Periodontal Medicine: From Teeth To Total Body Health
Learning to identify and manage systemic conditions as part of a multidisciplinary team can improve outcomes in periodontal care.
This course was published in the March 2021 issue and expires March 2024. The author has no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Describe the relationship between chronic systemic conditions and various oral complications.
- Explain the effects periodontal therapy may have on various systemic diseases.
- Identify risk factors and appropriate dental screening based on the patient’s systemic disease.
Patients with advanced periodontal breakdown pose a unique challenge to the restorative dentist. Tooth loss, increased mobility and severe attachment loss complicate efforts to restore a patient’s function and esthetics. Arriving at the proper diagnosis and prognosis for patients with poor oral and overall health will improve the way clinicians rehabilitate an otherwise terminal dentition.
Systemic health has a tremendous impact on the periodontal/restorative interface. Diabetes mellitus, cardiovascular disease, obesity, and other inflammatory conditions can contribute to the rate of disease progression and responsiveness to treatment. Learning to identify and manage systemic conditions as part of a multidisciplinary team can improve outcomes in periodontal care and positively impact the patient’s quality of life.
DIABETES MELLITUS
Diabetes mellitus is a dysregulation of carbohydrate and lipid metabolism due to an inability to secrete or respond to insulin. Type 1 diabetes mellitus (T1DM) is the result of an absolute insulin deficiency via autoimmune destruction of pancreatic beta cells. In a cross-sectional study of patients living with T1DM for more than 50 years, only 13.5% had severe periodontitis — lower than most prevalences of the same cohort. It is hypothesized there are residual, functional beta cells in T1DM patients that protect against autoimmune stress and tissue destruction.1
Chronic periodontitis is often considered the sixth complication of type 2 diabetes mellitus (T2DM), a condition that increases systemic inflammation in different ways as glucose tolerance gradually decreases. The primary cells of the innate immune system become less active, thereby leaving the periodontium vulnerable to the dental biofilm. Infection onset triggers a massive cytokine release from macrophages, leading to impaired collagen turnover and bone loss over time.2 Multiple cross-sectional studies among Pima Indians show a near threefold increase in clinical and radiographic parameters of periodontitis for T2DM patients.3,4 In the same population, an almost 11-time increased risk for bone loss in patients with a hemoglobin A1c (HbA1c) ≥ 9% was identified.4 Patients with poorly controlled T2DM are clearly more susceptible to the sequelae of a down regulated immune response.
On average, an 0.5% reduction in a patient’s HbA1c can be anticipated with nonsurgical and surgical treatment. The largest multicenter randomized controlled trial for patients with moderate to severe periodontitis and T2DM found a 0.3% to 0.6% reduction in HbA1c levels after one year of interventional periodontal care, as compared to those who received supragingival prophylaxis alone. Additionally, these patients had a diminished 10-year cardiovascular disease risk.5 However, the state of glycemia is more important than having T2DM alone. A recent meta-analysis found no significant differences in clinical outcomes after initial therapy for well-controlled T2DM patients compared to healthy controls.6
Tighter management of glycemic control results in better periodontal and overall health. In fact, an HbA1c ≤ 6.5% is the desired range for T2DM patients, per the American Association of Clinical Endocrinologists. Systemic complications, such as neuropathy, atherosclerosis and poor wound healing, occur thereafter. Since the vast majority (70.1%) of T2DM patients present with HbA1c > 6.5%, advanced screening and consultation should be considered to meet this threshold before surgery is pursued.7 Reducing inflammation via nonsurgical therapy and improved self-care should be prioritized in poorly controlled patients.
Individuals with severe forms of periodontitis should be screened for diabetes as part of their initial exam, along with other risk factors that include obesity, age, ethnicity and family history. For example, those with signs of polydipsia, polyurea, and/or polyphagia with a casual blood glucose > 200 mg/dL should be referred. However, this only provides a snapshot of their glycemic control. Chairside HbA1c readings are better point-of-care tests because they reflect HbA1c levels over a 12-week period, without the need for separate medical consultation. As of 2017, oral health professionals who use these monitoring devices chairside must register the practice as a laboratory and receive a Clinical Laboratory Improvement Amendment Certificate of Waiver every two years. Both chairside tests are now associated with billable procedure codes.
CARDIOVASCULAR DISEASE
A causal link between periodontitis and cardiovascular disease has long been difficult to establish. They share common risk factors, such as male gender, age, cigarette smoking, T2DM, and low socioeconomic status. And, after adjusting for confounders, most studies report a positive association between these disease processes. This is due to the direct and indirect pathways that connect the pathogenesis of periodontitis to cardiovascular disease, and vice versa.8
Oral bacteria and their byproducts can enter the bloodstream, damaging the vascular endothelium and increasing oxidative stress. In fact, periopathogens were found in 30% of resected human carotid plaques, with 44% containing at least one or more oral pathogens.9 Local oxidation attracts macrophages to the site of injury. As these cells ingest various lipoproteins and lipids that accumulate along the vascular wall, they become pathogenic foam cells. A release in platelet-derived growth factor then causes collagen deposition and fibrous encapsulation. This fatty plaque or atheroma increases tunica intima-media wall thickness, which is a preclinical measure of atherosclerosis associated with coronary heart disease and stroke.8
Indirectly, periodontitis increases systemic inflammation and alters macrophage activity to promote atheroma formation.10 Nonsurgical and surgical periodontal treatment, however, can reduce proinflammatory markers over time. A clinical trial of 65 healthy subjects showed a significant decrease in C-reactive protein and interleukin-6 (IL-6) two months after prophylaxis and scaling and root planing.11 In another interventional study, three months after full-mouth tooth extractions, inflammatory and thrombotic markers decreased when severe periodontitis was eliminated.12
Periodontal patients should be informed of the comorbidities associated with cardiovascular disease. In the nationwide Atherosclerosis Risk in Communities Study, the odds of carotid artery intima-media wall thickness ≥ 1 mm were 1.4 to 2.09 times higher for patients with moderate to severe periodontitis compared to healthy controls.13 A meta-analysis later revealed the prevalence and incidence of coronary heart disease were significantly increased in periodontitis, especially for subjects with less than 10 teeth.14 Periodontal treatment will not guarantee reduced risk for cardiovascular disease, but instead should be seen as a means to offset the body’s inflammatory burden, thereby improving overall health.
OBESITY
Adipose tissue is an active endocrine organ, and the destructive wake of central adiposity is far-reaching. Obesity is defined as having a body mass index (BMI) ≥ 30%, with severe or morbid obesity as a BMI ≥ 40%. The age-adjusted prevalence of obesity in the United States is 42.4%, and is highest among non-Hispanic black adults.15 These patients have hyperactive, enlarged adipocytes that attract macrophages and together produce tumor necrosis factor alpha, adiponectin and IL-6, which increase systemic inflammation and insulin resistance by directly changing the sensitivity of its receptors.16
The complete list of obesity-related complications is exhaustive and includes T2DM, coronary heart disease, obstructive sleep apnea, osteoarthritis, stroke, and end-stage renal disease.16 Compared to nonobese subjects, obese patients are 1.81 times more likely to have periodontitis, per a systematic review.17 Weight gain was determined to be just as detrimental. Another systematic review found a 33% likelihood of developing periodontitis among subjects who became obese.18
The type and location of fat contribute differently to the rate of periodontal disease. Visceral fat, for example, is more “active” than peripheral fat in its release of proinflammatory markers and adipokines.16 In fact, central obesity is a more important parameter for disease onset and progression than BMI. According to a national cross-sectional study, central obesity is a significant risk factor for tooth loss. A waist circumference of 37 to 39 inches for men, and 31 to 34.6 inches for women is considered high risk for having or developing periodontitis.19 A similar study design in Korea supports these findings, as the authors suggest that a larger waist circumference was significantly associated with periodontitis compared to participants of normal weight.20
Obesity is one of several disorders that increase the risk for metabolic syndrome, which requires having at least three of five criteria relating to elevated waist circumference, triglycerides, blood pressure, fasting glucose and lipoprotein levels. A random effects meta-analysis identified an almost twofold risk of having periodontitis with metabolic syndrome, and an increasing magnitude of association with every additional component.21 Obesity also compromises treatment response. In a clinical trial, obese participants showed less probing depth reduction and clinical attachment gain, similar to that of smokers, compared to the normal weight group two months after nonsurgical therapy.22 Significantly better clinical results were noted three months after nonsurgical therapy with dietary weight loss intervention.23
Dental professionals have a unique opportunity to engage with patients more frequently about nutrition than other healthcare providers. Overweight or obese patients with periodontitis should be screened for T2DM and hypertension as part of their initial exam, and encouraged to visit their primary care provider. This is to help slow the rate of periodontitis, improve their responsiveness to interventional treatment, and prevent further systemic complications.
OTHER INFLAMMATORY CONDITIONS
Rheumatoid arthritis (RA) is an autoimmune disorder that causes inflammation of the joint lining in the hands, wrists and knees, eventually leading to damaged bone and cartilage, restricted movement, and organ failure. There are direct and indirect pathways that link RA and periodontitis.3 Porphyromonas gingivalis, a keystone periopathogen, alters local antigens in early RA. These autoantigens are then recognized by anti-citrullinated protein antibodies (ACPAs), a highly diagnostic marker of RA. In fact, ACPA status has been significantly associated with periodontitis.24
Periodontitis also affects the microvasculature and can result in erectile dysfunction (ED). After adjusting for confounders, a case-control study found a 74% prevalence of mild chronic periodontitis in ED patients, with the oral condition leading to a 2.17-fold greater likeliness of developing ED.25 In a randomized controlled trial, patients with periodontitis and ED had improved erectile function after scaling and root planing due to a short-term decrease in serum cytokines.26
CASE REPORT
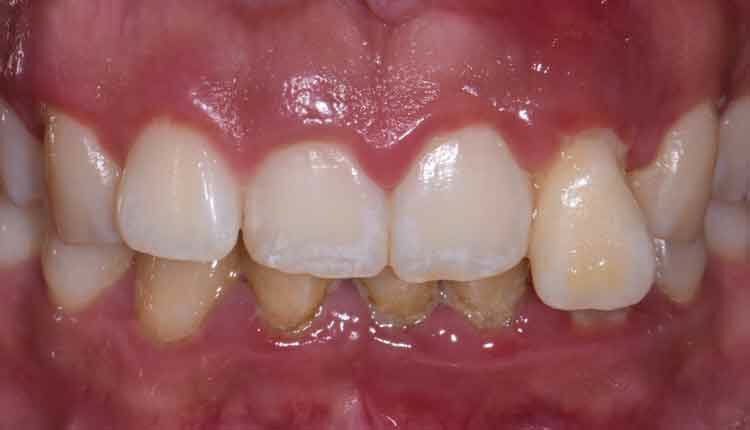
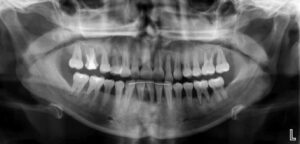
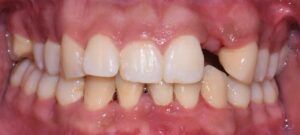
The following case outlines the interprofessional steps taken to treat severe periodontitis and its associated systemic conditions. A 24-year old Hispanic female presented to the graduate periodontics clinic at the University of Texas Health San Antonio School of Dentistry interested in comprehensive periodontal care (Figure 1 through Figure 10). She had uncontrolled T2DM and morbid obesity, yet no other comorbidities.
Her periodontal tissues were generally pink-red and loosely attached, with heavy plaque, calculus and debris noted throughout. She also presented with severe horizontal bone loss and vertical intrabony defects, multiple endo-periodontal lesions, furcation invasions, and secondary occlusal trauma. Probing depths ranged from 3 to 10 mm, with moderate bleeding on probing and suppuration; clinical attachment levels ranged from 1 to 11 mm. According to the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions, she was classified as having periodontitis, Stage IV, generalized, Grade C.27
After a thorough patient evaluation and review of various techniques and materials, a comprehensive treatment plan was developed, and consent was obtained. Nonsurgical and surgical procedures were performed using intravenous conscious sedation. The treatment plan included:
- Medical consultation to improve glycemic control.
- Full-mouth scaling and root planing, with extractions of periodontally hopeless teeth #10 and 15. She was prescribed amoxicillin 500 mg and metronidazole 500 mg every eight hours for eight days during the perioperative period.
- Reevaluation of clinical parameters and further assessment of oral hygiene compliance was completed 12 weeks after initial therapy.
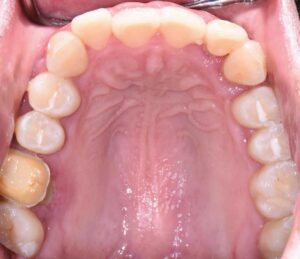
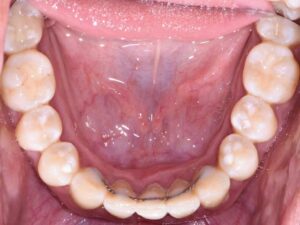
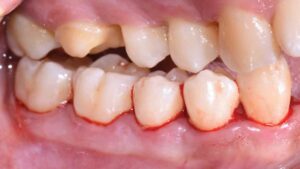
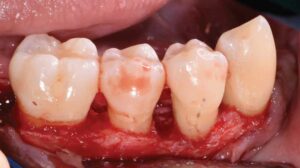
- Open flap debridement, with apically positioned flaps of the upper right, upper left, lower left, and lower right quadrants, and extractions of periodontally hopeless teeth #2, 4, 5, 19, 23, 24, 25 and 31 to arrest disease progression.
- Maxillary and mandibular interim removable dental prostheses to replace missing teeth, and a long-term provisional crown on #3 to stabilize her occlusion.
- Periodontal maintenance every three months, transitioning to a longer recall period based on stability and oral hygiene compliance.
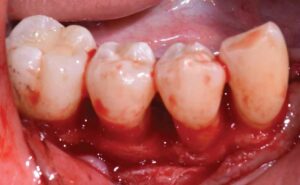
The patient was first referred to her physician for proper management of her T2DM due to diabetes-related symptoms and a casual blood glucose reading of 333 mg/dL at her initial exam. With nutritional counseling, weight loss, and medication adherence, her HbA1c decreased from 12.7% to 6.5% over six months. The patient continued care with her physician every three months.
Full-mouth scaling and root planing and extractions of teeth #10 and 15 were completed at one appointment under an eight-day antibiotic regimen to maximize the clinical efficacy of nonsurgical therapy. Site #10 was not simultaneously augmented for future implant placement due to limited patient finances, a lack of ridge height, and a chief goal of stabilizing her aggressive periodontal breakdown. While oral hygiene measures were reviewed in-depth, they were reinforced six weeks after initial therapy owing to continued poor plaque control. Periodontal reevaluation was performed after 12 weeks, at which time she presented with definite improvements in her self-care and clinical parameters.
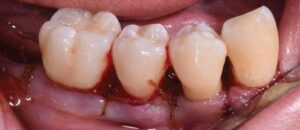
Surgical therapy was initiated to better visualize and debride the root surfaces, assess and modify the bony architecture, improve access for oral hygiene and maintenance, reduce pocketing and inflammation, gain clinical attachment, and slow/arrest disease progression. Open flap debridement with apically positioned flaps was performed in all four quadrants, with osseous recontouring as indicated. Periodontal regeneration was not attempted due to limited patient finances. The remaining periodontally hopeless teeth were extracted, with immediate delivery of maxillary and mandibular Essix retainers for esthetics. Interim removable dental prostheses and a long-term provisional crown #3 were later fabricated to stabilize her occlusion before proceeding with definitive removable appliances.
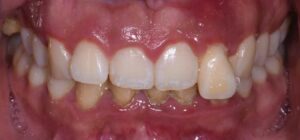
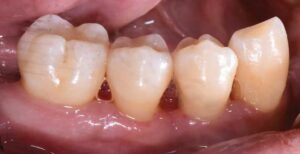
The patient continued in a supportive periodontal maintenance program every three months. Oral hygiene techniques were reviewed, and a prescription fluoride dentifrice was provided to reduce the incidence of root caries and sensitivity. Almost two years later, she presented with probing depths of 2 to 3 mm, with nonbleeding 4-mm pocketing in select posterior teeth. Her HbA1C values were routinely < 7%. Despite her initial presentation, she maintained most of her otherwise terminal dentition and renewed her quality of life.
SUMMARY AND CONCLUSIONS
The impact of periodontal medicine can truly be life-changing. Diabetes mellitus, cardiovascular disease, obesity and other inflammatory disorders can accelerate periodontal disease progression and diminish therapeutic response. As the links between periodontitis and systemic disease become more apparent, it is important to be mindful that association does not imply causation. An interprofessional approach to managing systemic conditions as part of periodontal care can improve clinical outcomes and patients’ overall well-being.
REFERENCES
- Shinjo T, Ishikado A, Hasturk H, et al. Characterization of periodontitis in people with type 1 diabetes of 50 years or longer duration. J Periodontol. 2019;90:565–575.
- Sczepanik, FS, Grossi ML, Casati M, et al. Periodontitis is an inflammatory disease of oxidative stress: we should treat it that way. Periodontol 2000. 2020;84:45–68.
- Emrich LJ, Shlossman M, Genco RJ. Periodontal disease in non-insulin-dependent diabetes mellitus. J Periodontol. 1991;62:123–131.
- Taylor GW, Burt BA, Becker MP, et al. Severe periodontitis and risk for poor glycemic control in patients with non-insulin-dependent diabetes mellitus. J Periodontol. 1996;67(Suppl 10):1085–1093.
- D’Aiuto F, Gkranias N, Bhowruth D, et al. Systemic effects of periodontitis treatment in patients with type 2 diabetes: a 12 month, single-centre, investigator-masked, randomised trial. Lancet Diabetes Endocrinol. 2018;6:954–965.
- Hsu YT, Nair M, Angelov N, Lalla E, Lee CT. Impact of diabetes on clinical periodontal outcomes following non–surgical periodontal therapy. J Clin Periodontol. 2019;46:206–217.
- Geisinger ML, Morris AB, Kaur M, et al. Glycemic control among patients with physician-managed type 2 diabetes. Gen Dent. 2018;66:52–55.
- Demmer RT, Desvarieux M. Periodontal infections and cardiovascular disease: the heart of the matter. J Am Dent Assoc. 2006;137(Suppl):14S–20S.
- Haraszthy VI, Zambon JJ, Trevisan M, Zeid M, Genco RJ. Identification of periodontal pathogens in atheromatous plaques. J Periodontol. 2000;71:1554–1560.
- Ridker PM, Cushman M, Stampfer MJ, Tracy RP, Hennekens CH. Inflammation, aspirin, and the risk of cardiovascular disease in apparently healthy men. N Engl J Med. 1997;336:973–979.
- 11. D’Aiuto F, Nibali L, Parkar M, Suvan J, Tonetti MS. Short-term effects of intensive periodontal therapy on serum inflammatory markers and cholesterol. J Dent Res. 2005;84:269–273.
- Taylor BA, Tofler GH, Carey HM, et al. Full-mouth tooth extraction lowers systemic inflammatory and thrombotic markers of cardiovascular risk. J Dent Res. 2006;85:74–78.
- Beck JD, Elter JR, Heiss G, Couper D, Mauriello SM, Offenbacher S. Relationship of periodontal disease to carotid artery intima-media wall thickness: the atherosclerosis risk in communities (ARIC) study. Arterioscler Thromb Vasc Biol. 2001;21:1816–1822.
- Bahekar AA, Singh S, Saha S, Molnar J, Arora R. The prevalence and incidence of coronary heart disease is significantly increased in periodontitis: a meta-analysis. Am Heart J. 2007;154:830–837.
- Hales CM, Carroll MD, Fryar CD, Ogden CL. Prevalence of Obesity and Severe Obesity Among Adults: United States, 2017–2018. NCHS Data Brief. Available at: https://www.cdc.gov/nchs/data/databriefs/db360-h.pdf. Accessed February 10, 2021.
- Suvan JE, Finer N, D’Aiuto F. Periodontal complications with obesity. Periodontol 2000. 2018;78:98–128.
- Suvan J, D’Aiuto F, Moles DR, Petrie A, Donos N. Association between overweight/obesity and periodontitis in adults. A systematic review. Obes Rev. 2011;12:e381–e404.
- Nascimento GG, Leite FR, Do LG, et al. Is weight gain associated with the incidence of periodontitis? A systematic review and meta-analysis. J Clin Periodontol. 2015;42:495–505.
- Kang J, Smith S, Pavitt S, Wu J. Association between central obesity and tooth loss in the non-obese people: results from the continuous National Health and Nutrition Examination Survey (NHANES) 1999–2012. J Clin Periodontol. 2019;46:430–437.
- Kim EJ, Jin BH, Bae KH. Periodontitis and obesity: a study of the Fourth Korean National Health and Nutrition Examination Survey. J Periodontol. 2011;82:533–542.
- Nibali L, Tatarakis N, Needleman I, et al. Clinical review: association between metabolic syndrome and periodontitis: a systematic review and meta-analysis. J Clin Endocrinol Metab. 2013;98:913–920.
- Suvan J, Petrie A, Moles DR, et al. Body mass index as a predictive factor of periodontal therapy outcomes. J Dent Res. 2014;93:49–54.
- Martinez-Herrera M, López-Domènech S, Silvestre FJ, et al. Dietary therapy and non-surgical periodontal treatment in obese patients with chronic periodontitis. J Clin Periodontol. 2018;45:1448–1457.
- Loutan L, Alpizar-Rodriguez D, Courvoisier DS, Finckh A, Mombelli A, Giannopoulou C. Periodontal status correlates with anti-citrullinated protein antibodies in first-degree relatives of individuals with rheumatoid arthritis. J Clin Periodontol. 2019;46:690–698.
- Martín A, Bravo M, Arrabal M, Magán-Fernández A, Mesa F. Chronic periodontitis is associated with erectile dysfunction. A case-control study in European population. J Clin Periodontol. 2018;45:791–798.
- Eltas A, Oguz F, Uslu MO, Akdemir E. The effect of periodontal treatment in improving erectile dysfunction: a randomized controlled trial. J Clin Periodontol. 2013;40:148–154.
- Caton JG, Armitage G, Berglundh T, et al. A new classification scheme for periodontal and peri-implant diseases and conditions — introduction and key changes from the 1999 classification. J Clin Periodontol. 2018;45(Suppl 20):S1–S8.
From Decisions in Dentistry. March 2021;7(3):25–28,31.




