
Risk Factors in Oral Cancer Screening
A review of oral screening and oral cancer risk factors — including the old, the new, and the unusual.
This course was published in the February 2019 issue and expires February 2022. The author has no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Explain the prevalence of oral cavity and oropharyngeal squamous cell carcinoma (SSC), and the potential impact of the human papillomavirus.
- Discuss the etiology of oral cavity and oropharyngeal SSC, and associated risk factors.
- Describe clinical approaches to screening for oral cancer in the dental setting, risk management and diagnosis.
As with many cancers, the development of oral SCC is multifactorial, which means multiple contributing factors often play a role in carcinogenesis. Additionally, etiologies vary by geographic location and anatomic subsite. An older man with a long history of cigarette smoking and alcohol use is no longer the only portrait of a patient with oral SCC, as today’s research shows a much more diverse collection of risk factors.
As such, the objective of this article is to briefly review both common and unusual risk factors found in the literature for oral SCC.
TOBACCO
Tobacco is the leading cause of cancer worldwide4 and a well-established risk factor for oral SCC. The International Agency for Research on Cancer (IARC) classifies tobacco smoke as a Group 1 carcinogen, designating there is sufficient evidence of carcinogenicity.4 Tobacco smoke contains nitrosamines, arsenic, benzo[a]pyrene and benzene, which are all capable of causing cancer.1 Cigarettes, pipes, cigars and water pipes produce tobacco smoke through combustion. The prevalence of cigarette smoking in the United States is 20%.1 The risk of developing oral SCC is dose-dependent and increases the longer a person smokes.1 While cigar and pipe smoking produces higher levels of carcinogens, the risk for oral SCC is comparable to cigarettes.4
Smokeless tobacco is primarily either chewed or snuffed, with wet snuff commonly used in the United States.5 The prevalence of smokeless tobacco use in this country is 2.3%.4 The risk for smokeless tobacco use and oral SCC differs in the literature due in large part to variation in product types. Of the various products, dry snuff tends to have the highest risk for tumor development.1,4 Habitual use of smokeless tobacco creates mucosal alterations in the area of placement, along with gingival recession and tooth staining. Following habit cessation, however, the majority of users will see resolution of mucosal alterations.
The process of inhaling nicotine through vapor (via devices such as e-cigarettes) has grown in popularity; in many cases, the goal of smoking cessation is cited as a reason for use.6 The prevalence of vaping in the United States is 3.7%.4 Tobacco companies promote these noncombustible alternatives as reduced-harm products in response to the addictiveness of nicotine,7 but practitioners should be cautious of this perception. Tobacco addiction is complex and, in addition to pharmacological factors, involves social and environmental factors.7 Vaping does however, eliminate secondhand smoke.
BETEL QUID
India has the highest global incidence of oral SCC, and ethic enclaves of Asian Indians in the United States show high rates of oral SCC.8 Culturally popular products that contain betel leaf, areca nut and sometimes tobacco are contributors to this high incidence. As part of efforts to raise oral cancer awareness, dental professionals are advised these products go by a variety of names, including betel nut, betel quid, paan, paan masala and gutka. Formulations can vary and may include slaked lime, catechu and spices. The mixture is chewed and can leave a characteristic brown-red staining of the teeth and mucosa. Habitual use results mucosal pallor and fibrosis, which is not reversible with habit cessation. The IARC has designated that products containing betel quid — with or without tobacco — are carcinogenic.8 The prevalence of betel quid use in Asian Indian enclaves in the Unites States is 13.0%, with higher rates in New York City and Los Angeles.8
Betel quid is used for religious events (especially in Hinduism), holidays and celebrations. Asian Indians with higher education levels and greater household incomes have higher odds of using betel quid, presumably due to participation in social settings.8 This is in contrast with the inverse relationship with socioeconomic status and cigarette smoking in the rest of the United States.
ALCOHOL
Alcohol consumption is associated with 3.5% of cancers worldwide, and the IARC designates alcohol as a Group 1 carcinogen.4,9 However, there is no clear threshold for carcinogenesis.9 Additionally, tobacco and alcohol are a synergistic risk factor and impart a 35-fold increased risk of oral SCC,4 as alcohol allows easier penetration of the oral mucosa by tobacco-related carcinogens.10 It is hard to study alcohol as an independent risk factor for oral SCC due to concomitant use with tobacco.
Researchers have also looked at alcohol-containing mouthwash as a potential risk factor for oral SCC. The alcohol content in popular mouthwashes may exceed that of beer, wine and liquor. Several reviews have identified no overall increase in risk for oral SCC, but mention an association in long-term frequent users.11 Moreover, alcohol-containing mouthwash may be an ill-advised choice for patients dealing with alcohol addiction. Systemic effects found in patients with alcohol addiction, such as nutritional deficiencies and liver dysfunction, may also be contributing factors to oral SCC.
ULTRAVIOLET RADIATION
Ultraviolet radiation (UV) is a risk factor for skin SCC, including the lips. The classic picture of a patient with lip SCC is an elderly, fair-skinned male with an outdoor occupation. With repeated UV exposure to the lip, the vermilion border can disappear and the lip can become atrophic and ulcerated. Patients with certain genetic disorders, such as xeroderma pigmentatosum, have an increased susceptibility to skin SCC due to a decreased ability to repair DNA mutations caused by UV radiation.1 Sun protection strategies — such as avoiding exposure, and use of sunscreen and protective clothing — are recommended.
Radiation therapy is used for treating several malignancies of the head and neck, including nasopharyngeal carcinoma and oral SCC. This treatment modality imparts a risk of developing a later primary carcinoma or sarcoma. Factors that influence this incidence include radiation dose and latency after radiation.12
HUMAN PAPILLOMAVIRUS
Worldwide, 16% of cancers are related to an infectious agent,13 and human papillomavirus (HPV) has been implicated in cervical, anal, vaginal, oropharyngeal, vulvar and penile cancer.14 In the United States, HPV is the most common sexually transmitted infection, and the majority of people infected with HPV will clear the infection within two years.4,14 Transmission to the oral cavity is not fully understood, but is presumed to be related to oral sex.
Researchers have recently determined specific characteristics of HPV-related head and neck tumors, and clinicians are learning how to translate this information to oral screening and patient care. The timing of this research is critical, as oropharyngeal SCC is now the most common HPV-associated cancer.14 The first important distinction regarding HPV-associated head and neck cancer is anatomical location. The 2017 edition of the World Health Organization Classification of Head and Neck Tumours designates oropharyngeal SCC as a distinct entity from oral cavity SCC.15 While HPV does not play a major role in oral cavity SCC, it accounts for 70% of oropharyngeal SCC.14,16 These tumors occur in the lymphatic-rich tissue of the oropharynx, which includes the tonsils, soft palate, base of the tongue, and posterior pharyngeal wall.17 A second important point to consider is HPV subtype, which is classified as low-risk or high-risk based on cancer association. For example, HPV high-risk type 16 is responsible for the majority of cervical cancer and oropharyngeal SCC.4,16 Commercially available HPV vaccines protect against subtype 16.
A patient with HPV-associated oropharyngeal SCC tends to be a younger male with increased sexual activity.16 Due to the posterior location, the most common symptoms of a patient with oropharyngeal SCC are a sore throat, dysphagia and odynophagia.1 Furthermore, it is not uncommon to detect this cancer through an enlarged, stony hard and nontender positive cervical lymph node prior to visualizing a tumor in the oropharynx. This again points to the value of oral cancer screening by oral health professionals.
UNKNOWN FACTORS
A clear risk factor has not been elucidated with a certain patient profile described in the literature: The profile is a younger patient without significant tobacco and alcohol risk factors who has a tumor on the anterior two-thirds of the tongue.18–20 These tumors often demonstrate aggressive features, such as perineural infiltration.18 Studies have looked at genetic alterations, such as p53, but more research is needed.21 As part of oral screening, oral SCC should be included on a differential diagnosis for a suspicious lesion, despite a lack of traditional risk factors.
POTENTIALLY MALIGNANT ORAL DISORDERS
Many of the risk factors discussed above are associated with developing a potentially malignant disorder (PMD), which is defined as a mucosal lesion with an increased risk of developing into oral SCC.19 Table 1 demonstrates PMDs associated with each risk factor. The worldwide prevalence for PMDs is 4.47%, with leukoplakia and oral submucous fibrosis being the most common;22 however, prevalence rates for PMDs differ based on the population.
In addition, PMDs vary with respect to malignant transformation potential. For example, erythroplakia — which is less common but can accompany leukoplakia — demonstrates a higher rate of malignant transformation.22 Proliferative verrucous leukoplakia, which is a special high-risk form of leukoplakia, has one of the highest malignant transformation potentials.1 Although not classified as a PMD, an ulceration on the lateral border of the tongue or floor of the mouth that does not heal in two weeks is suspicious for a malignancy. According to the evidence-based practice guidelines recently released by the American Dental Association, PMDs and suspected malignant lesions should be biopsied immediately.19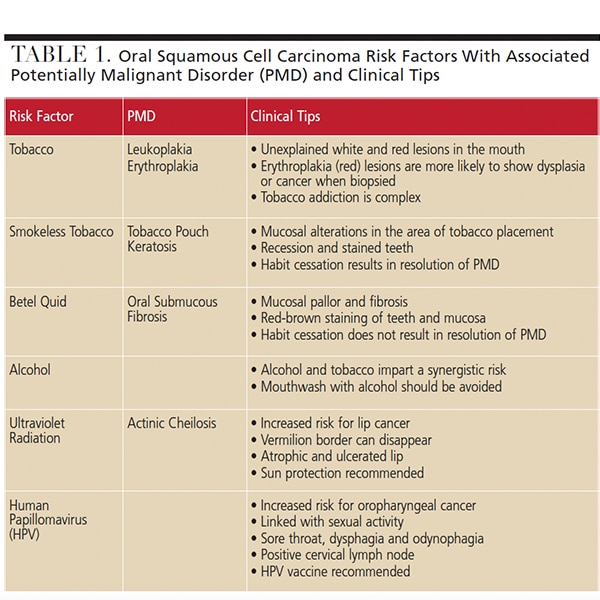
CLINICAL RESPONSIBILITIES
With the goal of raising oral cancer awareness, providers are encouraged to remain current on oral SCC risk factors, and perform oral screening of the head and neck. Along with dental team training, patient interviews and education are an essential part of efforts to identify and mitigate oral cancer risk factors.
A cross-sectional investigation evaluating oral SCC awareness demonstrates that many patients are knowledgeable about tobacco and alcohol as risk factors for oral SCC, but tend to be less knowledgeable about additional risk factors, such as HPV.23 Another cross-sectional study investigating HPV awareness among dentists and dental hygienists reported knowledge deficits.24 These studies suggest more clinically applicable HPV information should be disseminated at the provider and patient levels.
Due to their training, oral health professionals are ideally suited for oral cancer screening. In fact, dental schools routinely teach head and neck cancer examination techniques, and advocate their importance in early detection of oral SCC. By comparison, 56% of physicians report limited training on oral cancer examinations during their education.25 Additionally, 44% of medical professionals infrequently refer patients with suspicious oral lesions to specialists.25 Obviously, more needs to be done to raise oral cancer awareness throughout the health care professions. Ultimately, interprofessional collaboration and continuing education are paramount for detection of potentially malignant disorders and early diagnosis of oral cavity and oropharyngeal SCC.
REFERENCES
- Neville B, Douglas DD, Allen C, Bouquot J. Oral and Maxillofacial Pathology. 3rd ed. St. Louis, Missouri: Elsevier Health Sciences; 2015.
- American Cancer Society. Key Statistics for Oral Cavity and Oropharyngeal Cancers. Available at: www.cancer.org/ cancer/ oral-cavity-and-oropharyngeal-cancer/ about/ key-statistics.html#references. Accessed January 15, 2019.
- Shield KD, Ferlay J, Jemal A, et al. The global incidence of lip, oral cavity, and pharyngeal cancers by subsite in 2012. CA: Cancer J Clin. 2017;67:51–64.
- Cohen N, Fedewa S, Chen AY. Epidemiology and demographics of the head and neck cancer population. Oral Maxillofac Surg Clin North Am. 2018;30:381–395.
- Asthana S, Labani S, Kailash U, Sinha DN, Mehrotra R. Association of smokeless tobacco use and oral cancer: a systematic global review and meta-analysis. Nicotine Tob Res. 2018;doi.org/ 10.1093/ ntr/ nty074.
- Yeh JS, Bullen C, CH B, Glantz SA. E-cigarettes and smoking cessation. N Engl J Med. 2016;374:2172–2174.
- Elias J, Hendlin YH, Ling PM. Public versus internal conceptions of addiction: an analysis of internal Philip Morris documents. PLoS Med. 2018;1:e1002562.
- Mukherjea A, Modayil M, Tong EK. Moving toward a true depiction of tobacco behavior among Asian Indians in California: prevalence and factors associated with cultural smokeless tobacco product use. Cancer. 2018;124(Suppl 7):1607–1613.
- Amin G, Siegel M, Naimi T. National Cancer Societies and their public statements on alcohol consumption and cancer risk. Addiction. 2018;113:1802–1808.
- Kumar M, Nanavati R, Modi TG, Dobariya C. Oral cancer: Etiology and risk factors: A review. J Can Res Ther. 2016;12:458–463.
- Boffetta P, Hayes RB, Sartori S, et al. Mouthwash use and cancer of the head and neck: a pooled analysis from the International Head and Neck Cancer Epidemiology Consortium. Eur J Cancer Prevention. 2016;25:344–348.
- Fu X, Chen S, Chen W, et al. Clinical analysis of second primary gingival squamous cell carcinoma after radiotherapy. Oral Oncol. 2018;84:20–24.
- de Martel C, Ferlay J, Franceschi S, et al. Global burden of cancers attributable to infections in 2008: a review and synthetic analysis. Lancet Oncol. 2012;13:607–615.14.
- Van Dyne EA, Henley SJ, Saraiya M, Thomas CC, Markowitz LE, Benard VB. Trends in human papillomavirus-associated cancers — United States, 1999–2015. MMWR Morb Mortal Wkly Rep. 2018;67:918–924.
- WHO Classification of Head and Neck Tumours. El-Naggar AK, Chan JK, Grandis JR, Takata T, Slootweg PJ, eds. 4th ed. Lyon, France: IARC Publications; 2017:24–26.
- Westra WH. The changing face of head and neck cancer in the 21st century: The impact of HPV on the epidemiology and pathology of oral cancer. Head Neck Pathol. 2009;3:78–81.
- Westra WH, Lewis JS. Update from the 4th edition of the World Health Organization Classification of Head and Neck Tumours: oropharynx. Head Neck Pathol. 2017;11:41–47.
- Farquhar DR, Tanner AM, Masood MM, et al. Oral tongue carcinoma among young patients: An analysis of risk factors and survival. Oral Oncol. 2018;84:7–11.
- Lingen MW, Abt E, Agrawal N, et al. Evidence-based clinical practice guideline for the evaluation of potentially malignant disorders in the oral cavity: A report of the American Dental Association. J Am Dent Assoc. 2017;148;712–727.
- Tota JE, Engels EA, Madeleine MM, et al. Risk of oral tongue cancer among immunocompromised transplant recipients and human immunodeficiency virus-infected individuals in the United States. Cancer. 2018;124:2515–2522.
- dos Santos Costa SF, Brennan PA, Gomez RS, et al. Molecular basis of oral squamous cell carcinoma in young patients: Is it any different from older patients? J Oral Pathol Med. 2018;47:541–546.
- Mello FW, Miguel AF, Dutra KL, et al. Prevalence of oral potentially malignant disorders: a systematic review and meta-analysis. J Oral Pathol Med. 2018;47:633–640.
- Shimpi N, Jethwani M, Bharatkumar A, Chyou PH, Glurich I, Acharya A. Patient awareness/ knowledge toward oral cancer: a cross-sectional survey. BMC Oral Health. 2018;18:86.
- Daley EM, Thompson EL, Vamos CA, et al. HPV-related knowledge among dentists and dental hygienists. J Cancer Educ. 2016;33:1–6.
- Shimpi N, Bharatkumar A, Jethwani M, et al. Knowledgeability, attitude and behavior of primary care providers toward oral cancer: a pilot study. J Cancer Educ. 2018;33:359–364.
Featured image by GIVAGA/ISTOCK/GETTY IMAGES PLUS
From Decisions in Dentistry. February 2019;5(2):26—29.




