Peer to Peer: Preprocedural Rinsing and Dental Aerosols
Preprocedural rinses have recently been recommended to help reduce viral loads in dental aerosols.
Aerosols generated during dental procedures result in increases in aerosolized bacteria, viruses and fungi during and following treatment — and these aerosols can travel significant distances from treatment sites.1,2 While preprocedural rinsing is an off-label use, mouthrinses used to reduce bacterial loads in dental aerosols include chlorhexidine gluconate (CHX), cetylpyridinum chloride (CPC), essential oils (EO) and povidone-iodine (PVP-I). Preprocedural rinsing has recently been recommended with the objective of reducing viral loads with SARS-CoV-2 during the COVID-19 pandemic.
Bacterial Load Reduction — A recent systematic review of 13 randomized controlled clinical trials concluded there was moderate evidence for the efficacy of CHX, CPC and EO in reducing bacterial loads in dental aerosols.2 Studies measuring salivary bacterial levels lend support for prerinsing.3,4 At bactericidal concentrations, CHX and CPC disrupt bacterial cell membranes, resulting in leakage of intracellular proteins and cell death. In general, the mechanism of action for EO is similar. The use of PVP-I releases free iodine, which disrupts cell membranes and oxidizes proteins and fatty acids, resulting in cell death.
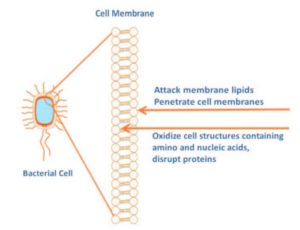
Viral Aerosols and SARS-CoV-2 — Although SARS-CoV-2 is transmitted primarily through droplets and contact, aerosols represent an alternative mode of transmission.5,6 High viral loads of SARS-CoV-2 have been found in saliva in infected individuals and in the oropharynx of asymptomatic individuals.7,8 Recently, preprocedural rinsing with 1.5% hydrogen peroxide or 0.2% PVP-I was suggested for dental patients.5 Both are oxidizing agents, and SARS-CoV-2 is susceptible to oxidation (Figure 1). In other geographic areas, 1% hydrogen peroxide has been recommended, and 3% hydrogen peroxide rinses have been suggested in patients with early COVID-19 infection.6,9 Hydroxyl free radicals are produced by hydrogen peroxide and in vitro are known to attack membrane lipids, genetic material (which contains nucleic acids), and other cell components.10 Hydrogen peroxide rinses have a history of safe use. In addition, PVP-I has demonstrated in vitro efficacy against SARS-CoV and MERS-CoV, and was recommended for use against MERS-CoV during the MERS epidemic.7,11 Free iodine oxidizes cell components containing amino and nucleic acids and disrupts proteins.7,11
Conclusions — Preprocedural rinsing has been recommended to help reduce transmission of pathogenic microorganisms. At this time, there is no evidence for the use of any preprocedural rinse against SARS-CoV-2.12 However, biologic plausibility suggests hydrogen peroxide and PVP-I could be expected to be virucidal intraorally, and may reduce SARS-CoV-2 viral load in dental aerosols. More research is needed on preprocedural rinses, the minimal infectious dose for SARS-CoV-2, and a quantifiable risk of SARS-CoV-2 transmission from dental aerosols.
REFERENCES
- Gralton J, Tovey E, McLaws ML, Rawlinson WD. The role of particle size in aerosolised pathogen transmission: a review. J Infect. 2011;62:1‐13.
- Marui VC, Souto ML, Rovai ES, Romito GA, Chambrone L, Pannuti CM. Efficacy of preprocedural mouthrinses in the reduction of microorganisms in aerosol: A systematic review. J Am Dent Assoc. 2019;150:1015–1026.
- Balbuena L, Stambaugh KI, Ramirez SG, Yeager C. Effects of topical oral antiseptic rinses on bacterial counts of saliva in healthy human subjects. Otolaryngol Head Neck Surg. 1998;118:625–629.
- Veksler AE, Kayrouz GA, Newman MG. Reduction of salivary bacteria by pre-procedural rinses with chlorhexidine 0.12%. J Periodontol. 1991;62:649–651.
- American Dental Association. Interim Guidance for Minimizing Risk of COVID-19 Transmission. Available at: https://www.ada.org/~/media/CPS/Files/COVID/ADA_COVID_Int_Guidance_Treat_Pts.pdf. Accessed May 29, 2020.
- Peng X, Xu X, Li Y, Cheng L, Zhou X, Ren B. Transmission routes of 2019-nCoV and controls in dental practice. Int J Oral Sci. 2020;12:9.
- Kirk-Bailey J, Combes J, Sunkaraneni S, Challacombe S. The Use of Povidone Iodine Nasal Spray and Mouthwash During the Current COVID-19 Pandemic for the Reduction of Cross Infection and Protection of Healthcare Workers. Available at: https://papers.ssrn.com/sol3/papers.cfm?abstract_id=3563092. Accessed May 29, 2020.
- Zou L, Ruan F, Huang M, et al. SARS-CoV-2 viral load in upper respiratory specimens of infected patients. N Engl J Med. 2020;382:1177–1179.
- Caruso AA, Del Prete A, Lazzarino AI, Capaldi R, Grumetto L. May hydrogen peroxide reduce the hospitalization rate and complications of SARS-CoV-2 infection? Infect Control Hosp Epidemiol. April 22, 2020. (e-Pub ahead of print.)
- U.S. Centers for Disease Control and Prevention. Chemical Disinfectants. Guideline for Disinfection and Sterilization in Healthcare Facilities (2008). Available at: https://www.cdc.gov/infectioncontrol/guidelines/disinfection/disinfection-methods/chemical.html#Hydrogen. Accessed May 29, 2020.
- Eggers M. Infectious disease management and control with povidone iodine. Infect Dis Ther. 2019;8:581–593.
- American Dental Association. Return to Work Interim Guidance Toolkit. Available at: https://success.ada.org/~/media/CPS/Files/Open%20Files/ADA_Return_to_Work_Toolkit.pdf. Accessed May 29, 2020.
From Decisions in Dentistry. June 2020;6(6):8.


