Head and Neck Lesions Associated With the Human Papillomavirus
An examination of risk factors, modes of transmission, and prevention and management strategies for these common viral infections.
This course was published in the July 2022 issue and expires July 2025. The author has no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Describe various types of human papillomavirus (HPV) infection, its prevalence, and clinical features of HPV-induced lesions that occur in the head and neck.
- Discuss the risks of transmission, and modes of transmission, for HPV.
- Identify preventive measures that can help reduce HPV
infection rates.
BENIGN LESIONS
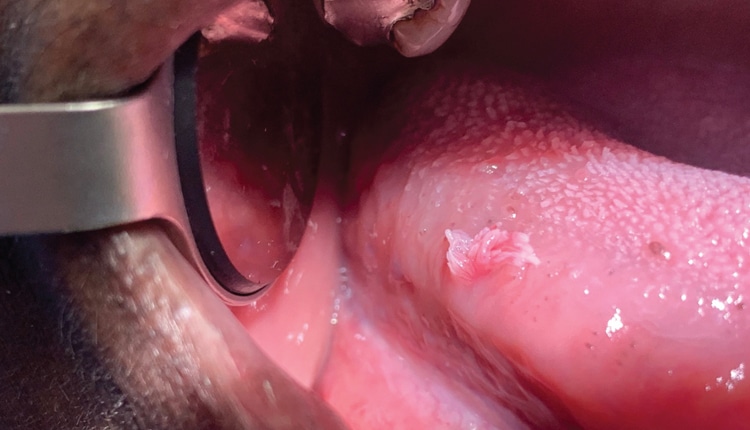
Squamous Papilloma: This benign exophytic (growing outward from the surface) lesion presents with a papillary or fingerlike surface architecture and color ranging from pink to white (Figure 1). It can occur anywhere in the oral cavity, but is frequently found as a solitary lesion on the tongue, palate or lips.1,5 The lesion is usually pedunculated, but it can be sessile. Squamous papilloma can occur at any age, but the average age on onset is between 30 and 50.6 This infection is most often associated with low-risk HPV types 6 and 11 (Table 1).6,7 Diagnosis is made by biopsy and histologic examination. While most cases are treated with surgical excision by laser, additional treatment modalities include electrocautery, cryosurgery, and intralesional injections of interferon.6 Except for lesions in patients infected with human immunodeficiency virus, recurrence is rare.8
Verruca Vulgaris: This common wart is primarily found on the hands. Its clinical presentation is similar to a papilloma with an exophytic papillary appearance (Figure 2). The lesions are sessile and can appear pink to white. While rarely found on the mucosa, it can be transferred to the vermillion border, gingiva or anterior tongue through autoinoculation from the hands.1,5,9 The virus affects children and young adults, with a peak incidence in those age 12 to 16.7 Verruca vulgaris is most frequently associated with HPV low-risk types 2 and 4 (Table 1).7 Diagnosis is made by biopsy and histologic examination, with topical salicylic acid or liquid nitrogen a common treatment approach.1 For lesions occurring intraorally, management may include surgical excision or removal using lasers, cryotherapy or electrosurgery.
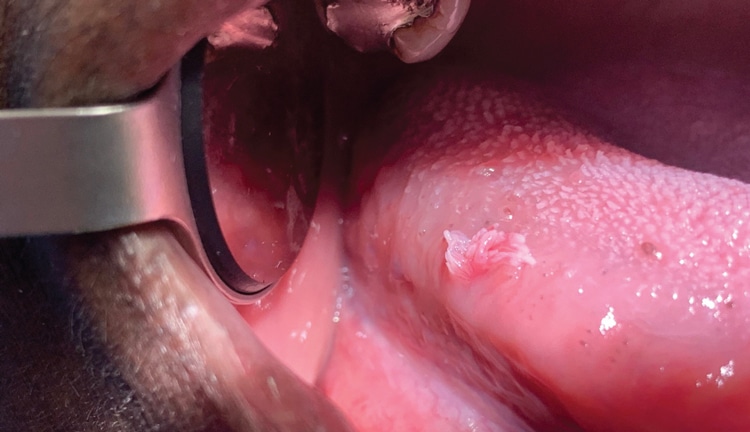
Condyloma Acuminatum: This venereal wart primarily occurs in the anogenital region. It is the most commonly reported sexually transmitted infection in the United States7 and occurs in approximately 1% of sexually active individuals.1 Condyloma acuminatum presents as exophytic pink nodules, with short, blunt surface projections (Figure 3). Common oral locations include the labial mucosa, lingual frenum and soft palate.1,7 While uncommon in the oral cavity, the lesion can be transferred to the mouth through orogenital contact or self-inoculation.5 These lesions are more commonly seen in adults ages 30 to 40.7 Low-risk HPV types 6 and 11 contribute to 90% of condyloma acuminatum cases,1 and many of these patients are coinfected with high-risk HPV types 16 and 18 (Table 1). Diagnosis is made by biopsy and histologic examination, with treatment via surgical excision.
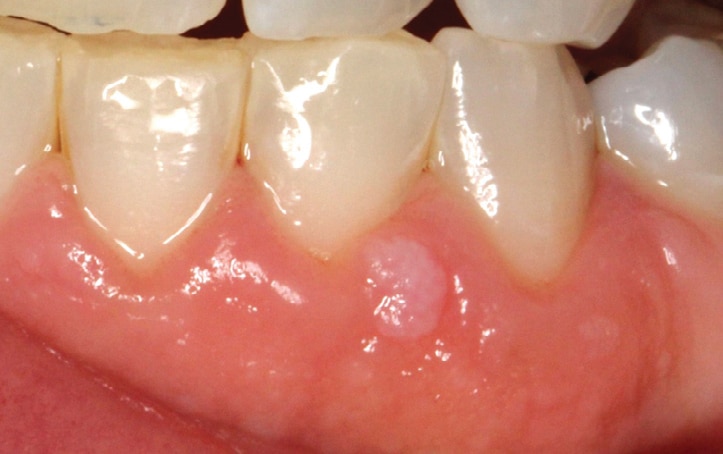
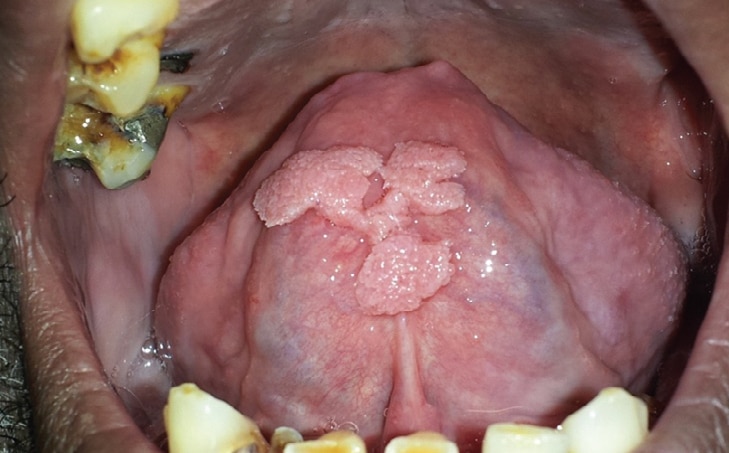
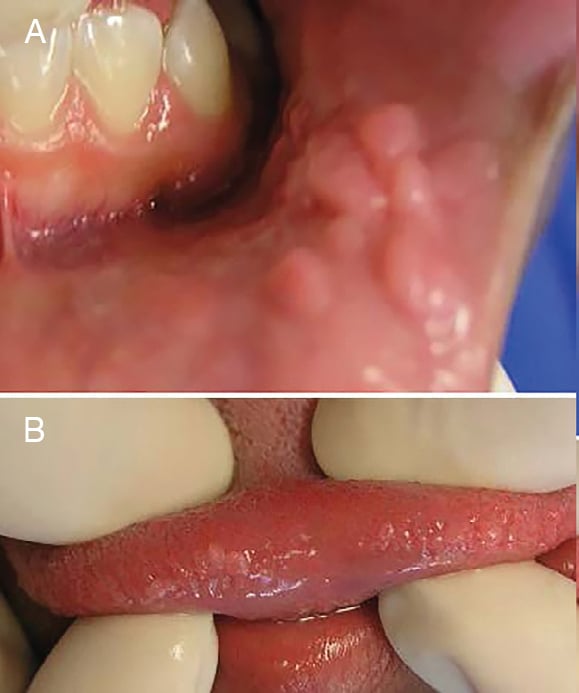
Heck Disease: Also called multifocal epithelial hyperplasia, Heck disease is a benign condition that presents as solitary nodular lesions or multiple whitish to pale pink masses occurring primarily on the buccal and labial mucosa and tongue (Figures 4A and 4B).1,7,10 The lesions can present as smooth surface nodules or exhibit a pebbly surface texture.1 Genetics is believed to play a role in the development of this condition, as many with the infection test positive for the human lymphocytic antigen DR4.7,11 Multifocal epithelial hyperplasia can occur at any age, but presents most commonly in children and adolescents.1,10 Heck disease is associated with low-risk HPV types 13 and 32 (Table 1).7,11 These lesions are asymptomatic and rarely require treatment, as they have been reported to regress.1,5 Surgical excision is the treatment modality for longstanding lesions.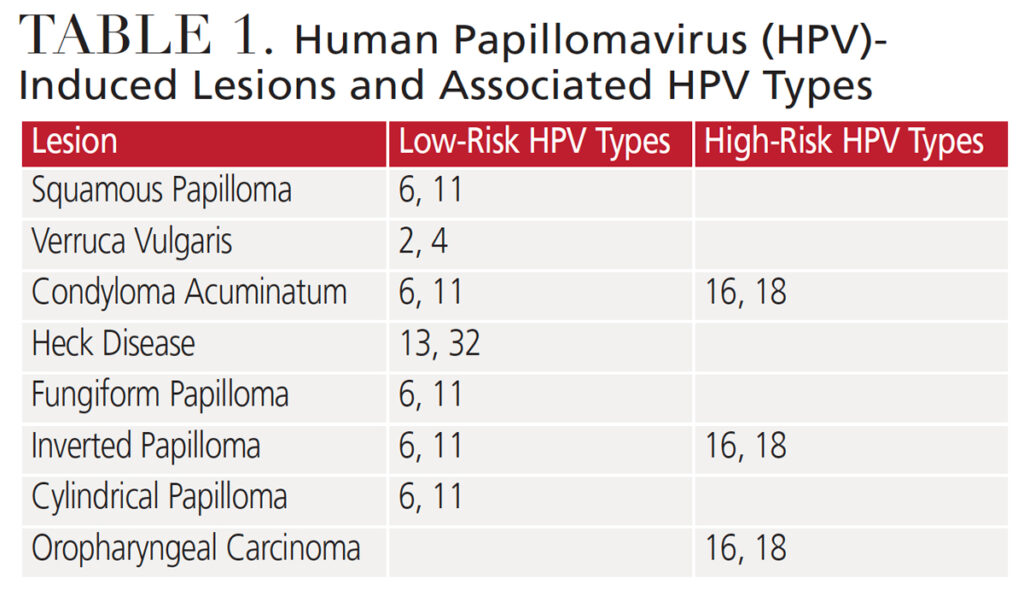
Sinonasal Papillomas: These benign growths arise from the sinus and nasal mucosa. They can develop in various locations, including the nasal septum, lateral walls of the nasal cavity, and maxillary and ethmoid sinuses. Comprising 10% to 25% of all tumors found in this region, their etiology is not well understood, but a history of allergies, sinusitis and nasal polyps has been suggested.1 Infection with HPV is also a possibility. Syrjänen and Syrjänen12 reported that 39% of sinonasal papillomas studied were positive for HPV. Based on clinical and histological features, papillomas are classified as fungiform, inverted or cylindrical.
Fungiform papillomas present as a pinkish nodule, with a papillary surface architecture similar to squamous papillomas. These growths arise on the nasal septum and occur more often in men than women. The most common presenting symptom is nasal obstruction or bleeding.1 Most cases test positive for HPV 6 or 11 (Table 1). Treatment involves surgical excision.
The inverted papilloma is the most common form and presents with significant clinical behavior, as it can be locally destructive and extend to surrounding structures. Approximately 3% to 24% of cases become malignant.1 Similar to the fungiform papilloma, the inverted papilloma presents as a pink nodule, but arises on the lateral nasal walls. Occurring more often in men, inverted papilloma may cause nasal obstruction; symptoms include bleeding, headaches, and a compromised olfactory sense. The most prevalent viral types identified are HPV 6, 11, 16 and 18 (Table 1). The treatment of choice is transnasal endoscopic surgery, which may be combined with an external surgical approach in aggressive cases.1
The cylindrical papilloma is the least common and presents as a red or brown nodule with a pebbly surface texture. It arises on the lateral nasal walls, ethmoid or maxillary sinuses. Nasal obstruction is the most common presenting symptom. Though prevalence of HPV is low in cylindrical papilloma, HPV types 6 and 11 are the forms most often detected (Table 1). Treatment involves transnasal endoscopic surgery or an external surgical approach.1
MALIGNANT LESIONS
Oropharyngeal Squamous Cell Carcinoma: This subset of squamous cell carcinoma occurs in the head and neck. It has a predilection for the pharyngeal walls, soft palate, base of the tongue, and tonsillar region. Approximately 70% to 80% of cases occur in the tonsillar region,1 a location that makes diagnosis more challenging. A definitive diagnosis is often delayed until the disease is advanced. Symptoms include a prolonged sore throat, dysphagia, and pain upon swallowing.1 Due to the delay in diagnosis, lesions are commonly greater in size than carcinomas that present in the oral cavity, and they are more likely to have metastasized.1
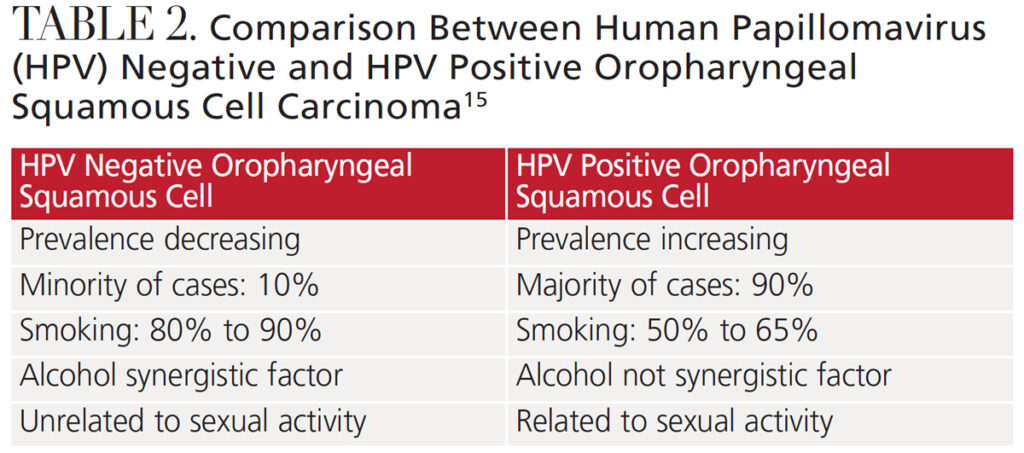
The primary causes for most cancers in the head and neck are smoking and alcohol use.13 Over the past two decades, the incidence of oropharyngeal squamous cell carcinoma has been increasing, particularly in men. This increase is associated with HPV and sexual behavior. Approximately 72% of oropharyngeal carcinomas are caused by HPV, with 62% induced by the high-risk types HPV 16 and 18.4 In addition, HPV type 16 has been identified in more than 90% of oropharyngeal squamous cell carcinoma cases.13 The prevalence of non-HPV-associated cases has been decreasing, and smoking and alcohol use are more significant in these cases compared to HPV-associated disease (Table 2).14,15
In early cases, the treatment of oropharyngeal squamous cell carcinoma involves surgery or radiation therapy. For advanced disease, a combination of surgery, radiation and chemotherapy is used. Determination of active HPV infection is imperative, as HPV-positive oropharyngeal squamous cell carcinoma has a better prognosis than non-HPV oropharyngeal squamous cell carcinoma.1,14,15
DIAGNOSIS AND DISCUSSION
Lesions induced by HPV are diagnosed by biopsy and microscopic evaluation. Oral lesions identified upon examination should be referred to a dentist, oral pathologist or oral surgeon for evaluation and biopsy. Although benign lesions do not generally recur, follow-up at subsequent visits is recommended. Cases of oral pharyngeal squamous cell carcinoma require a multidisciplinary approach that includes the expertise of otolaryngologists, head and neck surgeons, and oncologists.
Transmission of HPV occurs via skin or mucosal contact. The ease of infectivity is attributed to these viruses’ affinity for human skin and mucosa, as HPV invades the most superficial layers of the epithelium, making it more likely to avoid the body’s immune surveillance system. On contact, microtrauma occurs to the epithelium, allowing the virus to easily enter and infect epithelial cells.16
The virus can be transmitted sexually and nonsexually through skin-to-skin, skin-to-mucosa, or mucosa-to-mucosa contact with an infected person. Horizontal transmission occurs via contact with mucosa, skin, fingers, mouth and objects. The virus has been reported to live on surfaces for several days.7,16 Vertical transmission occurs between a mother and child either during pregnancy through the placenta or during natural delivery by contact with genital mucosa.16,17
Self-inoculation is another mode of transmission, as patients can transmit the virus from one part of the body to another. This is particularly true with young children. When lesions are noted in children, the mode of transmission warrants investigation by a healthcare provider. Auto inoculation from skin warts to genital areas can occur. However, the presence of anogenital-type warts in children should prompt further evaluation to rule out sexual abuse.18
 According to the CDC, approximately 14 million people become infected with HPV each year.3 The risk for infection is high, with 80% of women and 90% of men becoming infected with at least one type of HPV during their lifetimes.16 Most people are asymptomatic when infected and will clear the virus within one to two years without any sequela.1–3 In a certain percentage of people, the virus will persist, however, and, depending on the type, can lead to benign and/or malignant lesions.12 An impaired immune system, older age, and sexual behavior are linked with persistent infections.1
According to the CDC, approximately 14 million people become infected with HPV each year.3 The risk for infection is high, with 80% of women and 90% of men becoming infected with at least one type of HPV during their lifetimes.16 Most people are asymptomatic when infected and will clear the virus within one to two years without any sequela.1–3 In a certain percentage of people, the virus will persist, however, and, depending on the type, can lead to benign and/or malignant lesions.12 An impaired immune system, older age, and sexual behavior are linked with persistent infections.1
While lesions can develop with any of the HPV types, the risk of oral pharyngeal squamous cell carcinoma is the most significant concern. Even though the prevalence for HPV-induced oropharyngeal carcinoma has increased over the past 20 years, the risk remains low. The overall prevalence of HPV in the oral cavity is approximately 6.9%.5 A five-year study conducted by D’Souza et al13 revealed the incidence of HPV oral infections was 6% for men and 1% for women across all age groups. The prevalence for men ages 50 to 59 was slightly higher at 8.1%, while the prevalence was lower for oral HPV 16. Engaging in oral sex with an increased number of partners also showed a connection with higher infections in men than women.13
As the leading cause of sexually transmitted disease in the United States,2–4 HPV infection risk increases for those who have multiple sex partners, unprotected sex, adolescent start of sexual activity, and oral sex. More than 85% of sexually active Americans ages 18 to 44 report having had oral sex at least once with a partner of the opposite sex.3 Smoking is another risk factor due to its systemic and immunosuppressive effects. The prevalence of HPV 16 is higher among tobacco users.15 D’Souza et al13 reported that smoking doubled the prevalence of HPV infections in men (Table 3).
Currently, there is no treatment for HPV infections. The most effective mitigation strategy is to decrease infection rates via prevention. Sexual activity is the most significant causative factor. Participating in low-risk sexual activities, such as those that do not involve the exchange of bodily fluids or direct contact between mucus membranes with another person, is an effective way to avoid infection.19 Other preventive strategies include monitoring health status (e.g., undergoing regular Pap tests and pelvic exams for women, and being regularly tested for HPV). Limiting sexual activity to a monogamous relationship significantly diminishes exposure to infection.20 Another way to mitigate risk is to use condoms and other barriers, such as dams, when engaging in sexual activity.19
Another highly effective preventive strategy to not only reduce HPV transmission but also prevent cancers is vaccination. Most HPV vaccines protect against the viral strains frequently associated with cancer (HPV types 16 and 18) and genital warts (HPV types 6 and 11). Currently, the only vaccine distributed in the United States is Gardasil 9, a nine valent formula that protects against HPV types 6, 11, 16, 18, 31, 33, 45, 52 and 58.3 The CDC reports that since HPV vaccination was first recommended in 2006, there has been a significant reduction in HPV infections, but the number of preventable cancer cases remains high.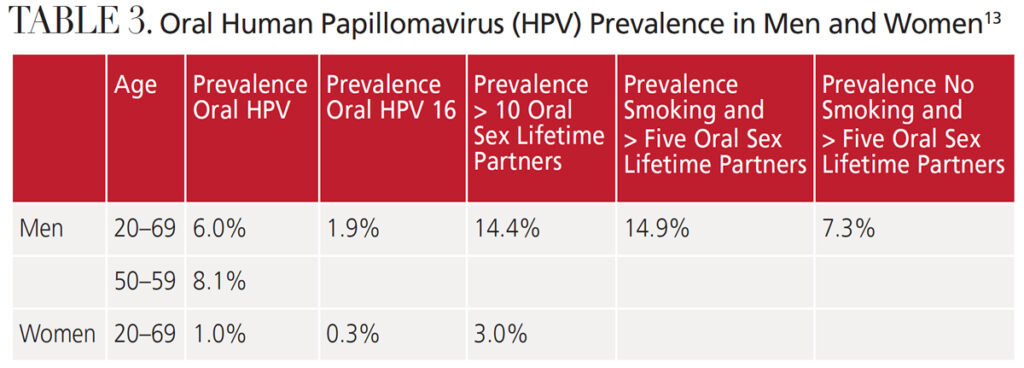
The CDC calculates the number of HPV-associated cancers (a specific type of cancer that is diagnosed in a part of the body where HPV is often found, including the cervix, vagina, vulva, penis, anus and oropharynx) to estimate the percentage of HPV-attributable cases. Such cases are estimated by multiplying the number of HPV-associated cases by the percentage attributable to HPV based on genotype studies.21 Using data from 2014 to 2018, the CDC estimates 79% of these cancers were attributable to HPV each year. Out of these cases, approximately 92% could have been prevented with the HPV vaccine.21
Inoculation has proven most effective if administered prior to exposure to the virus.4,12 The CDC recommends vaccination for children at ages 11 to 12, but advises the vaccine can be administered as early as age 9. Vaccination is also recommended for individuals up to age 26 if they were not vaccinated at a younger age. Routine vaccination is not recommended for those older than 26. For individuals ages 27 to 45, vaccination should be based on risk, and determined by medical guidance from a physician.
As healthcare providers, dentists play an integral role in disease prevention and patient education — not only in clinical practice as part of the oral cancer examination, but also by participating in community outreach events. These settings allow clinicians to educate patients about strategies to mitigate risks of infection, and advocate for the benefits of HPV vaccination.
CONCLUSION
As noted, HPV infections are common and can lead to benign and malignant lesions. Although some lesions possess shared viral subtypes, they differ in clinical presentation and prognosis. To adequately diagnose and treat these conditions, clinicians should be familiar with the various clinical presentations and patient demographics. Due to the possible sexual transmissibility of some of these lesions, patient education is critical to help limit infections. Although vaccination has proven effective in reducing infection rates, patients should be educated on the value of vaccination and the relationship between HPV and behaviors such as smoking and sexual activity.
REFERENCES
- Neville BW, Damm DD, Allen CM, Bouquot JE. Oral and Maxillofacial Pathology. 4th ed. St. Louis: Saunders Elsevier; 2016:331–340, 378–372.
- National Cancer Institute. HPV and Cancer. Available at: https://www.cancer.gov/about-cancer/causes-prevention/risk/infectious-agents/hpv-and-cancer. Accessed May 27, 2022.
- U.S. Centers for Disease Control and Prevention. HPV, the Vaccine for HPV, and Cancers Caused by HPV. Available at: https://www.cdc.gov/hpv/index.html. Accessed May 27, 2022.
- Vázquez-Otero C, Vamos CA, Thompson EL, et al. Assessing dentists’ human papillomavirus-related health literacy for oropharyngeal cancer prevention. J Am Dent Assoc. 2018;149:9–17.
- Ibsen OA, Phelan JA. Oral Pathology for the Dental Hygienist. 7th ed. St. Louis: Saunders Elsevier; 2014:125–126.
- Babaji P, Singh V, Chaurasia VR, Masamatti VS, Sharma AM. Squamous papilloma of the hard palate. Indian J Dent. 2014;5:211–213.
- Betz SJ. HPV-related papillary lesions of the oral mucosa: a review. Head and Neck Pathol. 2019;13:80–90.
- Jaju PP, Suvarna PV, Desai RS. Squamous papilloma: case report and review of literature. Int J Oral Sci. 2010;2:222–225.
- Ural A, Arslan S, Ersoz Ş, Değer B. Verruca vulgaris of the tongue: a case report with a literature review. Bosn J Basic Med Sci. 2014;14:136–138.
- Regezi JA, Sciubba JI, Jordan RC. Oral Pathology Clinical Pathologic Correlations. 7th ed. St. Louis: Saunders Elsevier; 2017:148–152.
- Bennett LK, Hinshaw M. Heck’s disease: diagnosis and susceptibility. Pediatr Dermatol. 2009;26:87–89.
- Syrjänen K, Syrjänen S. Detection of human papillomavirus in sinonasal papillomas: systematic review and meta-analysis. Laryngoscope. 2013;123:181–192.
- D’Souza G, McNeel TS, Fakhry C. Understanding personal risk of oropharyngeal cancer: risk-groups for oncogenic oral HPV infection and oropharyngeal cancer. Ann Oncol. 2017;28:3065–3069.
- Rettig EM, D’Souza G. Epidemiology of head and neck cancer. Surg Oncol Clin N Am. 2015;24:379–396.
- Kalmar J. Oral and Tonsillar Cancer: What About HPV? Lecture presented at: Thomas P. Hinman Dental Meeting; March 31, 2019; Atlanta.
- Petca A, Borislavschi A, Zvanca ME, Petca RC, Sandru F, Dumitrascu MC. Non-sexual HPV transmission and role of vaccination for a better future (Review). Exp Ther Med. 2020;20:186.
- Syrjanen S, Puranen M. Human papillomavirus infections in children: the potential role of maternal transmission. Crit Rev Oral Biol Med. 2000;11:259–274.
- Cao CD, Merjanian L, Pierre J, Balica A. A discussion of high-risk HPV in a 6-year-old female survivor of child sexual abuse. Case Rep Obstet Gynecol. 2017;2017:6014026.
- John Hopkins Medicine. Safer Sex Guidelines. Available at: https://www.hopkinsmedicine.org/health/wellness-and-prevention/safer-sex-guidelines. Accessed May 27, 2022.
- Stanley M. Prevention strategies against the human papillomavirus: the effectiveness of vaccination. Gynecol Oncol. 2007;107(2 Suppl 1):S19–S23.
- U.S. Centers for Disease Control and Prevention. Cancers Associated With Human Papillomavirus, United States — 2014–2018. Available at: https://www.cdc.gov/cancer/uscs/about/data-briefs/no26-hpv-assoc-cancers-UnitedStates-2014-2018.htm. Accessed May 27, 2022.
From Decisions in Dentistry. July 2022;8(7)40-43.




