation therapy and photodynamic therapy (PDT). Topical fluorouracil creams interfere with DNA synthesis and cell proliferation, and may be used for AK and superficial BCC (sBCC) in low-risk areas. Due to its inability to penetrate deeply into the skin, topical fluorouracil is recommended only for superficial lesions. Imiquimod, which has a high cure rate, is a topical cream that modifies the skin’s immune system to stimulate the body’s rejection of precancerous cells. Patients using topical creams will experience mild to moderate skin irritation during treatment.
Nonsurgical PDT uses a photosensitizing agent that is activated by a specific type of light to kill cancer cells.8 Administered orally or parenterally, PDT is also applied topically and localized into tumor cells. The photosensitizer in the cells absorbs the light and produces an active form of oxygen that destroys cancer cells. Research by Calzavara-Pinton et al8 demonstrates that PDT yields a 50% cure rate for sBCC versus an 83% cure rate for nodular BCC.
SURGICAL TREATMENT
The two forms of complete circumferential and peripheral margin assessment (CCPMA) surgery include Mohs micrographic surgery (MMS), and standard surgical excision, coupled with margin assessment. The former may require surgical reconstruction by a plastic surgeon afterward, whereas surgical excision has the advantage of immediate surgical closure. Developed by Frederic E. Mohs in the 1930s, MMS involves the surgical removal of skin cancer, followed by immediate microscopic examination of histologic sections of the entire surgical margin of excised tissue to ensure complete removal of malignant tissues. This procedure is considered one of the best approaches for treating BCC and SCC, especially on the face, because it achieves the lowest recurrence rate with maximal preservation of uninvolved skin.9 Typically used in outpatient settings, MMS is performed under local anesthesia by a surgical team consisting of a dermatologist, histotechnician, pathologist and plastic surgeon. While smaller defects are often closed by the Mohs surgeon, a plastic surgeon, oral-maxillofacial surgeon or ear, nose and throat specialist can perform reconstruction of complex wounds. Both CCPMA techniques are widely accepted and used regularly to remove malignant lesions.
Although BCC and SCC usually do not spread to other parts of the body, skin cancer located on high-risk areas of the head and neck regions, especially the periparotid area, may require evaluation of lymph nodal basins by a head and neck oncologist, oral-maxillofacial surgeon or ear, nose and throat surgeon. Research by Kraus et al10 showed that for a small subset of individuals with regional metastasis from cutaneous SCC, survival was poor. Additionally, in cases in which MM has progressed beyond Stage II, the lymph nodes that are closest to the tumor may be involved. Currently, there are two techniques to help determine possible node involvement. Lymphoscintigraphy, a mapping technique in which a small amount of a radioactive substance is injected at the site to track drainage, and sentinel lymph node biopsy, which requires removal of some sentinel nodes. If no cancer cells are found in the sentinel nodes (i.e., the first node to receive drainage from the tumor area), it is much less likely that cancer has invaded the lymphatic system and metastasized. These specialized techniques have addressed the question of whether to perform radical lymph node dissection in the absence of clinically palpable nodes.
SKIN CANCER DETECTION APPS
With the proliferation of smartphones and mobile devices, numerous skin cancer detection applications have been developed. Apps such as SkinVision, Skin Checker, MoleScope, Mole Mapper and FotoSkin provide guidance on how patients can check skin lesions and moles. The user can take photos of any suspicious area and the app will assess them. In a study by Maier et al,11 researchers found that in an analysis of 195 photos of skin lesions taken with the SkinVision app, 81% of MM lesions were assessed accurately, with 73% sensitivity and 83% specificity. However, the study revealed that clinical diagnoses through dermatologists presented 88% sensitivity and 97% specificity, which suggest that although the these apps have promise, they are not a substitute for professional examination.
Additionally, Wolf et al12 compared the diagnostic accuracy of four smartphone apps for MM detection; three apps used algorithms to assess lesions, and one apps sent photos to a dermatologist for assessment. Data revealed the performance of smartphone apps in assessing MM risk was highly variable, with the three apps using algorithms incorrectly classifying 30% or more of the lesions. The app that sent the image directly to a dermatologist for analysis resulted in the highest sensitivity for MM diagnosis. Poor image quality was cited as problematic. Patients should be cautioned that reliance on skin cancer detection apps, in lieu of medical consultation, could delay diagnosis and treatment of skin cancers.
RISK MANAGEMENT
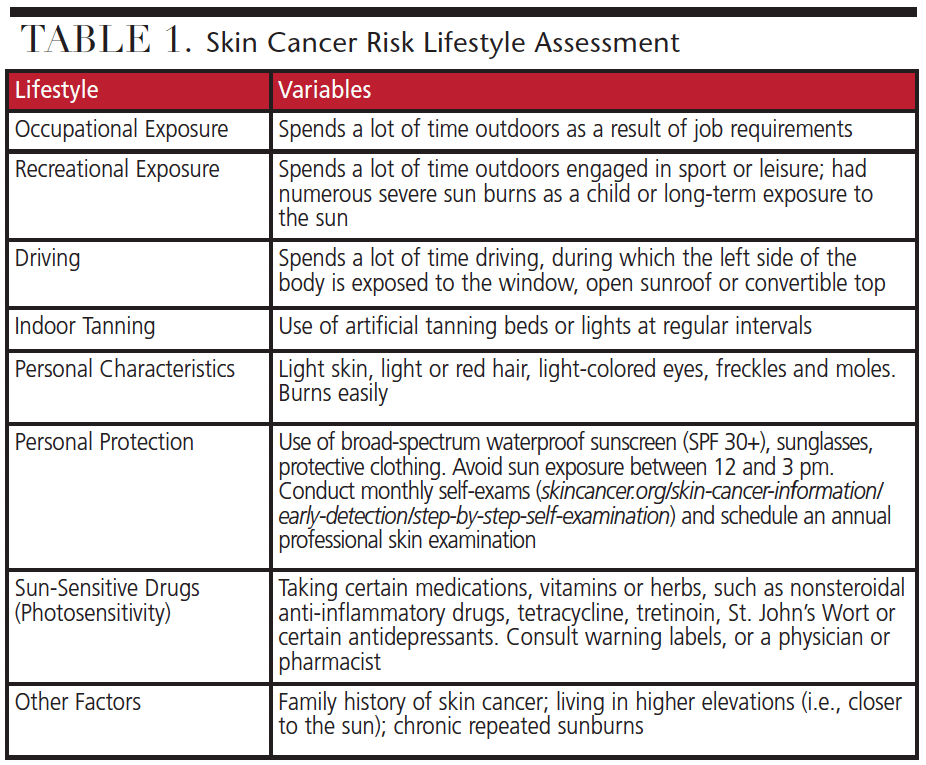
All individuals who are exposed to UV radiation are at risk for skin cancer; however, some are at greater risk. Individual risk assessment is based on numerous variables, such as family history, geographic location, occupational and recreational exposure, and personal characteristics — including eye, hair and skin color, whether or not freckles and moles are present, how the skin reacts to the sun (burn easily or tans), and use of sun protection. Individuals who work outdoors or engage in outdoor activities, or use tanning beds regularly, are especially vulnerable. Driving is also thought to be a risk factor. The Skin Cancer Foundation and American Academy of Dermatology have released skin cancer alerts that recommend drivers protect the side of their body that faces the driver’s-side window.1,7
Early detection is vital in reducing the incidence of skin cancer. Annual skin examinations by a physician, coupled with monthly self-examination, are the best ways to detect early signs of skin cancer.1 Table 1 (page 37) provides a simple risk assessment tool to help identify high-risk behaviors.
IN THE OPERATORY
Considering the three most common types of skin cancer (BBC, SCC and MM) often present on the face, head and neck areas, the notion that oral health professionals include a skin cancer examination in their routine extraoral clinical assessment is not a new concept.13–17 Because skin cancers are often first identified visually, dental teams are well positioned to educate patients, identify suspicious lesions, and arrange for further assessment. As part of Healthy People 2020, the Surgeon General’s Call to Action to Prevent Skin Cancer should serve as an advisory to dental professionals.18,19 Dentists and dental hygienists are already performing routine extraoral and intraoral examinations and are most often the clinicians who find precancerous or cancerous lesions of the head, neck and oral cavity. By adding focused, deliberate skin cancer screenings to existing extraoral examinations, practitioners can play a vital role in reducing incidences of skin cancers. The proposed role of the dental practitioner in screening should include skin cancer history and risk factor assessment, visual examination, patient education, counseling and referral to a medical professional, if indicated.
The greatest strides in combating skin cancer can come from increased awareness. Approximately 64% of the U.S. adult population sees a dentist annually, which places dental teams in a unique position to help identify skin cancers in their earliest stages.20 Just as the dental community has assumed a leadership role in reducing oral cancer, there is an opportunity to expand the traditional oral examination to include skin cancer surveillance.

CLINICAL PRESENTATIONS
Most skin cancers will present with a visible lesion that knowledgeable practitioners would be able to recognize opportunistically. As noted, skin cancers vary in appearance and symptoms; therefore, a rule of thumb is that if the lesion does not heal within a week or two, or something looks suspicious, a dermatologist should evaluate the area. Patients should be informed that change is the universal warning sign of skin cancer. It is important for practitioners to check suspicious moles using the A-B-C-D-E rule — which stands for asymmetry, border, color, diameter and evolution (Table 2, page 37). Additional symptoms of NMSC and MM skin cancers include a change in skin sensation (such as tingling, itchiness, pain or tenderness), and a spread of pigmentation outside the border of a mole or mark on the skin.
As a general guide, practitioners should be aware of the following clinical features:
- Lesions that are dissimilar on one side compared to the other
- Areas of unexplained color changes
- Flat, firm, pale or yellow areas, similar to a scar
- A thick, rough spot, crust or small eroded area
- Pink growths with raised edges and a lower area in their center that might contain abnormal blood vessels
- Small, pink or red, translucent, shiny and pearly bumps, which might have blue, brown or dark areas
- Rough or scaly red patches that might itch, crust, ooze or bleed easily
- Lesions that don’t heal within two weeks, or that heal and come back
educate, track AND DOCUMENT
Effective communication between patients and practitioners is critical to assist in the reduction of skin cancers. Take time to talk to the patient. Ask the patient if he or she would like a skin cancer assessment, and explain how the exam will be conducted and the rationale behind it. It is important that dental providers perform a thorough head and neck examination at every appointment and note any new growths. If a lesion or mole has changed or looks different from surrounding skin, the patient should be questioned. Findings should be recorded and photographed, which can be measured against future appointments. If a suspicious lesion is identified and a referral is necessary, explain why. The patient should be provided with written documentation to avoid misunderstandings, delays in treatment or lack of follow-through. Additionally, track patient referrals, which should include information on the possible condition, expected timeframe and the process. By creating awareness, educating patients, and referring to a specialist if warranted, dental teams can help ensure early diagnosis and treatment of skin cancers.
CONCLUSION
Cases of NMSC and MM are on the rise and considered an increasing public health concern. Reducing overexposure to UV radiation from the sun and indoor tanning is vital to reversing the increasing incidence rate. Skin cancers are due to many factors, but predominantly associated with individuals living longer, and chronic exposure to natural sun and artificial tanning.
As dental providers increasingly encounter patients who present with suspicious lesions, it is necessary to understand the signs, symptoms and treatment options for skin cancer. Screening and education, along with the preferences of the patient and the practitioner’s professional judgment, should be the basis for providing evidence-based care. Awareness is the first line of defense in the early detection of skin cancers. Working collaboratively with other health care professionals, dental teams can share in the responsibility for patients’ overall health.
Dentists and dental hygienists are encouraged to initiate a dialogue with each patient and make a positive impact in reducing skin cancers. In the future, it is possible that skin cancer screening will be an indispensable part of a comprehensive extraoral examination.
BONUS WEB CONTENT
DANGERS OF INDOOR TANNING
Indoor tanning is considered unsafe. 21 Many individuals express they feel better and healthier with a tan; however, tanned skin is damaged skin. The skin tans in self-defense by producing more melanin, yet, over time, this constant insult can lead to skin cancer. Tanning salons use devices that emit both UV-A and UV-B radiation. Both types of rays are harmful to the skin, whether from the sun or artificial tanning. 5 Individuals who use tanning beds have a much higher risk of getting squamous cell carnicoma (SCC), and they also tend to get SCC earlier in life.21
In 2015, the U.S. Food and Drug Administration responded to the increased number of skin and eye injuries related to use of indoor tanning by proposing new safety guidelines for the protection of consumers.22 Tanning facilities would need to put into place certain restrictions for minors and additional requirements for users age 18 and older. For instance, adults would need to sign a risk acknowledgement before their first tanning session and every six month thereafter.23 Other proposed changes would include visible warning signs and statements, limiting the amount of light allowed through protective eyewear, and installing emergency or panic button shut-off switches. At least 44 states and Washington, D.C., regulate the use of indoor tanning for minors.23
In order to help fund the Affordable Care Act, the Obama administration imposed a 10% “tanning tax” to help reduce skin cancer and increase tax revenues. 24 The American Academy of Dermatology (AAD) supported the tanning tax because of the significant health risks associated with specific indoor tanning devices. According to the AAD, indoor tanning before the age of 35, especially in young females, is linked to a 75% increase in the risk of melanoma.7 Whether these policies will remain is yet to be seen, nonetheless, this should serve as a warning to consumers that the use of indoor tanning is unsafe.
REFERENCES
- The Skin Cancer Foundation. Available at: skincancer.org. Accessed December 6, 2017.
- American Cancer Society. Available at: cancer.org/cancer/skin-cancer.html. Accessed December 6, 2017.
- Melanoma Research Foundation. Melanoma facts and statistics. Available at: melanoma.org/understand-melanoma/ what-melanoma/melanoma-facts-and-stats. Accessed December 6, 2017.
- U.S. Centers for Disease Control and Prevention. National Program of Cancer Registries (NPCR). Available at: cdc.gov/cancer/npcr/index.htm. Accessed December 6, 2017.
- U.S. Environmental Protection Agency. Health Effects of UV Radiation. Available at: epa.gov/sunsafety/health-effects-uv-radiation. Accessed December 6, 2017.
- World Health Organization. Ultraviolet radiation (UV). Skin cancers. Available at: who.int/uv/faq/skincancer/en/index1.html. Accessed December 6, 2017.
- American Academy of Dermatology. Skin cancer. Available at: aad.org/public/diseases/skin-cancer. Accessed December 6, 2017.
- Calzavara-Pinton PG, Szeimies RM, Ortel B, Zane C. Photodynamic therapy with systemic administration of photosensitizers in dermatology. J Photochem Photobiol B. 1996;36:225–231.
- American Society for Mohs Surgery. Available at: mohssurgery.org. Accessed December 6, 2017.
- Kraus DH, Carew JF, Harrison LB. Regional lymph node metastasis from cutaneous squamous cell carcinoma. Arch Otolaryngol Head Neck Surg. 1998;124:582–587.
- Maier T, Kulichova D, Scotten K, et al. Accuracy of a smartphone application using fractal image analysis of pigmented moles compared to clinical diagnosis and histological result. J Eur Acad Dermatol Venereol. 2015;29:663–667.
- Wolf JA, Moreau JF, Akilov O, et al. Diagnostic inaccuracy of smartphone applications for melanoma detection. JAMA Dermatol. 2013;149:422–426.
- Corcoran P, Depaola L, Nern K, Urquhart J. Head and neck skin cancer: dentists’ responsibility in early detection. Compend Contin Educ Dent. 2013;34:198–204.
- Main B, Felstead A, Hughes C, Thomas S. A guide to skin cancer of the face for the dental team. Dent Update. 2014;41:111–112,114–116,118.
- Sibai L, Kudsi Z. Facial skin lesions dentists should know. Dent Update. 2015;42:520–552,524.
- Steel BJ. Skin cancer-an overview for dentists. Br Dent J. 2014;216:575–581.
- The Oral Cancer Foundation. The role of dental and medical professionals. Available at: oralcancerfoundation.org/dental/role-dental-medical-professionals/. Accessed December 6, 2017.
- Office of Disease Prevention and Health Promotion. Evidence-Based Resource Summary. Healthy People 2020. Available at: healthypeople.gov/2020/tools-resources/evidence-based-resource/behavioral-counseling-prevent-skin-cancer. Accessed December 6, 2017.
- U.S. Centers for Disease Control and Prevention. The Surgeon General’s Call to Action to Prevent Skin Cancer. Available at: cdc.gov/cancer/skin/call_to_action/index.htm. Accessed December 6, 2017.
- U.S Centers for Disease Control and Prevention. National Center for Health Statistics. Oral and Dental Health. Available at: cdc.gov/nchs/fastats/dental.htm. Accessed December 6, 2017.
- U.S. Centers for Disease Control and Prevention. Indoor Tanning Is Not Safe. Available at: cdc.gov/cancer/skin/basic_info/indoor_tanning.htm. Accessed December
6, 2017. - U.S. Food and Drug Administration. FDA Proposes New Safety Measures for Indoor Tanning Devices: The Facts. Available at: www.fda.gov/ForConsumers/ConsumerUpdates/ucm350790.htm. Accessed December 6, 2017.
- National Conference of State Legislatures. Indoor Tanning Restrictions for Minors — A State-By-State Comparison. Available at: ncsl.org/research/health/indoor-tanningrestrictions.aspx. Accessed December 6, 2017.
- Internal Revenue Service. Excise Tax on Indoor Tanning Services Frequently Asked Questions. Available at: irs.gov/businesses/small-businesses-self-employed/excise-taxon-indoor-tanning-services-frequently-asked-questions. Accessed December 6, 2017.
The authors have no commercial conflicts of interest to disclose.
FEATURED IMAGE BY CREDIT/ISTOCK/GETTY IMAGES PLUS
From Decisions in Dentistry. January 2018;4(1):35-39.




