 DR_MICROBE/ISTOCK/GETTY IMAGES PLUS
DR_MICROBE/ISTOCK/GETTY IMAGES PLUS
Manifestations of Genetic Disorders in Periodontal Care
Genetic disorders can negatively impact oral health, and indications for periodontal care may be the first sign of an undiagnosed genetic disease.
This course was published in the May 2020 issue and expires May 2023. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Identify genetic disorders that are associated with periodontal disease and other oral conditions, including the incidence of oral manifestations in individuals with neurofibromatosis Type I.
- Describe the clinical significance of the association between genetic disorders and oral health issues.
- Discuss various means of detecting risk for genetic disorders, as well as their associated oral conditions.
Until recently, the relationship between genetic disorders and periodontal disease was unclear. As our understanding of the underlying mechanisms and systemic pathophysiology of numerous genetic disorders broadens, new research has uncovered a possible association between these types of disorders and periodontal disease. Of interest to clinicians who provide periodontal care, this paper reviews several genetic disorders that have periodontal manifestations, including Marfan syndrome, Papillon Lefèvre syndrome, Ehlers-Danlos syndrome Type VIII, neurofibromatosis Type I, and Alzheimer’s disease.
In light of the fact periodontal disease may be the first detectable sign of an undiagnosed genetic disorder and/or indicate its activity or progression, it is important for dental practitioners to be familiar with genetic disorders and their impact on the periodontal tissues.
MARFAN SYNDROME
Marfan syndrome (MFS) is an autosomal dominant genetic disorder caused by mutations in the fibrillin-1 gene that affect the structural integrity and the function of connective tissue. Fibrillin mutations manifest wherever microfibrils are present, such as the skeletal system, lungs, blood vessels and skin. Since the periodontium is rich in connective tissue, oral manifestations of MFS are common. Systemically, MFS is associated with cardiovascular disease complications that increase morbidity and mortality for MFS-affected individuals.
According to Suzuki et al,1 periodontal pathogens provoke a host immuno-inflammatory response characterized by the release of cytokines and matrix metalloproteinases. The release of oral pro-inflammatory mediators, such as TGF-β, can potentially influence the onset and development of systemic inflammatory disease. As such, it is especially noteworthy that MFS-affected individuals exhibit elevated levels of TGF-β in the serum and saliva.1
Although an inflammatory mediator, conflicting studies challenge the role of TGF-β in collagen synthesis. Suzuki et al1 concluded TGF-β not only stimulates the synthesis of the connective tissue matrix in periodontal ligament (PDL) cells, but also inhibits the activity of matrix metalloproteinases. However, in other studies TGF-β has been implicated in periodontal breakdown. Ohshima et al2 demonstrated that TGF-β enhances matrix degradation in gingival fibroblasts in patients with periodontitis, thereby explaining the inhibitory role of TGF-β in collagen synthesis.
Other MFS-associated oral manifestations include hypoplastic enamel spots, root deformity, abnormal pulp shape, pulpal inclusions, calculus and gingival inflammation, which increase the risk for dental disorders. Additionally, Suzuki et al1 found that individuals with MFS and cardiovascular disease exhibited a higher incidence of periodontitis compared to healthy controls. Moreover, it was discovered that serum levels of anti-Prevotella intermedia antibodies were significantly lower in individuals with MFS and cardiovascular disease compared to controls, thus suggesting these individuals may have weaker immune systems that are unable to adequately respond to periodontal pathogens.1
Another interesting finding is the effect of MFS on the PDL, which is structurally damaged due to a mutation of the fibrillin-1 gene. Studies conducted to clarify the function of elastic fibers in the PDLs of healthy patients as compared to PDL cells of individuals with MFS reported a mutation in the cbEGF domain, which is a critical alteration of the fibrillin-1 gene.3,4 As a result, mutations of the fibrillin-1 gene affect the normal development and function of the PDL fibers and make it more susceptible to periodontal breakdown.
EHLERS-DANLOS SYNDROME
Clinically and genetically, Ehlers-Danlos syndrome (EDS) is a heterogeneous connective tissue disorder characterized by variable degrees of skin hyperextensibility, fragility, scarring, minimal-to-moderate joint hypermobility (usually limited to the digits), and increased likelihood of bruising upon light trauma. The Type VIII form of EDS is a rare autosomal dominant disorder that not only has the aforementioned signs and symptoms, but also exhibits periodontal manifestations. Rahman et al5 reported that EDS is a disorder of the metabolism of fibrillar collagen. Mao and Bristow6 demonstrated that gene mutations (COL5A1 or COL5A2) play a role in the classic EDS presentation. Additional mutations (tenascin-X) have been reported, suggesting effects beyond that of collagen metabolism alone.7 Currently, there are no consistent molecular or biochemical markers that can detect EDS Type VIII. Unlike other types of EDS, severe early onset periodontitis is a primary oral feature of EDS Type VIII.5
Rahman et al5 examined the dental features, dermatological features and other clinical abnormalities consistent with the diagnosis of EDS in a large family consisting of five generations. The investigation showed demonstrable gingival recession and severe periodontal inflammation first identifiable in childhood, with rapid progression through adolescence and adulthood. Radiographs of selected family members indicated progressive loss of the hard and soft tissues of the periodontium, leading to edentulism.5 They identified a candidate gene on chromosome 12, but this was not consistent across the families, suggesting that EDS Type VIII is a genetically heterogeneous disorder.
PAPILLON-LEFÈVRE SYNDROME
Papillon-Lefèvre syndrome (PLS) is a rare autosomal recessive keratodermal disorder that can manifest as periodontitis. Affected individuals exhibit a homozygous mutation within the gene that encodes lysosomal protease cathepsin C (CTSC), which is mainly expressed in epithelial regions and on various immune cells (such as neutrophils and macrophages). As such, any genetic alterations in the CTSC gene will lead to the two distinguishing clinical hallmarks of PLS. The first is a deregulation of the coordination and function of many serine proteases, which are vital to host defense and increase susceptibility to periodontitis, especially at a young age. Second, since CTSC is expressed on epithelial cells, mutations of the CTSC gene will lead to gross thickening of the palmoplantar hyperkeratosis. Hence, diffuse palmoplantar keratosis and precocious (early onset) periodontitis are the two distinguishing clinical hallmarks of PLS.
Ragunatha et al8 detailed a case involving a male child who was born into a consanguineous family, which increased the risk for homozygous inheritance of the CTSC gene. At 6 months of age, dry, scaly hyperkeratotic thickening of the palms and soles was noted, which spread to the elbows and gluteal cleft. While the primary teeth developed normally, the patient experienced ongoing recurrent toothaches, severe halitosis, and progressive swelling of the gingiva, which eventually led to severe periodontitis by age 12. Periodontal care, including oral antibiotics and 20 mg/day of isotretinoin, resulted in significant dental and dermatologic improvements. A DNA sample confirmed homozygous nonsense mutations of the CTSC gene, which verified the diagnosis of PLS.
A late-onset form of PLS has also been reported. In some cases, the CTSC homozygous mutation has been completely absent, therefore presenting as heterozygous. This heterozygous presentation indicates there are variations of the cathepsin C pathways induced by single mutations of the CTSC gene that may contribute to the variable phenotype in late-onset periodontitis.
As a serious disorder with physical and psychosocial manifestation, PLS must be promptly identified and managed by a team of medical and dental providers. Otherwise, this recessive disorder may result in children losing all their teeth by adolescence, which has psychological and functional consequences. Features that should be further evaluated include hyperkeratosis, scaling skin lesions, gingival inflammation, and premature loss of incisors around early adolescence. This should be confirmed by a thorough clinical assessment, detailed patient and family history (which is important for a consanguineous family), physical examination, and genetic testing performed and assessed by an interdisciplinary team consisting of medical and dental practitioners.8
There is debate as to whether PLS is a separate and distinct disease entity. In a two-year study, Preus9 monitored two PLS-affected siblings and their family dog, who all exhibited rapidly destructive periodontitis. A high serum antibody titer against Aggregatibacter actinomycetemcomitans was discovered in the two affected children. This species, which was isolated in more than 50% of the diseased periodontal sites, suggested both patients were recently exposed to A. actinomycetemcomitans. Identifying both genetically and phenotypically identical clones of A. actinomycetemcomitans within the family’s environment led to the discovery the strains of bacteria were spread to the children by the dog.9 Treatment included a tetracycline regimen, along with concomitant extraction of all periodontally involved teeth.
Following periodontal care, the siblings and dog recovered from their rapidly destructive periodontitis. This finding led Preus9 to question the legitimacy of PLS as a distinct disease entity, which is different from other palmoplantar disorders. If the administration of antibiotics is sufficient to protect against A. actinomycetemcomitans, this may offer evidence PLS is nothing more than a dermatologically inherited disorder. Therefore, A. actinomycetemcomitans may, in fact, be the environmental trigger that causes periodontal infection, which is separate from the palmoplantar hyperkeratotic manifestations of this disorder. Despite Preus’ findings, it is still widely held that periodontitis and palmoplantar keratosis are distinguishing features of PLS.
NEUROFIBROMATOSIS
Neurofibromatosis is an autosomal dominant neurocutaneous syndrome “characterized by multiple cutaneous lesions and tumors of the central and peripheral nervous system.”10 Individuals with neurofibromatosis Type I (NF-1) exhibit oral manifestations in nearly 72% of cases.11 Figures 1A and 1B illustrate the extraoral and intraoral manifestations associated with this disorder.

Far more common than PLS and EDS Type VIII, NF-1 has an incidence of 1 in 3500 individuals worldwide. Although NF-1 primarily affects the skin and bones (as noted), it can also have oral manifestations. Javed et al10 reported that affected individuals exhibit unilateral non-tender enlargement of the gingiva, periodontitis, impacted and supernumerary teeth, morphological changes in teeth, and an Angle Class III molar relationship. Patients with NF-1 also tend to have poor and irregular oral hygiene, which can lead to gingival bleeding and excessive plaque. While present, oral melanotic pigmentation is an infrequent manifestation found in individuals with NF-1.10 However, it is noteworthy that oral pigmentation may appear unesthetic and contribute to psychosocial insecurities in those affected by the disorder.
Given the high incidence of this genetic disorder, there is great value in reinforcing proper oral hygiene in this patient population. Furthermore, while there is a tendency to focus only on extraoral features of NF-1, such as cafè-au-lait spots and Lisch nodules, the intraoral manifestations should not be overlooked. Similar to the findings of Ghalayani and colleagues,11 Shapiro et al12 noted that more than two-thirds of individuals with NF-1 have oral manifestations. Accordingly, oral examinations that assess patients’ restorative and periodontal needs are critical.
ALZHEIMER’S DISEASE
Alzheimer’s disease (AD) is a neurodegenerative disorder characterized by a progressive decline in memory, judgment and cognitive skills that slowly drains an individual’s ability to perform everyday tasks. This condition may also be associated with higher levels of inflammatory markers and antibodies in individuals with periodontal disease.13 As such, periodontal inflammation may be associated with brain inflammation, neurodegeneration and AD. In a 20-year longitudinal study involving 152 Danish 50-year-old subjects participating in the Golstrop Aging Study, Kamer et al13 found that patients with periodontal inflammation at baseline were nine times more likely to have lower digital symbol test (DST) scores at age 70 than those who had little or no periodontal inflammation at baseline. The strong association between periodontal inflammation and low DST scores held true even when taking other risk factors into account, such as cigarette smoking, obesity, and tooth loss unrelated to periodontal inflammation. Compared to subjects with no periodontal inflammation, a low DST score correlated to a higher incidence of periodontal inflammation, and therefore lower cognition. These findings point to a possible correlation between periodontal disease and late-onset AD. Furthermore, it suggests DST scores may have predictive value in forecasting the risk of AD in periodontally diseased individuals.
According to Kamer et al,13 specific pathogens contribute to the “initiation, maintenance and progression” of plaque biofilm and periodontal disease. As the biofilm matures, the periodontal bacteria thrive while simultaneously defending and protecting the ecosystem against foreign antigen invasion via exogenously derived antibacterial and host defense mechanisms. Potentially, the periodontal infection can metastasize to other areas distant from the oral cavity, triggering an inflammatory response that damages extraoral tissue. This relationship between inflammation and tissue destruction is known as the inflammatory hypothesis (Figure 2).
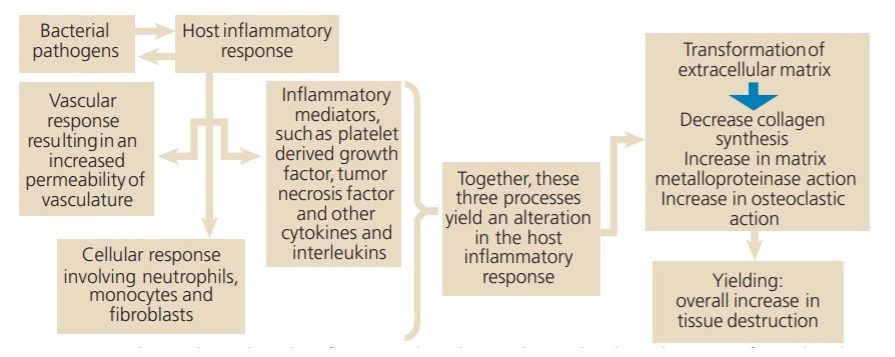
With respect to AD, Abbayya et al14 proposed two biological mechanisms that describe the interaction between periodontal disease and inflammatory markers. The first centers on the interaction between periodontal pathogens and host response that increases the level of pro-inflammatory cytokines. This initiates a cascade of inflammatory molecules that triggers systemic inflammation and, soon after, peripheral inflammation. These molecules have been shown to be capable of compromising the blood/brain barrier and entering cerebral areas. This activates the main inflammatory molecules within the brain — the microglial cells — which have the ability to cause neuronal damage via their pro-inflammatory mechanisms.
The second mechanism suggested by Abbayya et al14 postulates that oral microorganisms are capable of invading the brain. Irrespective of the entrance of these microorganisms, their products are capable of initiating an inflammatory response within the central nervous system. It has been generally accepted that inflammation within the central nervous system can result in cognitive decline.14 A few of the inflammatory mediators illustrated in Figure 2 (e.g., TNF-∝) have been implicated as playing contributing roles in the onset and progression of periodontal disease and AD.
Since there is no cure for AD, the emphasis is on preventive therapy. In a six-month study of 29 adults with mild AD, patients were classified as having gingivitis or periodontitis at baseline.15 Following periodontal care, the investigators reported reduced orofacial pain, improved mandibular function (including mastication and general movements), decreased periodontal inflammation, and improved quality of life. This study underscores the importance of prevention in mitigating the repercussions of the correlations between periodontal disease and AD.
CONCLUSIONS
Ongoing research is providing a clearer picture of the possible association between genetic disorders and periodontal disease. Thus, understanding the possible link between genetic disorders and periodontal health will be crucial as science continues to probe the etiology of these two conditions. Further understanding may help to define potential new mechanisms in disease development, including the role of periodontal health in relation to genetic diseases (and vice versa).
While each of these disorders exhibits distinct systemic and oral manifestations, the genetic disorders reviewed in this article share two common themes. First, oral infections — such as periodontitis — are manageable, and thus are modifiable risk factors for many of the incurable genetic disorders. The second theme is the inheritability of these conditions. Hence, both dentists and physicians should view genetic testing as another way to assess the risk of transmitting disease traits to offspring. Clinicians should also understand that due to the variable expressivity of these disorders, a genetic diagnosis might be useful in predicting an individual’s susceptibility to a wide range of oral and systemic conditions.
REFERENCES
- Suzuki J, Aoyama N, Izumi Y, Isobe M, Komuro I, Hirata Y. Effect of periodontitis on cardiovascular manifestations in Marfan syndrome. Critical common role of TGF-β. Int Heart J. 2015;56:121–124.
- Ohshima M, Yamaguchi Y, Matsumoto N, et al. TGF-β signaling in gingival fibroblast-epithelial interaction. J Dent Res. 2010;89:1315–1321.
- Shiga M, Saito M, Hattori M, et al. Characteristic phenotype of immortalized periodontal cells isolated from a Marfan syndrome type I patient. Cell Tissue Res. 2008;33:461–472.
- Suda N, Shiga M, Ganburged G, Moriyama K. Marfan syndrome and its disorder in periodontal tissues. J Exp Zool B Mol Dev Evol. 2009;312B:503–509.
- Rahman N, Dunstan M, Teare MD, et al. Ehlers-Danlos syndrome with severe early-onset periodontal disease (EDS-VIII) is a distinct, heterogeneous disorder with one predisposition gene at chromosome 12p13. Am J Hum Genet. 2003;73:198–204.
- Mao JR, Bristow J. The Ehlers-Danlos syndrome: on beyond collagens. J Clin Invest. 2001;107:1063–1069.
- Schalkwijk J, Zweers MC, Steijlen PM, et al. A recessive form of the Ehlers-Danlos syndrome caused by tenascin-X deficiency. N Engl J Med. 2001;345:1167–1175.
- Ragunatha S, Ramesh M, Anupama P, Kapoor M, Bhat M. Papillon-Lefèvre syndrome with homozygous nonsense mutation of cathepsin C gene presenting with late-onset periodontitis. Pediatr Dermatol. 2015;32:292–294.
- Preus HR. Treatment of rapidly destructive periodontitis in Papillon-Lefèvre syndrome. Laboratory and clinical observations. J Clin Periodontol. 1988;15:639–643.
- Javed F, Ramalingam S, Ahmed HB, et al. Oral manifestations in patients with neurofibromatosis type-1: a comprehensive literature review. Crit Rev Oncol Hematol. 2014;9:123–129.
- Ghalayani P, Saberi Z, Sardari F. Neurofibromatosis type I (von Recklinghausen’s disease): A family case report and literature review. Dent Res J (Isfahan). 2012;9:483–488.
- Shapiro SD, Abramovitch K, van Dis ML, et al. Neurofibromatosis: Oral and radiographic manifestations. Oral Surg Oral Med Oral Pathol. 1984;58:493–498.
- Kamer AR, Craig RG, Dasanayake AP, Brys M, Glodzik-Sobanska L, de Leon MJ. Inflammation and Alzheimer’s disease: possible role of periodontal diseases. Alzheimers Dement. 2008;4:242–250.
- Abbayya K, Puthanakar NY, Naduwinmani S, Chidambar YS. Association between periodontitis and Alzheimer’s Disease. N Am J Med Sci. 2015;7:241–246.
- Rolim Tde S, Fabri GM, Nitrini R, et al. Evaluation of patients with Alzheimer’s disease before and after dental treatment. Arq Neuropsiquiatr. 2014;72:919–924.
From Decisions in Dentistry. May 2020;6(5):25–28.




