Recognizing Common Lesions of the Tongue
Accurately diagnosing common presentations will allow appropriate management — and help rule out malignancies.
This course was published in the March 2016 issue and expires 03/31/19. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
OBJECTIVES
After reading this course, the participant should be able to:
- Identify the most common benign lesions found on the tongue.
- Describe the etiology and clinical manifestations of each of
these lesions. - Discuss lesion diagnosis and management.
Lesions affecting the tongue represent a substantial portion of oral mucosal lesions. According to the National Health and Nutrition Examination Survey (NHANES), the prevalence of tongue lesions at any given time is 15.5%.1 Many large scale, population-based screenings have identified the most common conditions affecting the tongue, however, these were performed on specific groups and the data may vary based on the population studied and assessment method.2,3
Although the tongue is primarily composed of muscle tissue, most pathologic lesions arise from the cells of the surface epithelium, which covers the tongue, and blood vessels.2 This article describes a few of the most common benign conditions that affect the tongue.
Geographic Tongue
Commonly referred to as benign migratory glossitis, geographic tongue was first identified by French physician Pierre François Olive Rayer in 1831. He described the condition as “a wandering rash of the tongue.” The most commonly affected site is the anterior two-thirds of the dorsal tongue, although these benign red and white lesions can also present on other oral mucosal sites.4,5 Geographic tongue has a number of monikers, including lingua geographica, erythema migrans, exfoliation areata lingua, superficial migratory glossitis, lingual dystrophy, pityriases linguae, transitory benign plaques of the tongue, marginal exfoliative glossitis, ectopic geographic tongue and glossitis areata migrans. The most commonly used terms, however, are geographic tongue or benign migratory glossitis.6
The reported prevalence of geographic tongue ranges from 0.28% to 14.4%,6 although most surveys note the range between 1.0% and 2.5%.6,7 Shulman’s and Carpenter’s population-based, case-control study, which looked at data from 16,833 American adults in NHANES 1988–1994, found that geographic tongue was more prevalent among whites and blacks compared to Mexican-Americans.4 There is debate about which age group is most frequently affected. Although some investigators believe that geographic tongue is most common among children and that frequency decreases with age, others believe that it can occur at any age. Even though many reports claim no gender predilection, others suggest that the disorder is more common among women.4,6,7 In a study of 188 Thai subjects, Jainkittivong and Langlais found that women were slightly more likely to be affected than men.8
While the exact etiology of geographic tongue is unclear, multiple causes have been proposed. There are theories that suggest that geographic tongue may be associated with hormonal fluctuations or allergies, or that it could be psychosomatic.4 Geographic tongue may have a genetic cause as well.6 Dermatological conditions, such as pustular psoriasis, seborrheic dermatitis and pityriasis pilaris, have been linked to the condition. The association between geographic tongue and pustular psoriasis has been investigated most closely, with conclusions suggesting that the presence of geographic tongue in an otherwise healthy individual may indicate a greater risk of generalized psoriasis.6 Systemic conditions — such as diabetes, Reiter’s syndrome and Down syndrome — appear to have a possible correlation with geographic tongue.
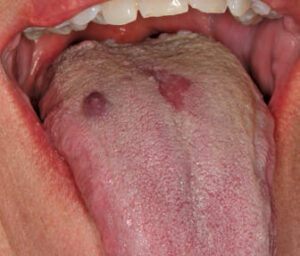
Most often, the patient is unaware of the presentation and is usually asymptomatic. The clinical appearance of geographic tongue can range from minimally evident to very prominent. During a flare up, lesions exhibit multiple red patches (ranging from 0.5 cm to 1.5 cm in diameter) due to atrophy or loss of the filiform papillae. Lesions appear as red patches surrounded by elevated, thin, white-to-yellow borders (Figure 1 and Figure 2).9 Lesions can also be multifocal.
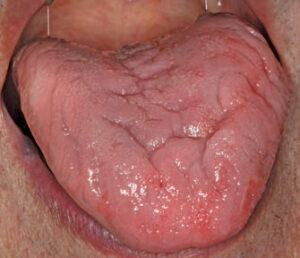
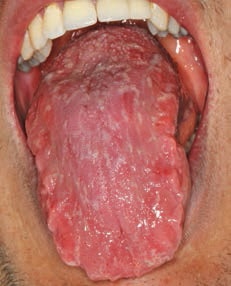
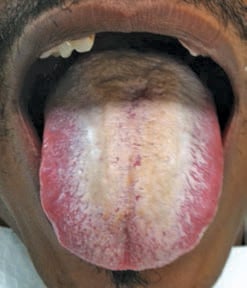
Geographic tongue is sometimes referred to as migratory because its location and pattern of presentation tend to change. The appearance of geographic tongue may fluctuate within hours, with lesions presenting for days or months — or even longer.6 This condition has a strong association with fissured tongue.4 Although geographic tongue is most often painless, patients can present with discomfort and a burning sensation. On occasion, candidiasis also may coexist with geographic tongue.10
The diagnosis of geographic tongue and its treatment are based on the patient’s medical history and clinical examination. On rare occasions, a biopsy is required to confirm a histologic diagnosis.6 Because the majority of patients are asymptomatic — and because geographic tongue is chronic or intermittent — no treatment is generally indicated. It is also possible that geographic tongue may not recur after the initial episode. To alleviate anxiety, patients often need to be assured that it is a benign process.6 Treating geographic tongue with topical corticosteroids, topical anesthetic agents and antihistamines may help alleviate symptoms, but no specific therapy has demonstrated consistent success.5,6
The differential diagnosis for geographic tongue includes erythroplakia or candidiasis. Other conditions, such as oral lichen planus, lupus erythematous and drug reactions, can occasionally mimic this condition. A biopsy may be needed to confirm the clinical diagnosis when the clinical presentation is not readily apparent. Due to the close association of geographic tongue in psoriatic patients, dentists are advised to inquire about skin problems in patients with geographic tongue, and refer these individuals for a skin evaluation if needed.11
HAIRY TONGUE
Hairy tongue, also referred to as black hairy tongue or coated tongue, affects just 0.5% of the adult population.5 Amato Lusitano, a Portuguese physician, first described this condition in 1557.12 In a study conducted on 5150 Turkish dental patients, 11.3% presented with hairy tongue that included filiform papillae elongated more than 3 mm, and 23.2% exhibited hairy tongue in which the filiform papillae were less than 3 mm.3 In a study of 3611 Minnesota schoolchildren, however, prevalence of this condition was very low (0.05%).7 There is no gender predilection. Other terminology has been ascribed to this condition, such as lingua villosa nigra, hyperkeratosis of the tongue, nigrities linguae, keratomyositis linguae and melanotrichia linguae — but none of these is commonly used today.12
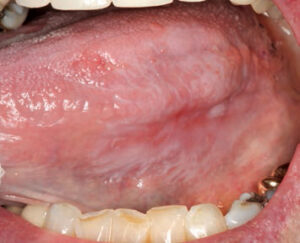
Hairy tongue affects the filiform papillae, which undergo hypertrophy and therefore elongate. The marked elongation of the filiform papillae makes them susceptible to discoloration and can present in varying shades, such as white, tan, brown or black (Figure 3).13 Darker discolorations are the result of entrapped debris, extrinsic staining from food, and chromogenic microorganisms in the filiform papillae.13 This condition may also be caused by a decrease in normal keratin desquamation.5 Because the filiform papillae are affected, this condition involves the mid-dorsal surface of the tongue, just anterior to the circumvallate papillae. The lateral borders and tip of the tongue remain uninvolved.5
The etiology for hairy tongue is not well understood. It presents among healthy individuals, as well as those with debilitating health conditions. Several predisposing factors have been implicated in its pathogenesis, including smoking and poor oral hygiene.12,14 A number of medications, such as antibiotics, antihypertensives, corticosteroids, psychotropics, and oxidizing agents such as hydrogen peroxide, are thought to contribute to the development of hairy tongue — primarily because they cause xerostomia symptoms. A few studies have implicated bismuth, tetracycline, erythromycin, linezolid and olanzapine as contributing to the development of keratin on the tongue’s filiform papillae.15 General debilitation and history of radiation therapy can predispose individuals to hairy tongue.5
Most patients with hairy tongue remain asymptomatic,5,13 although complaints of oral malodor, nausea or altered taste have been reported. In rare instances, patients may experience a gagging sensation.5 Clinical examination with gauze or a dental instrument can help assess the elongation of the filiform papillae.
Clinical presentation of hairy tongue is classic, and, as such, biopsies are generally unnecessary.5 As a benign condition, treatment is usually not required, although gentle debridement of the tongue with a soft toothbrush or commercial tongue scraper can expedite the removal of the keratin. Discontinuation of associated aggravating factors, such as tobacco, should be advocated, and good oral hygiene should be encouraged. If medication use is implicated, hairy tongue will resolve once the offending agent is eliminated.12 Hairy tongue should not be confused with oral hairy leukoplakia, which is caused by the Epstein-Barr virus.13 Oral hairy leukoplakia differs in its anatomic location and its association with immunosuppression.
MEDIAN RHOMBOID GLOSSITIS
Median rhomboid glossitis was initially thought to be a developmental condition because of its location on the dorsum of the tongue in the midline, preceding the circumvallate papillae. Embryologically, it was thought that the tuberculum impar was not covered by the lateral borders of the tongue, resulting in a smooth surface red spot on the midline of the posterior aspect of the dorsal tongue. This theory has been discarded, however, and median rhomboid glossitis is now considered a form of erythematous candidiasis.5,16 Central papillary atrophy and posterior midline atrophic candidiasis are other names for this condition.16
Median rhomboid glossitis affects approximately 1% of adults.5 In a Turkish study that included 5150 participants, only 0.5% were diagnosed with median rhomboid glossitis.3 This prevalence is consistent with another study that included 2000 Jordanian subjects.14 Although men are more commonly affected than women, in the Turkish study median rhomboid glossitis was more prevalent among women.3 The condition most commonly affects adults, and is only occasionally identified in children.5,7 There appears to be no ethnic predilection.
The prevalence of median rhomboid glossitis is slightly higher among individuals who have diabetes, patients who are immunosuppressed, and those who have recently taken a broad-spectrum antibiotic. In patients with insulin-dependent diabetes, median rhomboid glossitis is one of the most commonly reported presentations of oral candidiasis.17
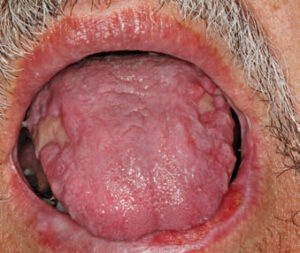
Clinically, median rhomboid glossitis is identified by a zone of depapillation of the filiform papillae, which manifests along the midline of the posterior dorsal surface of the tongue, anterior to the circumvallate papilla (Figure 4). It may arise away from the midline, as well.17 The affected area may vary in size between 1 cm and 2.5 cm. The lesion often appears well defined and erythematous, and is shaped like a rhomboid or oval lesion.3 Over time, the lesion can become more lobular. Often, signs of inflammation are noted in the area of the hard or soft palate where the tongue rests against the palate. Referred to as the kissing lesion, the presence of this condition is presumed by some to be a marker of immunosuppression and acquired immunodeficiency syndrome.16 The lesion may be caused by prolonged contact between the two anatomic locations, however.17 While patients with median rhomboid glossitis are generally asymptomatic, complaints of pain, burning, irritation or itching have been reported.17
Because the clinical presentation of median rhomboid glossitis is most often evident, clinicians may consider empirical treatment with antifungal agents. A biopsy or culture may also aid the diagnosis. Commonly used topical antifungal agents, such as nystatin or clotrimazole, as well as systemic medications, have demonstrated efficacy in resolving these lesions.9 Other conditions, such as irritation fibroma, granular cell tumor and tertiary syphilis, may be considered in the differential diagnosis.16
ORAL LICHEN PLANUS
Oral lichen planus is a relatively common disorder affecting the epithelium and oral mucosa.18 It affects 1% to 2% of the general adult population.19 It is more common among women than men. Although oral lichen planus is more frequently diagnosed in patients older than 40, it can affect children and young adults as well.
Oral lichen planus is a T-cell-mediated chronic inflammatory oral mucosal disease.18 Its proposed etiology ranges from genetics to bacterial or viral infections. Others theorize that oral lichen planus may be a reaction to medications or dental materials, or another allergic response — but these theories are not supported by clinical evidence. Its association with autoimmunity is most plausible. Previously, stress was considered to play a large factor in its etiopathogenesis. A relationship between etiopathogenesis and systemic conditions, such as hepatitis C, is controversial.18,19 Individuals with diabetes may have a slightly elevated risk of developing the condition.19
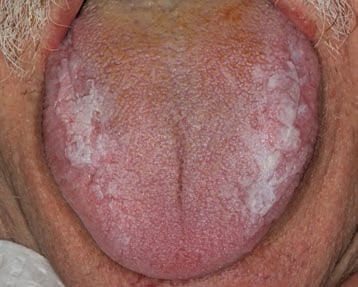
The two most common clinical presentations of oral lichen planus are a reticular, white lacy pattern (Figure 5), or an erosive form that presents with erythema and ulcerations with radiating striae (Figure 6). Another clinical variant is plaque-like oral lichen planus, which presents with white elevated patches (Figure 7).
Oral lichen planus affects the buccal mucosa, tongue and gingiva most frequently; in fact, nearly half of all oral lichen planus lesions (44%) affect the tongue.19 The lesions usually present in a bilateral and often symmetrical pattern. Oral lichen planus lesions that appear in a reticular- or plaque-like pattern are often asymptomatic or mildly symptomatic, however, patients with erosive oral lichen planus present with classic symptoms of pain, burning and irritation. At times, candida species can coexist with oral lichen planus. The condition has periods of flare-ups and remission.20 Extraorally, oral lichen planus can affect the skin, skin appendages and other sites, such as genital, ocular, nasal, laryngeal, gastric and anal mucosa. There has been debate as to whether oral lichen planus is likely to transform into oral cancer.20 The risk is thought to vary between 0.5% and 5% over a period of 1 to 20 years.18
Diagnosis of oral lichen planus should be made based on clinical and histologic findings, as many other conditions can clinically mimic the condition — such as graft-versus-host disease, leukoplakia and squamous cell carcinoma. The plaque-like form of oral lichen planus is easily confused with leukoplakia.18 For symptomatic patients, administering topical or systemic steroids remains the most common therapy. In refractory cases, the use of other topical or systemic immunomodulators and retinoids has been tried. Due to the potential for malignant transformation, there is some concern regarding treatment of oral lichen planus lesions with immunomodulators,18 and, owing to this premalignant potential, close monitoring during the course of treatment is recommended.21
CONCLUSION
Dental professionals frequently encounter oral lesions in practice and reaching a diagnosis requires clinicians to take a thorough medical history and perform an in-depth oral examination. Familiarity with common characteristics of oral lesions — in regard to color, shape, size, location and morphology — assists practitioners in making an accurate diagnosis. The history should include information regarding onset, duration and symptoms, as well as history of alcohol and tobacco use. Any lesion that cannot be easily or accurately diagnosed should be referred to a specialist for further evaluation.
References
- Shulman JD, Beach MM, Rivera-Hidalgo F. The prevalence of oral mucosal lesions in US adults: data from the Third National Health and Nutrition Examination Survey, 1988–1994. J Am Dent Assoc. 2004;135:1279–1286.
- Costa FW, Osterne RL, Mota MR, Alves AP, Soares EC, Sousa FB. Tongue lesions. J Craniofac Surg. 2012;23:548–551.
- Avcu N, Kanli A. The prevalence of tongue lesions in 5150 Turkish dental outpatients. Oral Dis. 2003;9:188–195.
- Shulman JD, Carpenter WM. Prevalence and risk factors associated with geographic tongue among U.S. adults. Oral Dis. 2006;12:381–386.
- Neville BW, Damm DD, Allen CM and Bouquot JE. Oral and Maxillofacial Pathology. 3rd ed. Philadelphia: WB Saunders; 2009.
- Assimakopoulos D, Patrikakos G, Fotika C, Elisaf M. Benign migratory glossitis or geographic tongue: an enigmatic oral lesion. Am J Med. 2002;113:751–755.
- Redman RS. Prevalence of geographic tongue, fissured tongue, median rhomboid glossitis, and hairy tongue among 3611 Minnesota schoolchildren. Oral Surg Oral Med Oral Pathol. 1970;30:390–395.
- Jainkittivong A, Langlais RP. Geographic tongue: clinical characteristics of 188 cases. J Contemp Dent Pract. 2005;6:123–135.
- Gonsalves WC, Chi AC, Neville BW. Common oral lesions: Part I. Superficial mucosal lesions. Am Fam Physician. 2007;75:501–507.
- Ching V, Grushka M, Darling M, Su N. Increased prevalence of geographic tongue in burning mouth complaints: a retrospective study. Oral Surg Oral Med Oral Pathol Oral Radiol. 2012;114:444–448.
- Tarakji B, Umair A, Babaker Z, Sn A, Gazal G, Sarraj F. Relation between psoriasis and geographic tongue. J Clin Diagn Res. 2014;8:ZE06–ZE07.
- Lawoyin D, Brown RS. Drug-induced black hairy tongue: diagnosis and management challenges. Dent Today. 2008;27:60,62–63.
- Reamy BV, Derby R, Bunt CW. Common tongue conditions in primary care. Am Fam Physician. 2010;81:627–634.
- Darwazeh AM, Almelaih AA. Tongue lesions in a Jordanian population. Prevalence, symptoms, subject’s knowledge and treatment provided. Med Oral Patol Oral Cir Bucal. 2011;16:745–749.
- Nisa L, Giger R. Black hairy tongue. Am J Med. 2011;124:816–817.
- Nelson BL, Thompson L. Median rhomboid glossitis. Ear Nose Throat J. 2007;86:600–601.
- Goregen M, Miloglu O, Buyukkurt MC, Caglayan F, Aktas AE. Median rhomboid glossitis: a clinical and microbiological study. Eur J Dent. 2011;5:367–372.
- Scully C, Carrozzo M. Oral mucosal disease: lichen planus. Br J Oral Maxillofac Surg. 2008;46:15–21.
- Carbone M, Arduino PG, Carrozzo M, et al. Course of oral lichen planus: a retrospective study of 808 northern Italian patients. Oral Dis. 2009;15:235–243.
- Farhi D, Dupin N. Pathophysiology, etiologic factors, and clinical management of oral lichen planus, part I: facts and controversies. Clin Dermatol. 2010;28:100–108.
- Fitzpatrick SG, Hirsch SA, Gordon SC. The malignant transformation of oral lichen planus and oral lichenoid lesions: a systematic review. J Am Dent Assoc. 2014;145:45–56.




