Periodontal Regeneration With Human Stem Cells
For patients with periodontal disease, mesenchymal stem cells hold promise for restoring the periodontium to its original architecture and function.
This course was published in the January 2019 issue and expires January 2022. The author has no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Discuss the various types and sources of human stem cells, and their potential application in periodontal regenerative therapy.
- Describe the concept of tissue engineering, and the factors that influence periodontal regeneration.
- Explain why the use of dental tissue-derived mesenchymal stem cells holds particular interest and promise in this field.
Periodontitis is a multifactorial inflammatory disease involving the complex interplay between the host’s immune response to microbial accumulation at the tooth/gingiva interface, as influenced by lifestyle, environmental factors and host response genes.1 If periodontitis is left untreated, ongoing bone loss may ultimately lead to tooth loss. The goal of therapy is to restore the periodontium to its original architecture and function — but this is difficult because it involves the reconstruction of at least four tissues: gingiva, periodontal ligament, bone and cementum.2 Although conventional treatment shows limited potential for complete periodontal regeneration, newer technologies hold promise for achieving predictable regenerative outcomes.3
Periodontal regeneration requires carefully orchestrated events to occur in spatial and temporal sequences reminiscent of periodontal development.4 Early in this process, migration of appropriate mesenchymal (or adult) stem cells into the periodontal defect is required. The cells proliferate and differentiate into specialized cells responsible for the reconstruction of a functional periodontal attachment apparatus. These processes are controlled by growth factors that affect gene expression.5 Mesenchymal stem cells are of interest because they are precursor cells capable of differentiating into the osteoblasts, cementoblasts and fibroblasts needed for periodontal regeneration.
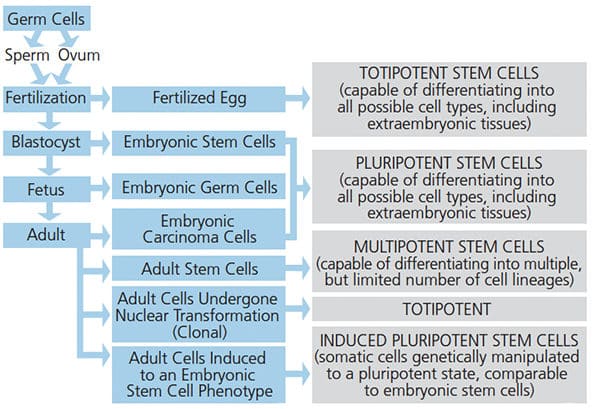
Defined by their capacity to self renew and proliferate, stem cells have the ability to reproducibly differentiate into functional cells indicative of different lineages.6 Stem/progenitor cells have been isolated from nearly every human tissue and organ, and have been categorized according to their source and developmental (or differentiation) potential (Figure 1). There are four main sources of stem cells: embryonic tissues, fetal tissues, postnatal tissues, and reprogrammed differentiated somatic cells, also known as induced pluripotent stem (iPS) cells.
Embryonic stem cells are pluripotent cells derived, meaning they can differentiate into cells with features of all three embryonic germ layers (mesoderm, endoderm and ectoderm). Despite their potent differentiation potential, obtaining human embryonic stem cells raises ethical issues that preclude their widespread use. However, advances have allowed genetic reprogramming of mesenchymal stem cells into iPS cells that are functionally similar to embryonic stem cells.7
Due to their differentiation capabilities, adult stem cells have been studied in considerable detail for use in regenerative medicine.8 Although they have limited differentiation potential compared to embryonic stem cells, mesenchymal stem cells are not associated with ethical concerns, are easily accessible and, importantly, immunocompatible. Due to their ubiquitous presence in adult tissues, including dental tissues, they have been investigated for use in periodontal regeneration.
CELL-BASED PERIODONTAL TISSUE ENGINEERING
While the simple injection of cells into osseous defects has been studied, a more efficient approach to periodontal regeneration is to combine mesenchymal stem cells with tissue growth factors and space maintaining and delivery systems. A key goal in periodontal tissue engineering is using cell culture methods (aided by growth and differentiation factors) to produce suitable quantities of stem cells that can be placed into periodontal defects. These regenerative technologies rely on input from a large number of fields, including nanotechnology, three-dimensional (3D) bioprinting, bioengineering, cell biology, molecular biology and gene manipulation; practical implementation, however, also requires efficient manufacturing processes.9,10 Key factors for using mesenchymal stem cells for periodontal regeneration include:11
- An adequate supply of progenitor cells with the capacity to differentiate into the required mature tissue-forming phenotypes, including osteoblasts, cementoblasts and fibroblasts
- The appropriate signals to modulate cellular differentiation and tissue neogenesis
- A conductive 3D extracellular matrix scaffold to support and facilitate these processe
- Angiogenic signals that promote new vascular networks are essential to provide the nutritional base for tissue growth and homeostasis
- Appropriate mechanical loading is needed for development of highly organized, functional periodontal ligament fibers
- As periodontal defect sites support a microbial load, controlling infection and host response will promote optimal periodontal regeneration
EXTRAORAL MESENCHYMAL STEM CELLS
Bone Marrow-Derived Mesenchymal Stem Cells: Early preclinical trials have shown bone marrow-derived adult stem cells can promote periodontal regeneration through enhanced neovascularization and regeneration of alveolar bone, cementum and periodontal ligament.12–15 However, difficulties in procuring these cells have limited their use and led to investigation of dental-tissue-derived mesenchymal stem cells that can easily be obtained chairside.
Adipose-Derived Stromal Cells: Human adipose-derived stromal cells can be obtained via liposuction procedures and may have potential for periodontal tissue engineering. When mixed with platelet rich plasma, adipose-derived stromal cells have been shown in rat models to promote regeneration of the periodontal ligament and alveolar bone.16,17
Intraoral Mesenchymal Stem Cells: Since the first identification of adult stem cells in the human periodontal ligament,18 mesenchymal stem cell-like populations have been isolated from all dental tissues except enamel.19 Although limited, the periodontium retains some ability to regenerate, and this appears to be facilitated by the presence of mesenchymal stem cells.20 But because dental tissues do not have the same capacity to remodel as bone tissues, dental-derived mesenchymal stem cells are more restricted in their differentiation capacity than bone marrow-derived cells.21
Periodontal ligament mesenchymal stem cell populations are of particular interest. Early studies demonstrated populations of stem cells within the periodontium have the potential to differentiate into either cementoblasts or osteoblasts.22,23 A recent systematic review reported that when implanted into periodontal defects, periodontal ligament-derived stem cells resulted in statistically significant improvement in 70.5% of the studies examined.24 The use of periodontal ligament cells cultured into cell sheets has also been studied.25,26 Preclinical animal models of dehiscence and intrabony defects have demonstrated periodontal regeneration involving formation of new bone, periodontal ligament and cementum following implantation of autologous periodontal ligament cell sheets.25,26 Transplanting allogeneic periodontal ligament cell sheets into periodontal defects in miniature swine has resulted in periodontal tissue regeneration in a fashion similar to transplantation of autologous cell sheets — and with no evidence of rejection.27
TISSUE ENGINEERING SCAFFOLD
As detailed above, periodontal tissue engineering involves placing cells into a prefabricated 3D construct that can be implanted into the defect site.4 This approach overcomes some of the limitations associated with conventional periodontal regeneration, as it allows direct delivery of growth factors and/or progenitor cells into the periodontal site. Central to this approach is the construction of 3D scaffolds to support and facilitate the delivery of cells and growth factors. The development of scaffolds for periodontal regeneration evolved from the early study of membrane guided tissue regeneration. The concepts of guided periodontal regeneration were based on the understanding it was critical to provide regenerative space. In this context, several “monophasic” constructs (in the form of barrier membranes) were developed. Bioactivity could be incorporated using various inorganic biomaterials and/or growth factors to promote regeneration of bone, periodontal ligament and cementum.28 Although this approach seems relatively straightforward, clinical results proved unpredictable. Accordingly, newer materials were developed based on the understanding of the spatial and temporal sequences of biologic events associated with periodontal regeneration. These materials can be distinguished as either biphasic or triphasic constructs.
Biphasic constructs for periodontal tissue engineering have incorporated an osteoconductive composite foam (or bone compartment) on one side of a traditional occlusive membrane in an attempt to better replicate an environment for periodontal regeneration.29–31 This approach has been further developed using sophisticated additive manufacturing technology to develop biphasic scaffolds composed of both bone and ligament compartments.32 Seeding of periodontal fibroblasts into the ligament compartments, and subsequent layering onto dentin slices prior to implantation in rats, resulted in formation of periodontal ligament and cementum-like tissue.32
Early studies using biphasic scaffolds focused mainly on regenerating periodontal ligament and alveolar bone. However, this is only part of the periodontal regenerative process, and a critical missing piece is the ability of biphasic constructs to induce new cementum formation. Accordingly, recent studies have begun to address how incorporating a third layer into these periodontal scaffolds might induce cementogenesis.
Several triphasic scaffolds have been described that can be produced using conventional scaffold fabrication technologies or additive manufacturing. Thus, triphasic scaffolds can be fabricated that, in addition to bone and ligament compartments, have an extra cementum layer. These possess the necessary compartments to produce bone, periodontal ligament and cementum.33 More recently, a tri-layered scaffold with gingival, bone and periodontal compartments demonstrated the potential to promote formation of bone, cementum and interposing soft tissue.33 These features were representative of neogenesis of the periodontium, although the fiber arrangement was not fully functional.34
CUSTOMIZED SCAFFOLDS
While cell-based therapies have considerable potential, they still pose limitations largely associated with regulatory barriers to how cells are sourced, as well as laboratory cell culturing processes and safety issues. As a result, the development of advanced synthetic scaffolds that incorporate drugs or growth factors has been pursued as an alternative to cell-based tissue engineering.
The next phase of cell/scaffold technology will involve using computer aided design software to customize inserts to precisely fit periodontal defects. In this process, computed tomography helps determine the attributes of a periodontal defect to aid production of customized 3D constructs. The technology’s potential has been demonstrated in a case report in which cone beam computed tomography of a periodontal defect was used to prefabricate a customized scaffold using selective laser sintering.15 The scaffold comprised an internal region, with pegs to support periodontal ligament regeneration and the delivery of platelet-derived growth factor, as well as an external resorbable compartment designed to be replaced with alveolar bone. The scaffold remained in place for 12 months without signs of dehiscence or chronic inflammation. However, due to design features that did not allow mechanical matching with gingival tissue, the construct eventually became exposed to the oral environment, resulting in its loss due to bacterial infection. Nonetheless, this report demonstrates the technical and biologic feasibility of such approaches.
STUDIES USING DENTAL-DERIVED HUMAN STEM CELLS
The periodontal regenerative capacity of dental-derived stem cells has been well demonstrated in animal models. More recently, several small human clinical trials have used periodontal ligament stem cells and dental pulp stem cells.35–38 These studies have confirmed the safety and efficacy of using implanted autologous cells for periodontal regeneration. Other stem cells, such as bone marrow stromal stem cells and adipose tissue stem cells, have also been used with some success.
CANDIDATES FOR THERAPY
Who should receive such therapy? Patient and site selection for these new approaches will be no different than protocols described for periodontal regeneration using membranes and biologic materials.39 An individual’s stage or grade of periodontitis, or if he or she has specific genotype features (such as an IL-1 beta polymorphism), appears to have no effect on outcomes.40,41 While postoperative infection is always an issue, an individual’s specific microbial genotype has not been shown to have an adverse effect on periodontal regeneration.
Compared to traditional open-flap debridement, the question of whether the slight clinical improvements offered by regenerative techniques are cost effective is yet to be answered. To date, no cost/benefit analyses for periodontal regeneration have been reported. However, with the introduction of the “number needed to treat” (NNT) concept for regenerative periodontics, some clues can be gleaned; (NNT is the average number of patients who need to be treated for one to benefit compared to a control). Two studies have reported the NNT for periodontal regeneration and suggested that at least eight to nine patients have to receive a regenerative procedure to achieve one patient gaining at least 2 mm of probing attachment level.42,43 Clearly, these are not remarkable outcomes — but whether the use of newer technologies as described in this paper will improve these outcomes remains to be seen.
CONCLUSION
The development of clinical procedures that allow complete and predictable periodontal regeneration of tissues damaged by disease or trauma remains challenging. Regeneration of periodontal tissues requires a thorough understanding of the temporal and spatial requirements, and includes the use of appropriate human stem cells, crucial effector molecules and well-designed scaffolds.
Extraoral- and dental tissue-derived stem/progenitor cells have been studied in preclinical animal models for potential use in periodontal tissue engineering. There is now good evidence that use of mesenchymal stem cells, in conjunction with highly structured scaffolds, can achieve periodontal regeneration. Yet many aspects must be addressed before this becomes a clinical reality. In particular, science needs more comprehensive information concerning the cell and molecular biology of stem cells and their capacity to facilitate regeneration.
Notwithstanding these issues, additional challenges must be addressed before this approach can be used as standard treatment. Further development is needed in scaffold fabrication, as well as the biosafety of using implanted cells. Concerns still exist over how the overall processes of tissue regeneration are controlled. And there is always a risk that while the process can be turned on, what happens if it cannot be turned off? Regulatory bodies will require stringent adherence to manufacturing protocols for clinical translation, and this raises considerable cost implications.
Perhaps the ultimate limiting factor for use of human stem cells for periodontal tissue engineering is not whether it can be done, but whether it will eventually prove cost effective for routine clinical use.
REFERENCES
- Bartold PM, Van Dyke TE. Periodontitis: a host-mediated disruption of microbial homeostasis. Unlearning learned concepts. Periodontol 2000. 2013;62:203–217.
- Narayanan AS, Bartold PM. Biochemistry of periodontal connective tissues and their regeneration: a current perspective. Connect Tissue Res. 1996;34:191–201.
- Bartold PM. Group C. Initiator paper. Periodontal regeneration — fact or fiction? J Int Acad Periodontol. 2015;17(Suppl 1):37–49.
- Bartold PM, McCulloch CA, Narayanan AS, Pitaru S. Tissue engineering: a new paradigm for periodontal regeneration based on molecular and cell biology. Periodontol 2000. 2000;24:2532–2569.
- Smith PC, Martínez C, Cáceres M, Martínez J. Research on growth factors in periodontology. Periodontol 2000. 2015;67:234–250.
- Smith A. A glossary for stem-cell biology. Nature. 2006;441:1060–1060.
- Takahashi K, Tanabe K, Ohnuki M, Narita M, Ichisaka T, Tomoda K, Yamanaka S. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 2007;131:861–872.
- Kanji S, Das H. Advances of stem cell therapeutics in cutaneous wound healing and regeneration. Mediators Inflamm. 2017;2017:5217967.
- Li JJ, Ebied M, Xu J, Zreiqat H. Current approaches to bone tissue engineering: the interface between biology and engineering. Adv Healthc Mater. 2018;7:e1701061.
- Oryan A, Kamali A, Moshiri A, Baghaban Eslaminejad M. Role of mesenchymal stem cells in bone regenerative medicine: what is the evidence? Cells Tissues Organs. 2017;204:59–83.
- Bruder SP, Fink DJ, Caplan AI. Mesenchymal stem cells in bone development, bone repair, and skeletal regeneration therapy. J Cell Biochem. 1994;56:283–294.
- Li H, Yan F, Lei L, Li Y, Xiao Y. Application of autologous cryopreserved bone marrow mesenchymal stem cells for periodontal regeneration in dogs. Cells Tissues Organs. 2009;190:94–101.
- Kawaguchi H, Hirachi A, Hasegawa N, et al. Enhancement of periodontal tissue regeneration by transplantation of bone marrow mesenchymal stem cells. J Periodontol. 2004;75:1281–1287.
- Hasegawa N, Kawaguchi H, Hirachi A. Behavior of transplanted bone marrow-derived mesenchymal stem cells in periodontal defects. J Periodontol. 2006;77:1003–1007.
- Yamada Y, Ueda M, Hibi H, Baba S. A novel approach to periodontal tissue regeneration with mesenchymal stem cells and platelet-rich plasma using tissue engineering technology: a clinical case report. Int J Periodontics Restorative Dent. 2006;26:363–369.
- Tobita M, Uysal AC, Ogawa R, Hyakusoku H, Mizuno H. Periodontal tissue regeneration with adipose-derived stem cells. Tissue Eng Part A. 2008;14:945–953.
- Lemaitre M, Monsarrat P, Blasco-Baque V, et al. Periodontal tissue regeneration using syngeneic adipose-derived stromal cells in a mouse model. Stem Cells Transl Med. 2017;6:656–665.
- Seo BM, Miura M, Gronthos S, et al. Investigation of multipotent postnatal stem cells from human periodontal ligament. Lancet. 2004;364:149–155.
- Chen Fm, Sun HH, Lu H, Yu Q. Stem cell-delivery therapeutics for periodontal tissue regeneration. Biomaterials. 2012;33:6320–6344.
- Ivanovski S, Gronthos S, Shi S, Bartold PM. Stem cells in the periodontal ligament. Oral Dis. 2006;12:358–363.
- Huang GT, Gronthos S, Shi S. Mesenchymal stem cells derived from dental tissues vs. those from other sources: their biology and role in regenerative medicine. J Dent Res. 2009;88:792–806.
- McCulloch CA, Bordin S. Role of fibroblast subpopulations in periodontal physiology and pathology. J Periodontal Res. 1991;26(3 Pt 1):144–154.
- Isaka J, Ohazama A, Kobayashi M, et al. Participation of periodontal ligament cells with regeneration of alveolar bone. J Periodontol. 2001;72:314–323.
- Bright R, Hynes K, Gronthos S, Bartold PM. Periodontal ligament-derived cells for periodontal regeneration in animal models: a systematic review. J Periodontal Res. 2015;50:160–172.
- Akizuki T, Oda S, Komaki M, et al. Application of periodontal ligament cell sheet for periodontal regeneration: a pilot study in beagle dogs. J Periodontal Res. 2005;40:245–251.
- Tsumanuma Y, Iwata T, Washio K, et al. Comparison of different tissue-derived stem cell sheets for periodontal regeneration in a canine 1-wall defect model. Biomaterials. 2011;32:5819–5825.
- Ding G, Liu Y, Wang W, et al. Allogeneic periodontal ligament stem cell therapy for periodontitis in swine. Stem Cells. 2010;28:1829–1838.
- Baba S, Yamada Y, Komuro A, et al. Phase I/II trial of autologous bone marrow stem cell transplantation with a three-dimensional woven-fabric scaffold for periodontitis. Stem Cells Int. 2016;2016:6205910.
- Carlo Reis EC, Borges AP, Araújo MV, Mendes VC, Guan L, Davies JE. Periodontal regeneration using a bilayered PLGA/calcium phosphate construct. Biomaterials. 2011;32:9244–453.
- Requicha JF, Viegas CA, Hede S, Leonor IB, Reis RL, Gomes ME. Design and characterization of a biodegradable double-layer scaffold aimed at periodontal tissue-engineering applications. J Tissue Eng Regen Med. 2016;10:392–403.
- Park CH, Rios HF, Jin Q, et al. Tissue engineering bone-ligament complexes using fiber-guiding scaffolds. Biomaterials. 2012;33:137–145
- Sowmya S, Mony U, Jayachandran P, et al. Tri-layered nanocomposite hydrogel scaffold for the concurrent regeneration of cementum, periodontal ligament, and alveolar bone. Adv Healthc Mater. 2017;6:doi:10.1002/adhm.201601251.
- Varoni EM, Vijayakumar S, Canciani E, et al. Chitosan-based trilayer scaffold for multi-tissue periodontal Regeneration. J Dent Res. 2018;97:303–311.
- Rasperini G, Pilipchuk SP, Flanagan CL, et al. 3D-printed bioresorbable scaffold for periodontal repair. J Dent Res. 2015;94(Suppl 9):153S–157S.
- McAllister BS. Stem cell-containing allograft matrix enhances periodontal regeneration: case presentations. Int J Periodontics Restorative Dent. 2011;31:149–155.
- Feng F, Akiyama K, Liu Y, et al. Utility of PDL progenitors for in vivo tissue regeneration: a report of 3 cases. Oral Dis. 2010;16:20–28.
- Chen FM. Gao LN, Tian B, et al. Treatment of periodontal intrabony defects using autologous periodontal ligament stem cells: a randomized clinical trial. Stem Cell Res Ther. 2016;7:33.
- Lin NH, Gronthos S, Bartold PM. Stem cells and periodontal regeneration. Aust Dent J. 2008;53:108–121.
- Tonetti MS, Cortellini P. Case selection and treatment considerations of guided tissue regeneration in deep intrabony defects. Curr Opin Periodontol. 1997;4:82–88.
- Corbella S, Weinstein R, Francetti L, Taschieri S, Del Fabbro M. Periodontal regeneration in aggressive periodontitis patients: a systematic review of the literature. J Investig Clin Dent. 2017;8:doi:10.1111/jicd.12245.
- Chatzopoulos GS, Koidou VP. Association between susceptible genotypes to periodontitis and clinical outcomes of periodontal regenerative therapy: a systematic review. Med Oral Pathol Cir Bucal. 2016;21:e456–e464.
- Needleman IG, Worthington HV, Giedrys-Leeper E, Tucker RJ. Guided tissue regeneration for periodontal infra-bony defects. Cochrane Database Syst Rev. 2006;2:CD001724.
- Esposito M, Grusovin MG, Papanikolaou N, Coulthard P, Worthington HV. Enamel matrix derivative (Emdogain) for periodontal tissue regeneration in intrabony defects. Cochrane Database Syst Rev. 2009;4:CD003875.
Featured image by PABLO_K/ISTOCK/GETTY IMAGES PLUS
From Decisions in Dentistry. January 2019;5(1):38–41.




