
Oral Complications of Medication Usage
An overview of oral side effects of commonly used drugs, and recommendations for prevention and management of these symptoms.
This course was published in the December 2017 issue and expires December 2020. The author has no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
OBJECTIVES
- Explain the prevalence of medication usage among the U.S. population, and common oral side effects.
- Describe key properties of saliva and mechanisms that can affect salivary flow.
- List risks and adverse oral sequelae associated with drug-induced xerostomia.
- Discuss the variety of drugs associated with oral side effects.
Patients who have multiple medical conditions often present for dental treatment with complex medical and pharmacological histories. Recent statistics from the U.S. Centers for Disease Control and Prevention (CDC) reveal that 49% of the U.S. population has taken at least one medication, 23% has taken three or more, and 12% has taken five or more prescription drugs within the last 30 days.1 Medication usage typically increases with age, and older adults present with both polypharmacy and polyherbacy. The more medications consumed, including herbal and dietary supplements (HDS), the greater the risk for a drug-drug or drug-HDS interaction.2 Clinicians should routinely assess medicated patients for complaints and/or presentation of adverse drug effects. This article will review common oral side effects of medication use and recommendations for prevention and management.
Each manufacturer reports known side effects of medication use in the drug monograph. Side effects are organized and reported by the body system affected, with adverse oral side effects listed under the gastrointestinal system. Adverse oral drug effects include xerostomia, oral ulcerations, gingival hyperplasia, taste alteration, lichenoid drug reaction and mucositis, which are discussed here. Others include dysesthesias (altered sensations), osteonecrosis, malignancy, intraoral pigmentation and angioedema.3 Clinicians should consult a drug reference resource to identify whether a patient’s medication use is negatively impacting oral health. With the notable exception of xerostomia, generally, oral side effects occur in only a small minority of users.
DRUG-INDUCED XEROSTOMIA
Drug-induced xerostomia or dry mouth is the most frequently reported oral side effect among older adults, who may present with multiple comorbidities and depression.4,5 A recent review suggests the incidence of xerostomia increases with the number and dose of medications taken.6
Salivary secretion is stimulated via excitation of both the parasympathetic and sympathetic divisions of the autonomic nervous system. Salivary gland cell-surface receptors receive stimuli from neurotransmitters, which transmit signals within the cells that comprise the glands. Serous saliva, which is high in volume and ions, but low in protein, comprises the majority of the volume of saliva. Parasympathetic stimulation also results in vasodilation of the blood vessels that support salivary glands. Serous saliva is essential for the breakdown of the food bolus, swallowing and digestion.7,8
Stimulation of the sympathetic nervous system produces saliva that is high in proteins, but low in volume. Stimulation produces vasoconstriction of the blood vessels that supply the glands, reducing the rate of salivary secretion and causing glandular cells to produce more mucinous saliva. Sympathetic stimulation inhibits flow rate due to central nervous system influences from the higher centers of the brain that control the salivary centers, thereby diminishing reflex activity. Mucinous saliva makes the mouth feel sticky, and, for some individuals, the oral cavity may feel dry. An example of this sensation is the dry mouth that is perceived while feeling anxious, nervous or frightened.7,8
Symptoms of drug-induced xerostomia vary with the type of medication used.8 Drugs alter the autonomic nervous system and production and secretion of saliva.9 Cholinergic drugs bind to muscarinic receptors in the parasympathetic nervous system to stimulate salivary flow and are approved by the U.S. Food and Drug Administration (FDA) for the treatment of Sjögren’s syndrome and post-radiation of head and neck cancers. Examples include pilocarpine and cevimeline.10 Conversely, many patients use anticholinergic medications that oppose the normal effects of the parasympathetic nervous system.9 Affected patients may complain of xerostomia symptoms, as these drugs significantly reduce the volume of serous saliva. It is important to note that most patients do not complain of xerostomia — which is the perception of dry mouth — until half of the normal salivary flow is lost.11 Patients who have lost significant parotid flow often complain of feeling parched, and clinicians may notice intraoral signs that include enamel demineralization and/or caries, increased biofilm, gingival disease, food packing, poor oral cleansing and halitosis. Medications that oppose the parasympathetic nervous system are used to treat a variety of systemic conditions and include antihypertensives, antidepressants and drugs for treating chronic obstructive pulmonary disease, notably emphysema.
Drugs that alter the sympathetic nervous system — including amphetamines used for the treatment of attention deficit disorder/attention deficit hyperactivity disorder — are taken by patients of all ages, including children. These individuals may exhibit ropey saliva and/or complain of a “sticky” feeling in the oral cavity. Other sympathomimetics include decongestants, appetite suppressants and bronchodilators.7,8,12,13
In addition, drugs may produce symptoms of xerostomia through other mechanisms that are not neutrally mediated. Diuretics contribute to a systemic loss of water, resulting in dehydration without any alteration in salivary flow rates. Some drugs produce vasoconstriction of salivary gland vessels. Inhaled medications have been reported to produce the sensation of dry mouth without actually altering salivary flow rates. Many drugs have been reported in the literature as contributing to xerostomia without adequate research to determine if or how salivary flow rates are affected.8
CONSEQUENCES OF CHRONIC XEROSTOMIA
Due to the loss of natural salivary immunoglobulins, patients with chronic xerostomia are at high risk for a variety of oral infections, including bacterial (e.g., caries and gingival disease), fungal and viral infections.14 Changes in the chemical composition of saliva include a drop in oral pH due to the loss of buffering by sodium bicarbonate, causing an acid shift that promotes demineralization and caries. The acidity of residual saliva and biofilm also contributes to dentinal hypersensitivity. In addition, changes in salivary proteins decrease efficacy of the breakdown of the food bolus during chewing, which may contribute to digestive complaints. Inadequate lubrication leads to oral trauma from foods, oral appliances and prosthetics. Loss of lubrication contributes to difficulty with chewing and swallowing, which increases aspiration and choking risks. Clinicians have likely observed that many of their patients take medications for gastroesophageal reflux disease that may be related to chronic xerostomia. Given the large number of individuals affected by xerostomia, it is not surprising that proton pump inhibitors (e.g., omeprazole and esomeprazole) are among the most commonly prescribed medications in the United States.15
Recurrent fungal infections may be observed in patients with xerostomia, who may present with a pseudomembranous appearance (i.e., bright red with overlying white pseudomembrane), hyperkeratotic denture stomatitis and/or symptomatic geographic tongue. Angular chelitis is frequently observed, especially in patients with loss of vertical dimension. Intraoral fungal infections are associated with use of steroid inhalers. Recently, the threat level for Candida has been moved up to “serious” by the CDC due to its emergence as a resistant organism.16 The systemic azole antifungals include fluconazole, itraconazole and ketoconazole. In 2013, the FDA changed the approval status of ketoconazole for the treatment of Candidiasis, as the drug is associated with risks for liver injury, adrenal insufficiency and serious drug interactions.17 Widespread use of systemic azole antifungals has been attributed to promoting resistance. Although systemic antifungal agents are anecdotally preferred by patients, compatibility must be checked with concurrent use of other medications due to the risk for drug interactions and growing concerns for promoting resistance.
Topical agents, such as nystatin, should be used for the treatment of oral fungal infections. The systemic azole antifungals should be reserved for extensive mucocutaneous infections. Compliance with topical agents is often poor due to the frequency of application and duration of treatment, so the need for therapy must be reinforced. Dentures and oral appliances must be treated as well, and toothbrushes and lip balms/lipsticks disposed, to reduce risk for reinfection. Twice-daily use of an antiseptic mouthrinse has been shown to reduce denture stomatitis (with the dentures out), as both chlorhexidine and essential oil mouthrinse have been shown to kill seven species of Candida.18
INTERVENTIONS
Numerous interventions exist to assist patients with restoring comfort and function, and to reduce adverse oral sequelae associated with chronic xerostomia. Patients should be encouraged to remain hydrated. While salivary replacement agents help temporarily replenish moisture and provide lubrication, not all agents are the same, and clinicians are discouraged from using a “one-size-fits-all” approach with product recommendations. For example, many agents contain carboxymethylcellulose to simulate the viscosity of natural saliva, as well as glycerin, which coats the tissues and creates a “slippery” feeling, reducing friction. However, patients who complain of sticky, mucinous saliva may not tolerate these products well, and may prefer products without glycerin.
Salivary replacement agents are available in a variety of forms, including lozenges, sprays, rinses and gum. Their effects are short-lived and typically last for only a few hours. To date, the quality of the evidence supporting the efficacy of these products is weak.19 Compliance is problematic for patients who must apply them frequently throughout the day. Further, salivary replacement agents often contain a sugar alcohol, such as xylitol, as a sweetener and anticaries agent. Given that these agents must be used frequently and are ingested, dose-control is an important consideration, as the sugar alcohols cannot be metabolized, resulting in adverse digestive effects. Clinicians should encourage patients to try a variety of products to identify which provide the greatest symptomatic relief.
Supplemental fluoride therapy is also recommended as a caries-preventive agent in this patient population. Various caries risk assessment models identify drug-induced xerostomia as a major risk factor.20,21 Topical fluoride recommendations from the American Dental Association can be used to determine the most age-appropriate and risk-appropriate fluoride interventions, including dosage and frequency of application.22 Several commercial fluoride products are available over the counter or by prescription that are specially-formulated for patients with xerostomia. To date, nonfluoride remineralization therapies lack sufficient evidence to substantiate their benefits,23 thus, clinicians should use their judgment to determine whether the addition of these products may be appropriate to reduce caries risk.
Good oral hygiene is essential for patients with chronic xerostomia. Practitioners should recommend a variety of oral aids to assist with mechanical disruption of biofilm. These include oral irrigators, air flossers and power brushes. Sonic toothbrushing has been shown to stimulate salivary flow.24 Patients with chronic dry mouth benefit greatly from the daily use of antiseptic mouthrinses and therapeutic dentifrices to reduce biofilm and resultant bacterial infections.18 In addition, dentifrices containing sodium bicarbonate may help raise oral pH levels.
APHTHOUS ULCERS AND GINGIVAL HYPERPLASIA
Several drug classes have been shown to cause aphthous ulcers.25 Although it may not be possible to switch the offending drug to another from a different medication class, if frequent ulceration affects the patient’s oral health-related quality of life, the prescribing physician should be contacted to determine feasibility of an alternate therapy. Topical over-the-counter and prescription anesthetics may help improve comfort.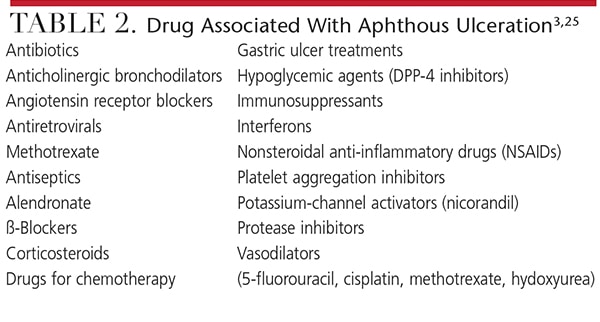
Gingival hyperplasia may be observed in patients taking anticonvulsants, calcium channel blockers and immunosuppressants used to prevent organ transplant rejection.3 While it is difficult for patients to perform adequate oral hygiene with hyperplastic tissues, effective self-care may reduce the extent and severity of the lesion. Susceptible individuals carry a genetic risk for this drug manifestation.26
TASTE ALTERATION
More than 250 drugs have been associated with altering taste (dysgeusia) and smell (dysosmia). Older adults frequently complain that food tastes bland or they cannot taste certain foods, resulting in behaviors such as adding salt, over-seasoning foods or simply not eating regularly. Several mechanisms of drug-induced taste alteration have been proposed, including secretion of the drug into saliva and gingival crevicular fluid, diminished turnover of taste buds, concentration of electrolytes in saliva, and altered neuronal and/or neurotransmitter function.3 Saliva is necessary to carry tastants over the taste buds, thus, patients may have multiple contributing factors to explain distortion of taste. Clinicians should assess patients’ nutritional status — including eating habits, use of salt and food selection — to ensure proper intake of essential nutrients and adequate consumption. A referral to a registered dietician may be warranted.
Patients complaining of an undesirable “aftertaste” may adopt a variety of behaviors to mask the taste. Clinicians should assess use of candy, gum, mints and excessive mouthwash, all of which may occur in this population. Patients should be assessed for oral signs of these behaviors and counseled accordingly.
LICHENOID DRUG REACTION
Lichenoid reaction is a delayed hypersensitivity reaction to a drug. Clinically, this condition is often confused with lichen planus, an autoimmune disease. Patients may or may not be symptomatic. There is a lack of consensus about diagnostic criteria because, with some cases, the condition may not resolve after the drug has been discontinued.6,27 Clinicians should assess a “new” presentation of lichen planus and correlate onset with the timing of the introduction of a medication since the last dental visit. Although many other drugs have also been implicated, common medications associated with lichenoid drug reaction include nonsteroidal anti-inflammatory drugs and antihypertensive agents.
MUCOSITIS
Radiation therapy and chemotherapy used for cancer treatment are associated with causing oral mucositis, which can occur directly as a burn from radiation and from exposure to the toxic drugs used during chemotherapy that are secreted into the oral cavity via saliva. Drugs used for chemotherapy cause systemic immunosuppression, which reduces salivary immunoglobulins and increases risk for oral infections. Mucositis causes significant pain and alters the patient’s ability to eat, swallow and speak.
Several systematic reviews have examined which agents are most efficacious for the treatment of mucositis.28–30 Efficacy varies depending upon the timing of the intervention and types of chemotherapy drugs used. There is insufficient and weak-quality evidence to support the use of chlorhexidine gluconate or povodine iodine for reducing either chemotherapy- or radiation therapy-induced mucositis.28–30 Current evidence supports the benefit of sucking on ice chips (i.e., cryotherapy).28–30 Emerging evidence suggests that use of a low-level laser may be beneficial in preventing mucositis, but more research is needed to support this intervention.31,32
“Magic mouthwash” describes a mixture of a topical anesthetic (e.g., over-the-counter liquid diphenhydramine or prescription lidocaine) with a coating agent. Patients rinse prior to eating and at bedtime. The coating allows the topical anesthetic agent to remain in contact with the tissues so the patient can take in nutrition, often in the form of a fortified liquid supplement. It is essential to help patients control pain and maintain nutrition during cancer treatment. Rinsing and swallowing the mouthwash at bedtime may also help patients sleep due to the sedative side effect of diphenhydramine.
CONCLUSION
Adverse oral drug effects negatively influence oral comfort, function and quality of life. Clinicians should assess their patients’ medication use to determine risks for oral complications. Utilizing recommended interventions for oral adverse effects associated with medications can prevent oral infections, reduce discomfort, facilitate chewing and swallowing, and promote optimal oral health. Interprofessional collaboration with physicians and registered dieticians may be warranted to ensure appropriate strategies are in place for effectively managing medications.
REFERENCES
- U.S. Centers for Disease Control and Prevention. National Center for Health Statistics. Health, United States, 2016 With Chartbook on Long-term Trends in Health. Available at: cdc.gov/nchs/data/hus/hus16.pdf#079. Accessed November 8, 2017.
- Loya AM, González-Stuart A, Rivera JO. Prevalence of polypharmacy, polyherbacy, nutritional supplement use and potential product interactions among older adults living on the United States-Mexico border: a descriptive, questionnaire-based study. Drugs Aging. 2009;26:423–436.
- Yuan A, Woo SB. Adverse drug events in the oral cavity. Oral Surg Oral Med Oral Pathol Oral Radiol. 2015;119:35–47.
- Tan EC, Lexomboon D, Sandborgh-Englund G, Haasum Y, Johnell K. Medications that cause dry mouth as an adverse effect in older people: a systematic review and metaanalysis. J Am Geriatr Soc. October 26, 2017. Epub ahead of print.
- Viljakainen S, Nykänen I, Ahonen R, et al. Xerostomia among older home care clients. Community Dent Oral Epidemiol. 2016;44:232–238.
- Aliko A, Wolff A, Dawes C, et al. World Workshop on Oral Medicine VI: clinical implications of medication-induced salivary gland dysfunction. Oral Surg Oral Med Oral Pathol Oral Radiol. 2015;120:185–206.
- Porter SR, Scully C, Hegarty AM. An update of the etiology and management of xerostomia. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2004;97:28–46.
- Wolff A, Joshi RK, Ekström J, et al. A guide to medications inducing salivary gland dysfunction, xerostomia, and subjective sialorrhea: a systematic review sponsored by the World Workshop on Oral Medicine VI. Drugs R D. 2017;17:1–28.
- Villa A, Wolff A, Narayana N, et al. World Workshop on Oral Medicine VI: a systematic review of medication-induced salivary gland dysfunction. Oral Dis. 2016;22:365–382.
- Miranda-Rius J, Brunet-Llobet L, Lahor-Soler E, Farré M. Salivary secretory disorders, inducing drugs, and clinical management. Int J Med Sci. 2015;12:811–824.
- Dawes C. Physiological factors affecting salivary flow rate, oral sugar clearance, and the sensation of dry mouth in man. J Dent Res. 1987;66(Spec No):648–653.
- Sreebny LM, Schwartz SS. A reference guide to drugs and dry mouth — 2nd edition. Gerodontology. 1997;14:33–47.
- Nähri TO, Meurman JH, Ainamo A. Xerostomia and hyposalivation: causes, consequences and treatment in the elderly. Drugs Aging. 1999;15:103–116.
- Dawes C, Pedersen AM, Villa A, et al. The functions of human saliva: a review sponsored by the World Workshop on Oral Medicine VI. Arch Oral Biol. 2015;60:863–874.
- Brown T. 100 Best-Selling, Most Prescribed Branded Drugs Through March. Available at: medscape.com/viewarticle/844317. Accessed November 9, 2017.
- U.S. Centers for Disease Control and Prevention. Antifungal Resistance. Available at: cdc.gov/fungal/antifungal-resistance.html. Accessed November 9, 2017.
- U.S. Food and Drug Administration. FDA Drug Safety Communication: FDA limits usage of Nizoral (ketoconazole) oral tablets due to potentially fatal liver injury and risk of drug interactions and adrenal gland problems. Available at: www.fda.gov/Drugs/DrugSafety/ucm362415.htm. Accessed November 7, 2017.
- DePaola LG, Spolarich AE. Safety and efficacy of antimicrobial mouthrinses in clinical practice. J Dent Hyg. 2007;81(Suppl, pt 2):13–25.
- Riley P, Glenny AM, Hua F, Worthington HV. Pharmacological interventions for preventing dry mouth and salivary gland dysfunction following radiotherapy. Cochrane Database Syst Rev. 2017:7:CD012744.
- American Dental Association. Caries Risk Assessement Form. Available at: ada.org/~/media/ADA/Science%20and%20Research/Files/topic_caries_over6.ashx. Accessed November 9, 2017.
- Doméjean S, White JM, Featherstone JD. Validation of the CDA CAMBRA caries risk assessment — a six-year retrospective study. J Calif Dent Assoc. 2011;39:709–715.
- American Dental Association. Professionally Applied and Prescription-strength, Home-use Topical Fluoride Agents for Caries Prevention Clinical Practice Guideline (2013). Available at: ebd.ada.org/en/evidence/guidelines/topical-fluoride. Accessed November 9, 2017.
- Rethman MP, Beltrán-Aguilar ED, Billings RJ, et al. Nonfluoride caries-preventive agents: executive summary of evidence-based clinical recommendations. J Am Dent Assoc. 2011;142:1065–1071.
- Papas A, Singh M, Harrington D, et al. Stimulation of salivary flow with a powered toothbrush in a xerostomic population. Spec Care Dentist. 2006;26:241–246.
- Jinbu Y, Demitsu T. Oral ulcerations due to drug medications. Japan Dent Sci Rev. 2014;50:40–46.
- Seymore RA, Thomason JM, Ellis JS. The pathogenesis of drug-induced gingival overgrowth. J Clin Periodontol. 1996;23:165–175.
- Fortuna G, Aria M, Schiavo JH. Drug-induced oral lichenoid reactions: a real clinical entity? A systematic review. Eur J Clin Pharmacol. September 13, 2017. Epub ahead of print.
- McGuire DB, Fulton JS, Park J, et al. Systematic review of basic oral care for the management of oral mucositis in cancer patients. Support Care Cancer. 2013;21:3165–3177.
- Peterson DE, Ohrn K, Bowen J, et al. Systematic review of oral cryotherapy for management of oral mucositis caused by cancer therapy. Support Care Cancer. 2013;21:327–332.
- Yarom N, Ariyawardana A, Hovan A, et al. Systematic review of natural agents for the management of oral mucositis in cancer patients. Support Care Cancer. 2013;21:3209–3221.
- Migliorati C, Hewson I, Lalla RV, et al. Systematic review of laser and other light therapy for the management of oral mucositis in cancer patients. Support Care Cancer. 2013;21:333–341.
- Oberoi S, Zamperlini-Netto G, Beyene J, Treister NS, Sung L. Effect of prophylactic low level laser therapy on oral mucositis: a systematic review and meta-analysis. PLoS One. 2014;9:e107418.
FEATURED IMAGE BY DNY59 / ISTOCK / GETTY IMAGES PLUS
From Decisions in Dentistry. December 2017;3(12):30—33.




